Blood Gas Interpretation 2005825 Before beginning Allens test

Blood Gas Interpretation 2005/8/25

Before beginning… Allen’s test for radial and ulnar artery n Common errors of arterial blood sampling n ¨ Air in sample: PCO 2↓, p. H↑, PO 2↨ ¨ Venous mixture: PCO 2↑, p. H↓, PO 2↓ ¨ Excess anticoagulant (dilution): PCO 2↓, p. H↑, PO 2↨ ¨ Metabolic effects: PCO 2↑, p. H↓, PO 2↓ n Simultaneous electrolytes panel
![Acid Base Physiology n The Law of Mass Action K 1 [A] + [B] Acid Base Physiology n The Law of Mass Action K 1 [A] + [B]](http://slidetodoc.com/presentation_image/fd19426160bb443442c9af9e4d70f0fa/image-3.jpg)
Acid Base Physiology n The Law of Mass Action K 1 [A] + [B] [C] + [D] K 2 K 1/K 2 = [C][D]/[A][B] n Dissociation constant for an acid Ka = [H+][A-]/[HA]

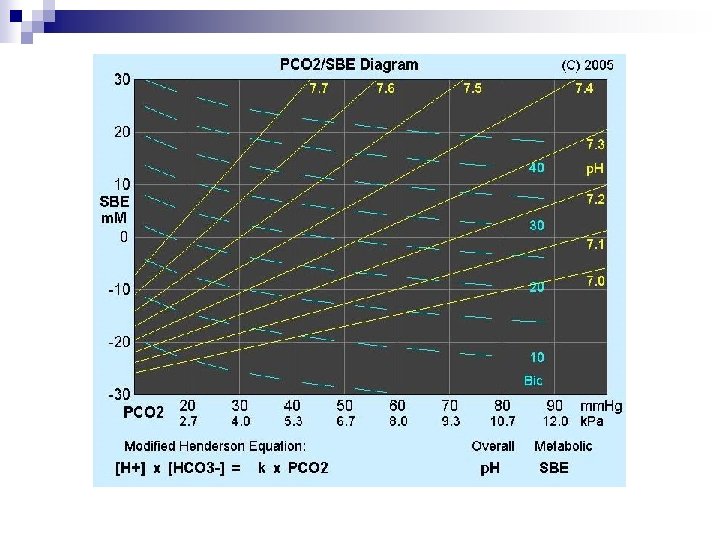
Henderson-Hasselbalch Equation CO 2 + H 2 O H 2 CO 3 H+ + HCO 3[H+] = K x [CO 2]/[HCO 3 -] = 24 PCO 2/[HCO 3 -] p. H = 6. 1 + log ([HCO 3 -]/0. 0301 x. PCO 2)

