BLOOD GAS INTERPRETATION Jose Gaudreault APN Montreal Childrens

BLOOD GAS INTERPRETATION Josée Gaudreault, APN Montreal Children’s Hospital Mc. Gill University Health Center

Why do we do blood gases?

Why do we do blood gases? Assessment of oxygenation Assessment of ventilation Assessment of acid-base balance

Few definitions Equilibrium is achieved at a normal p. H Acid substance able to give up hydrogen ions during chemical exchanges Base substance able of accepting hydrogen ions during chemical exchanges

Types of BG Venous No oxygen value Affected by local circulatory and metabolic environment Capillary § Accurate p. CO 2 and p. H § Avoid excessive squeezing – why? § Free flowing blood. Arterial Gold standard

Information from BG p. H p. CO 2 p. O 2 Bicarbonate (HCO 3) Lytes, glucose

p. H § Estimates total blood acid load (mostly reflects dissolved CO 2, but may also include metabolic acids such as lactic acid) § - log of [H+] ; logarithmic relationship (not linear) § Change in p. H of 1 = 10 x change in H+ § Small change in [H+ ] = fatal § Low p. H = acidic solution, high p. H = basic solution § p. H, p. CO 2 and HCO 3 give information about effectiveness of regulatory mechanisms of lungs and kidneys

CO 2 + H 2 O <-> H 2 CO 3 <-> HCO 3 CO 2 = respiratory acid removed by lungs HCo 3 - = basic buffer (neutralizes acids), produced by kidneys

p. H, p. CO 2 & HCO 3 determine the primary problem!!!

p. O 2 is carried by Hgb and plasma pa. O 2 = Measure of how much oxygen is delivered to the tissues Arterial >> capillary >> venous Represents oxygenation; regulated by lungs Can be correlated with oxygen saturation as measured by pulse oximeter

p. CO 2 Represents ventilation; regulated by lungs CO 2 is a by-product of cell metabolism Diffuses easily in the blood (as carbonic acid) Accumulates in blood when ventilation is impaired

- Bicarbonate (HCO 3 ) Base Most important buffer in blood (can accept 2 H+ for every molecule of HCO 3) Controlled at the level of the kidneys Keeps p. H within a tight range GI losses below the pylorus = loss of bicarbonate ion
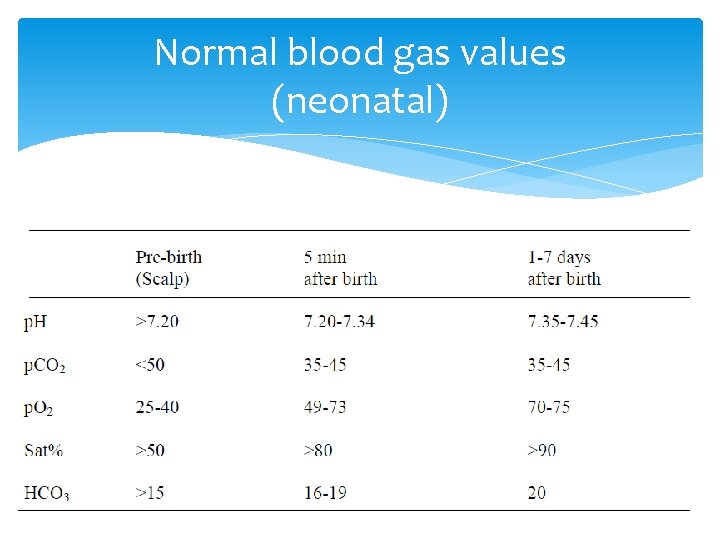
Normal blood gas values (neonatal)

Normal blood gas values ABG Normal value p. H p. O 2 p. CO 2 HCO 3 Base excess 7. 35 -7. 45 80 -100 35 -45 22 -26 -2 to +2

Back to the buffer systems 1. Lungs regulate p. CO 2 by resp rate. It is a quick response 2. Kidneys excrete H+, bicarbonate, and ammonium (NH 4+). It is a slow compensatory mechanism

Compensation Restoration of p. H to near normal levels with resp or renal buffer system Never results in « natural » overcorrection (we cause it) Example: respiratory acidosis p. H 7. 30, CO 2 60: when compensation occurs from the kidney, p. H stays on the acidotic side 7. 40

Acid-Base Imbalances: Four conditions Respiratory acidosis alkalosis Metabolic acidosis alkalosis

Respiratory acidosis Failure of alveolar exchange of CO 2 Common Causes in the NICU: RDS Other resp disorders – collapse, pneumothorax, air leak, pneumonia Choanal atresia ETT blocked / displaced Accumulation of secretions Anxiety/pain

Respiratory alkalosis Hyperventilation Causes: Anxiety/pain Mechanical ventilation – rate, pressures

Metabolic acidosis 3 causes Too many acids exogenous sources metabolic by-products Not enough bases

Common causes: metabolic acidosis Common causes in the NICU: Hypothermia Underperfusion, decreased CO (CHD, shock) Hypoxemia (asphyxia, RDS) Diarrhea (loss of HCO 3 - from bile salts in the stool) Dehydration (hemoconcentration of H+) Renal failure – kidneys cannot resorb bicarb Infection - sepsis - septic shock – why? Administration of solution with high content of chloride (eg Na. Cl) (Clexchanges with HCO 3 - in proximal tubules >> more Cl out of cell = more HCO 3 in) Sodium acetate (Na. CH 3 COO-) – alternate source of bicarb after conversion in liver – often used in TPN as buffer for acidosis

Metabolic alkalosis Common causes in the NICU: Thiazide, loop diuretics: loss of K+ in kidneys Persistent vomiting Gastric suction (unreplaced) – Why do we replace with K+? Urea cycle disorder (accumulation of NH 4+) Loss of K+ activates H+/K+ pump (K+ out of cell for H+ into cell) >> alkalosis Loss of H+ activates H+/K+ exchange pump (H+ out of cell, K+ into cell) – can lead to hypokalemia Compensation results in hypoventilation in body’s attempt to retain CO 2

Blood gas interpretation First, is there a problem? Any number that is out of range? Second, does the patient have an acidosis or an alkalosis? Is the p. H less than 7. 4 or more than 7. 4? Third, what is the primary problem – metabolic or respiratory? – Look at source. Is it your PCO 2 that is out of range, or your HCO 3? Third, is there any compensation by the patient – respiratory compensation is immediate while renal compensation takes time. If p. H is within range of 7. 35 – 7. 45, but other numbers are out of range, compensation has occurred. Fourth, evaluate the effectiveness of oxygenation (only for arterial).

Few exercises…
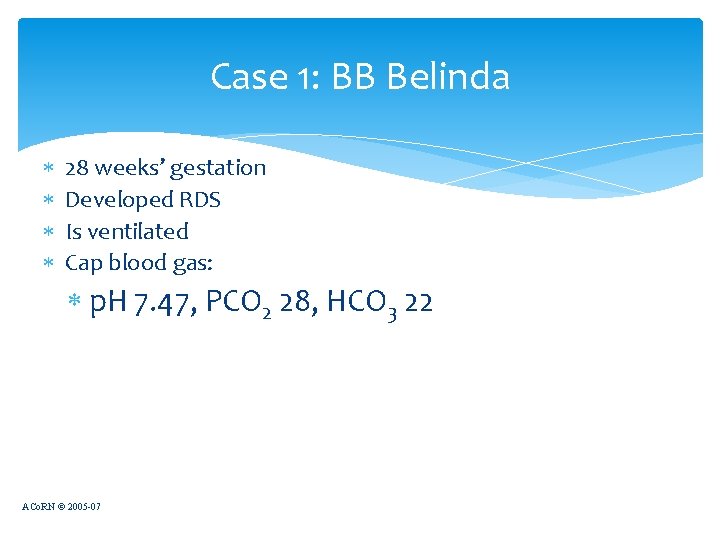
Case 1: BB Belinda 28 weeks’ gestation Developed RDS Is ventilated Cap blood gas: p. H 7. 47, PCO 2 28, HCO 3 22 ACo. RN © 2005 -07

Case 1: Bb Belinda has respiratory alkalosis Excessive ventilation Correct by decreasing ventilator rate decreasing tidal volume or decreasing inspiratory pressure ACo. RN © 2005 -07

Case 2: Bb Jacinthe 14 days, 38 weeks’ gestation, SGA Mother is a substance user Poor feeding and watery stools x 3 days One wet diaper in the last 24 hours Cool to touch and delayed capillary refill Respirations easy but rapid Blood gas: p. H 7. 36, PCO 2 25, HCO 3 12, Lactate 4. 7, Base deficit 10.

Case 2: Bb Jacinthe Compensated metabolic acidosis Fluid loss (diarrhea) plus poor intake dehydration + metabolic acidosis Mechanisms: tissue hypoperfusion lactic acidosis bicarbonate losses in stool respiratory compensation by “blowing off” CO 2 ACo. RN © 2005 -07

Case 3: Billy was delivered by emergency Caesarean delivery for fetal distress / loss of fetal heart beat RR 26, HR 124, his respiratory effort is weak Sp. O 2 90% in 50% oxygen He is hypotonic Blood gas: p. H 7. 16, PCO 2 70, BD 7, HCO 3 21 ACo. RN © 2005 -07

Case 3: Billy had an asphyxial insult. Inadequate respirations high PCO 2 (respiratory acidosis). Tissue hypoxia metabolic acidosis. What needs to be done? ? ? ACo. RN © 2005 -07

Merci!!!
- Slides: 31