Normal Range p. H = 7. 35 -7. 45 n PCO 2 = 35 -45 mm. Hg (40 mm. Hg) n HCO 3 - = 22 -26 m. Eq/L (24 m. Eq/L) n
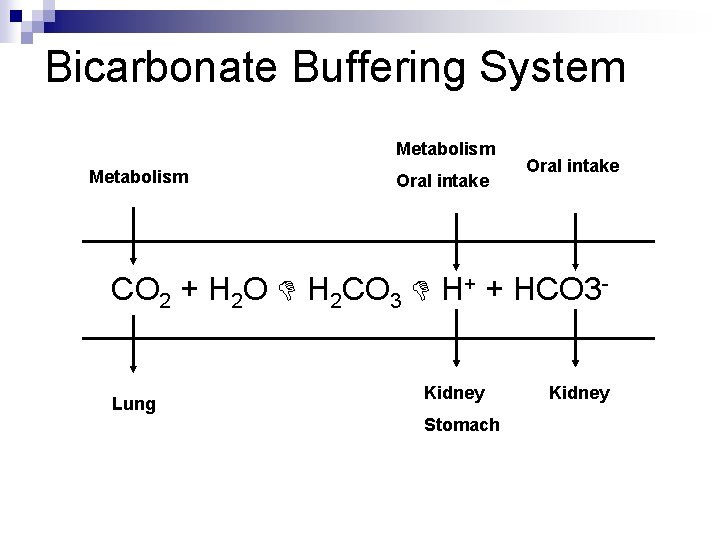
Bicarbonate Buffering System Metabolism Oral intake CO 2 + H 2 O H 2 CO 3 H+ + HCO 3 - Lung Kidney Stomach Kidney
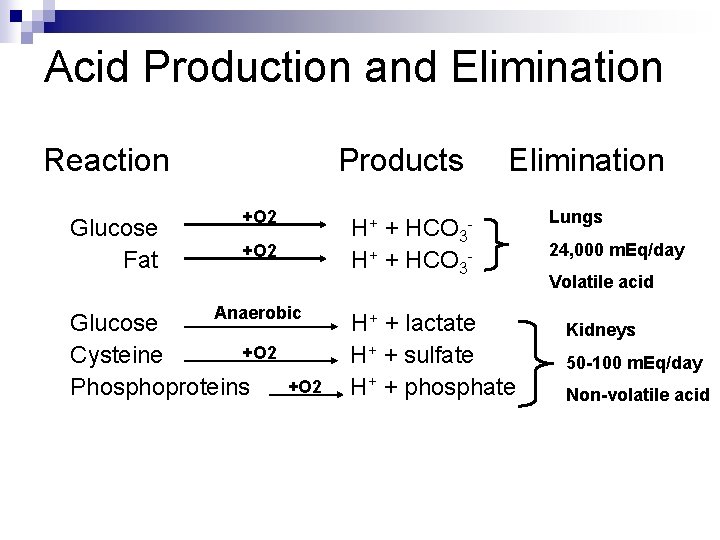
Acid Production and Elimination Reaction Products Elimination +O 2 Glucose H+ + HCO 3+O 2 + + HCO Fat H 3 Anaerobic + + lactate Glucose H + + sulfate +O 2 Cysteine H + + phosphate +O 2 Phosphoproteins H Lungs 24, 000 m. Eq/day Volatile acid Kidneys 50 -100 m. Eq/day Non-volatile acid

Determinants of CO 2 in the alveolus VA = VE – VD = VT x f (1 - VD/VT) PACO 2 = k x (VCO 2/VA) n Physiologic dead space = anatomic dead space + alveolar dead space
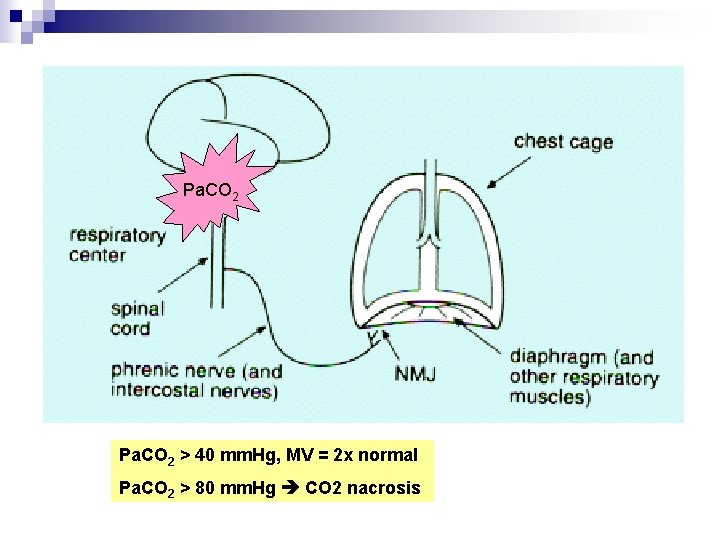
Pa. CO 2 > 40 mm. Hg, MV = 2 x normal Pa. CO 2 > 80 mm. Hg CO 2 nacrosis

Renal Regulation of Bicarbonate “Reabsorption“ of filtered HCO 3 - (4000 mmol/day) n Formation of titratable acid (4000 mmol/day H+) n Excretion of NH 4+ in the urine n 80 -90% of HCO 3 - : reabsorbed in the proximal tubule n Distal tubule: reabsorption of remained bicarbonate and secretion of hydrogen ion n
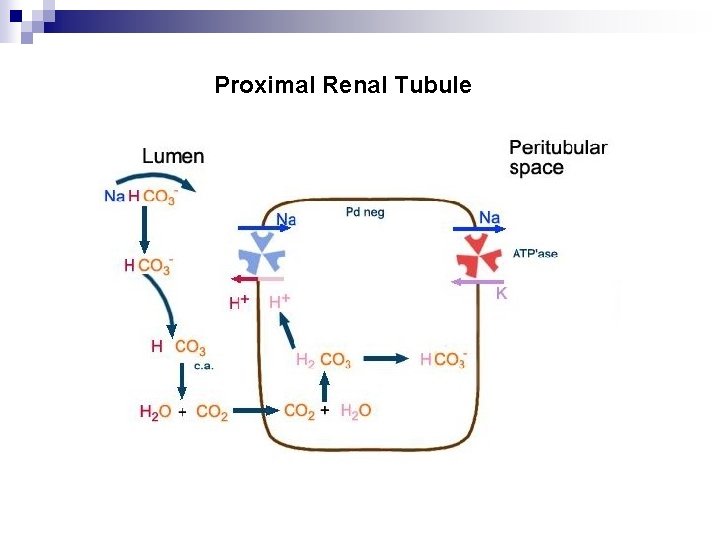
Proximal Renal Tubule
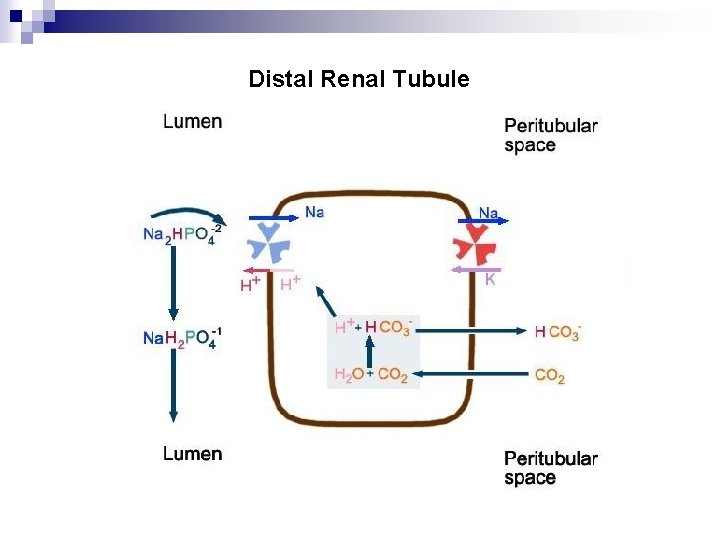
Distal Renal Tubule
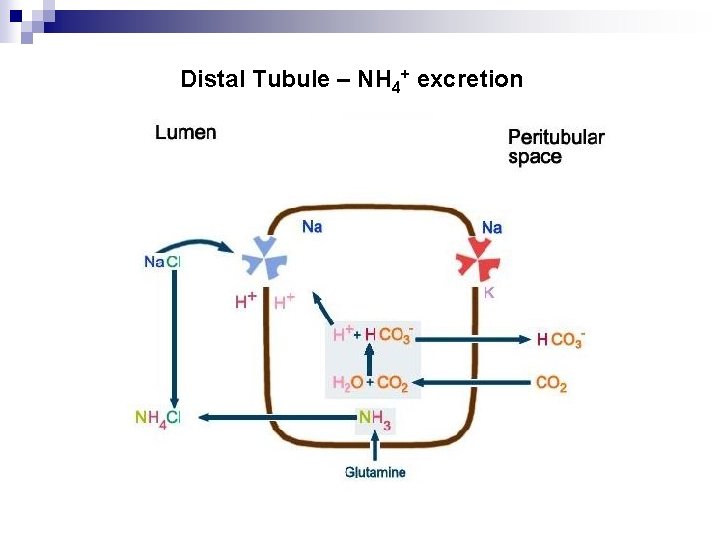
Distal Tubule – NH 4+ excretion

Acid Base Disturbance n n n n Metabolic acidosis: HCO 3 -↓ Metabolic alkalosis: HCO 3 - ↑ Respiratory acidosis: PCO 2↑ Respiratory alkalosis: PCO 2 ↓ Simple Primary Secondary mixed

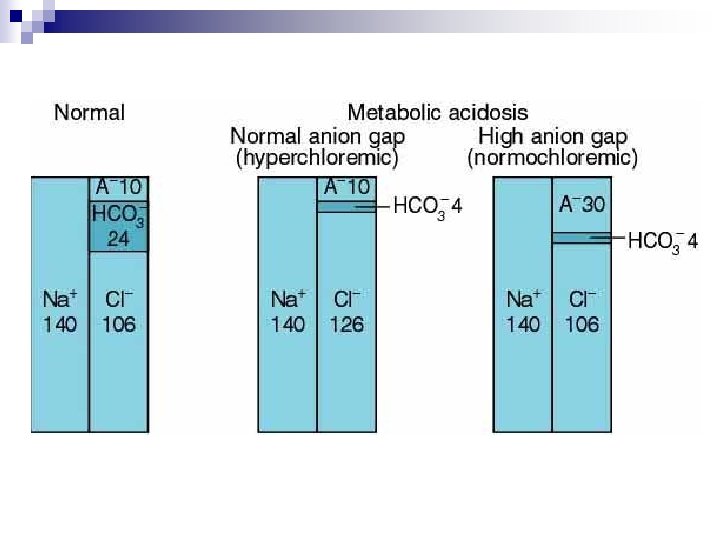
Metabolic Acidosis n n n Indogenous acid production (lactic acidosis, ketoacidosis) Indogenous acid accumulation (renal failure) Loss of bicarbonate (diarrhea) High anion gap Normal (hyperchloremic )

Pathophysiologic Effect of Metabolic Acidosis n n n Kussmaul respiration Intrinsic cardiac contractility↓, normal inotropic function Peripheral vasodilatation Central vasoconstriction pulmonary edema Depressed CNS function Glucose intolerance

Anion Gap AG = Na+ - (Cl- + HCO 3 -) n Unmeasured anions in plasma (normally 10 to 12 mmol/L) n Anionic proteins, phosphate, sulfate, and organic anions n Correction: if albumin < 4 Albumin ↓ 1 AG ↓ 2. 5 n

Anion Gap n Increased unmeasured anions ¨ Decreased unmeasured cations (Ca++, K+, Mg++) ¨ Increase in anionic albumin ¨ n Decrease ¨ ¨ ¨ Increase in unmeasured cations Addition of abnormal cations Reduction in albumin concentration Decrease in the effective anionic charge on albumin by acidosis Hyperviscosity and severe hyperlipidemia ( underestimation of sodium and chloride concentration)

Causes of High-Anion-Gap Metabolic Acidosis Lactic acidosis Toxins Ketoacidosi s Ethylene glycol Diabetic Methanol Alcoholic Salicylates Starvation Renal failure (acute and chronic)

Causes of Non-Anion-Gap Acidosis I. Gastrointestinal bicarbonate loss A. Diarrhea B. External pancreatic or small-bowel drainage C. Ureterosigmoidostomy, jejunal loop, ileal loop D. Drugs 1. Calcium chloride (acidifying agent) 2. Magnesium sulfate (diarrhea) 3. Cholestyramine (bile acid diarrhea) II. Renal acidosis A. Hypokalemia 1. Proximal RTA (type 2) 2. Distal (classic) RTA (type 1) B. Hyperkalemia 1. Generalized distal nephron dysfunction (type 4 RTA) a. Mineralocorticoid deficiency b. Mineralocorticoid resistance c. ØNa+ delivery to distal nephron d. Tubulointerstitial disease e. Ammonium excretion defect III. Drug-induced hyperkalemia (with renal insufficiency) A. Potassium-sparing diuretics (amiloride, triamterene, spironolactone) B. Trimethoprim C. Pentamidine D. Angiotensin-converting enzyme inhibitors and AT-II receptor blockers E. Nonsteroidal anti-inflammatory drugs F. Cyclosporine IV. Other A. Acid loads (ammonium chloride, hyperalimentation) B. Loss of potential bicarbonate: ketosis with ketone excretion C. Expansion acidosis (rapid saline administration) D. Hippurate E. Cation exchange resins
![Metabolic Alkalosis Net gain of [HCO 3 - ] n Loss of nonvolatile acid Metabolic Alkalosis Net gain of [HCO 3 - ] n Loss of nonvolatile acid](http://slidetodoc.com/presentation_image/fd19426160bb443442c9af9e4d70f0fa/image-23.jpg)
Metabolic Alkalosis Net gain of [HCO 3 - ] n Loss of nonvolatile acid (usually HCl by vomiting) from the extracellular fluid n Kidneys fail to compensate by excreting HCO 3 - (volume contraction, a low GFR, or depletion of Cl- or K+) n

Causes of Metabolic Alkalosis I. Exogenous HCO 3 - loads A. Acute alkali administration B. Milk-alkali syndrome II. Effective ECFV contraction, normotension, K+ deficiency, and secondary hyperreninemic hyperaldosteronism A. Gastrointestinal origin 1. Vomiting 2. Gastric aspiration 3. Congenital chloridorrhea 4. Villous adenoma 5. Combined administration of sodium polystyrene sulfonate (Kayexalate) and aluminum hydroxide B. Renal origin 1. Diuretics 2. Edematous states 3. Posthypercapnic state 4. Hypercalcemia/hypoparathyroidism 5. Recovery from lactic acidosis or ketoacidosis 6. Nonreabsorbable anions including penicillin, carbenicillin 7. Mg 2+ deficiency 8. K+ depletion 9. Bartter's syndrome (loss of function mutations in TALH) 10. Gitelman's syndrome (loss of function mutation in Na+-Cl- cotransporter in DCT)

Causes of Metabolic Alkalosis III. ECFV expansion, hypertension, K+ deficiency, and mineralocorticoid excess A. High renin 1. Renal artery stenosis 2. Accelerated hypertension 3. Renin-secreting tumor 4. Estrogen therapy B. Low renin 1. Primary aldosteronism a. Adenoma b. Hyperplasia c. Carcinoma 2. Adrenal enzyme defects a. 11 b-Hydroxylase deficiency b. 17 a-Hydroxylase deficiency 3. Cushing's syndrome or disease 4. Other a. Licorice b. Carbenoxolone c. Chewer's tobacco d. Lydia Pincham tablets IV. Gain of function mutation of renal sodium channel with ECFV expansion, hypertension, K+ deficiency, and hyporeninemic-hypoaldosteronism A. Liddle's syndrome

Respiratory Acidosis Severe pulmonary disease n Respiratory muscle fatigue n Abnormal ventilatory control n Acute vs. Chronic (> 24 hrs) n

Respiratory Acidosis Acute: anxiety, dyspnea, confusion, psychosis, and hallucinations and coma n Chronic: sleep disturbances, loss of memory, daytime somnolence, personality changes, impairment of coordination, and motor disturbances such as tremor, myoclonic jerks, and asterixis n Headache: vasocontriction n

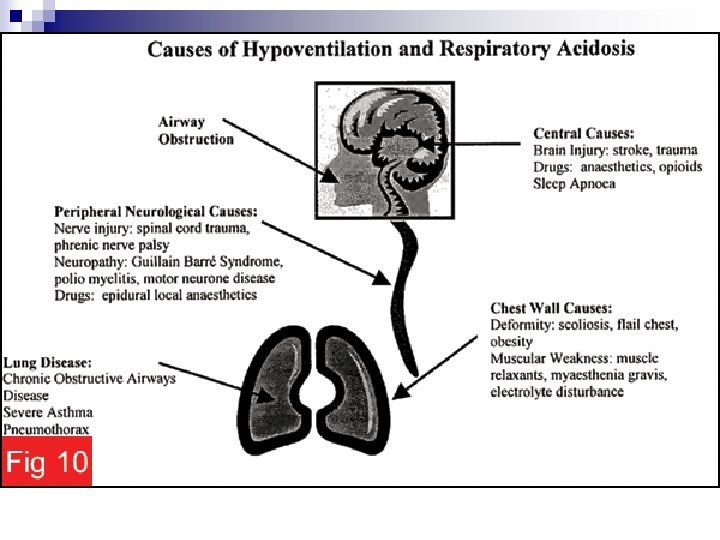
Respiratory Acid-Base Disorders II. Acidosis A. Central 1. Drugs (anesthetics, morphine, sedatives) 2. Stroke 3. Infection B. Airway 1. Obstruction 2. Asthma C. Parenchyma 1. Emphysema 2. Pneumoconiosis 3. Bronchitis 4. Adult respiratory distress syndrome 5. Barotrauma D. Neuromuscular 1. Poliomyelitis 2. Kyphoscoliosis 3. Myasthenia 4. Muscular dystrophies E. Miscellaneous 1. Obesity 2. Hypoventilation 3. Permissive hypercapnia

Respiratory Alkalosis Strong ventilatory stimulus with alveolar hyperventilation n Consuming HCO 3 - n > 2 -6 hrs: renal compensation (decrease NH 4+/acid excretion and bicarbonate reabsorption) n

Respiratory Alkalosis n Reduced cerebral blood flow ¨ dizziness, mental confusion, and seizures n n Minimal cardiovascular effect in normal health Cardiac output and blood pressure may fall in mechanically ventilated patients Bohr effect: left shift of hemoglobin-O 2 dissociation curve tissue hypoxia (arrhythmia) intracellular shifts of Na+, K+, and PO 4 - and reduces free [Ca 2+]

Respiratory Acid-Base Disorders I. Alkalosis A. Central nervous system stimulation 1. Pain 2. Anxiety, psychosis 3. Fever 4. Cerebrovascular accident 5. Meningitis, encephalitis 6. Tumor 7. Trauma B. Hypoxemia or Tissue hypoxia 1. High altitude, ØPa. CO 2 2. Pneumonia, pulmonary edema 3. Aspiration 4. Severe anemia C. Drugs or hormones 1. Pregnancy, progesterone 2. Salicylates 3. Nikethamide D. Stimulation of chest receptors 1. Hemothorax 2. Flail chest 3. Cardiac failure 4. Pulmonary embolism E. Miscellaneous 1. Septicemia 2. Hepatic failure 3. Mechanical hyperventilation 4. Heat exposure 5. Recovery from metabolic acidosis

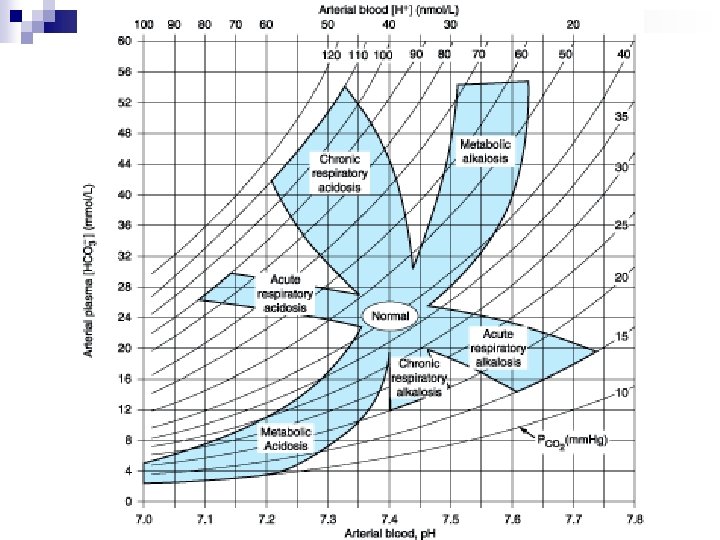
Stepwise Approach n n n Do comprehensive history taking and physical examination Order simultaneous arterial blood gas measurement and chemistry profiles Assess accuracy of data Direction of p. H: always indicates the primary disturbance Calculate the expected compensation Second or third disorders
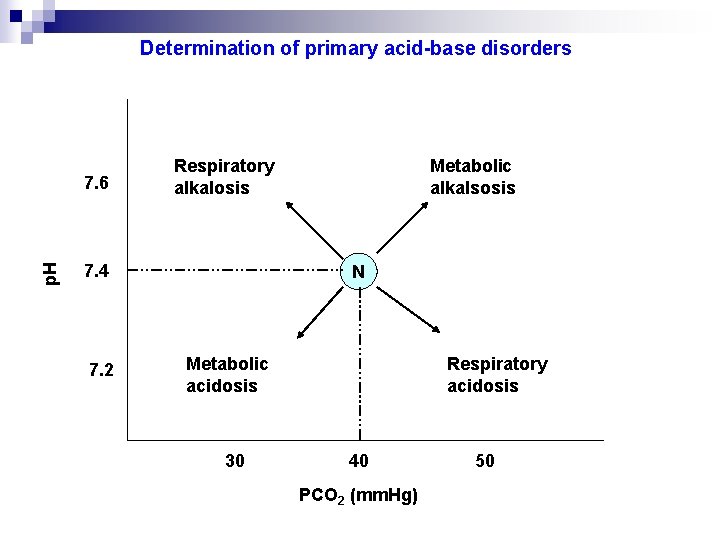
Determination of primary acid-base disorders p. H 7. 6 Respiratory alkalosis 7. 4 7. 2 Metabolic alkalsosis N Metabolic acidosis 30 Respiratory acidosis 40 PCO 2 (mm. Hg) 50
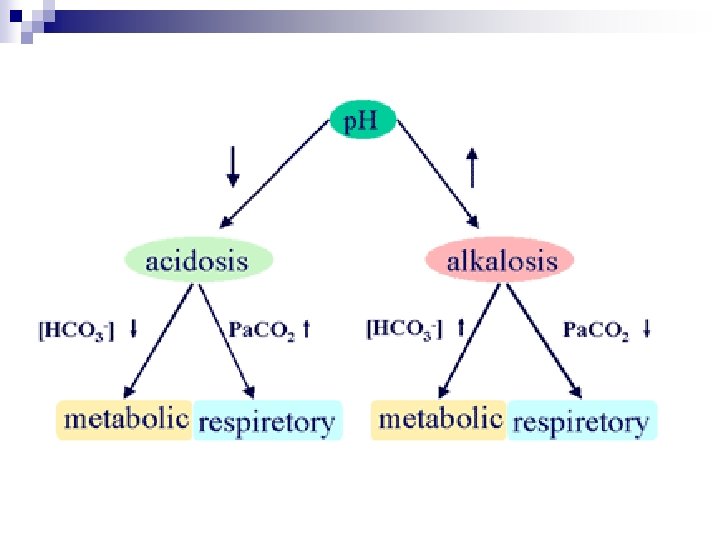

Compensatory Mechanisms n Respiratory compensation ¨ Complete within 24 hrs n Metabolic compensation ¨ Complete within several days n Both the respiratory or renal compensation almost never over-compensates
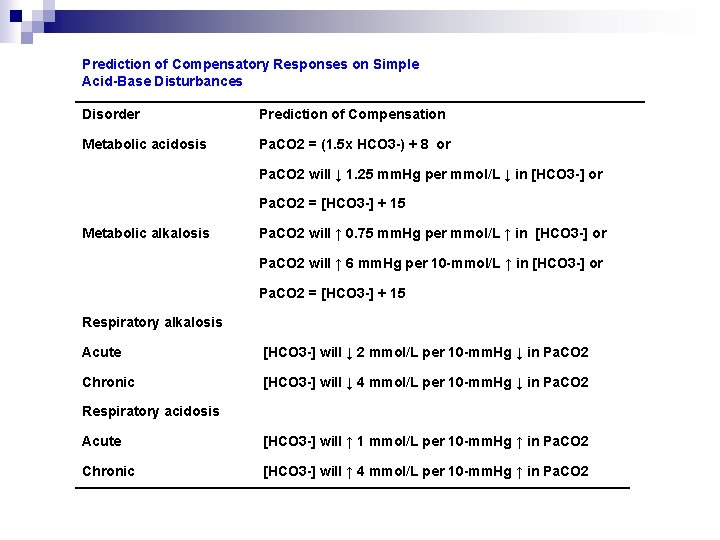
Prediction of Compensatory Responses on Simple Acid-Base Disturbances Disorder Prediction of Compensation Metabolic acidosis Pa. CO 2 = (1. 5 x HCO 3 -) + 8 or Pa. CO 2 will ↓ 1. 25 mm. Hg per mmol/L ↓ in [HCO 3 -] or Pa. CO 2 = [HCO 3 -] + 15 Pa. CO 2 will ↑ 0. 75 mm. Hg per mmol/L ↑ in [HCO 3 -] or Pa. CO 2 will ↑ 6 mm. Hg per 10 -mmol/L ↑ in [HCO 3 -] or Pa. CO 2 = [HCO 3 -] + 15 Metabolic alkalosis Respiratory alkalosis Acute [HCO 3 -] will ↓ 2 mmol/L per 10 -mm. Hg ↓ in Pa. CO 2 Chronic [HCO 3 -] will ↓ 4 mmol/L per 10 -mm. Hg ↓ in Pa. CO 2 Respiratory acidosis Acute [HCO 3 -] will ↑ 1 mmol/L per 10 -mm. Hg ↑ in Pa. CO 2 Chronic [HCO 3 -] will ↑ 4 mmol/L per 10 -mm. Hg ↑ in Pa. CO 2
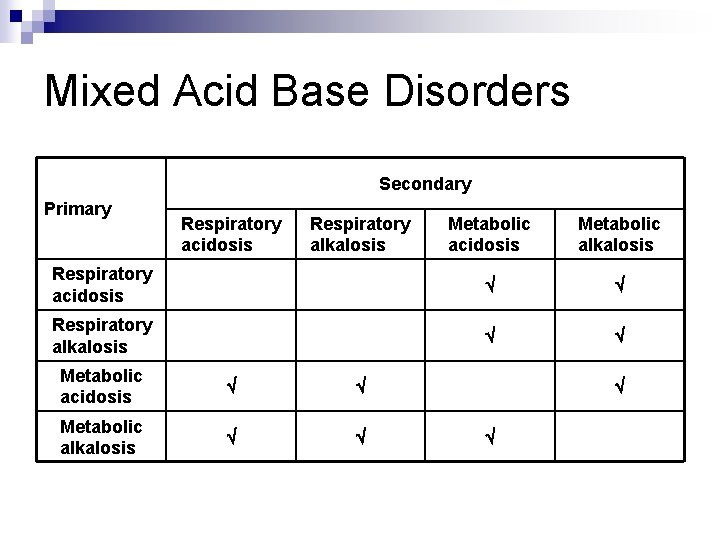
Mixed Acid Base Disorders Secondary Primary Respiratory acidosis Respiratory alkalosis Metabolic acidosis Metabolic alkalosis Respiratory acidosis Respiratory alkalosis Metabolic acidosis Metabolic alkalosis

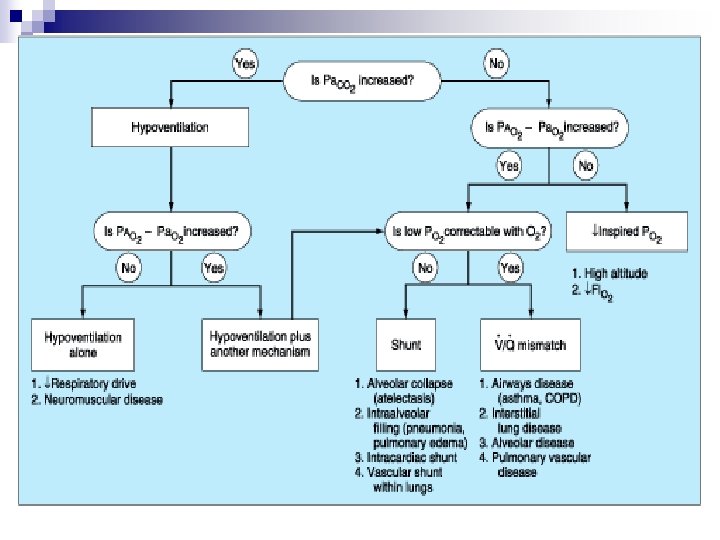
Oxygenation Poor diffusion across alveolar membrane n Small pressure gradient between PAO 2 and Pa. O 2 n Large alveolar area is required for gas transfer n Hemoglobin carries the majority of oxygen in the blood n
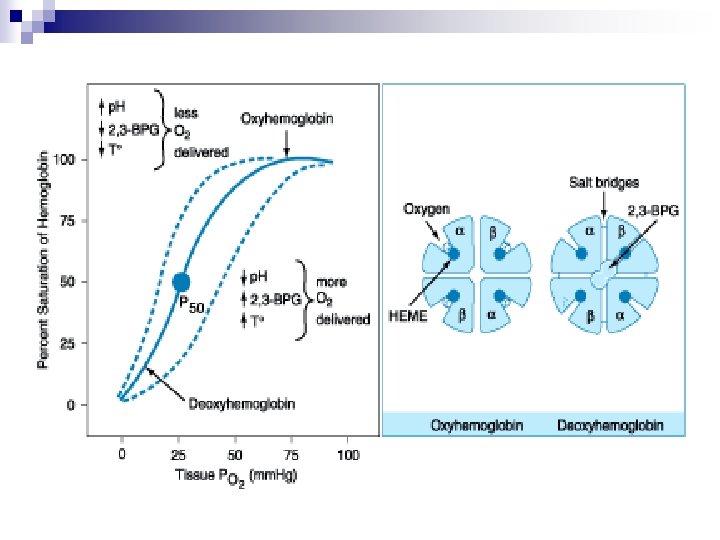

Oxygenation n Ventilation and alveolar disease Ventilation↓ PAO 2 ↓ Pa. O 2 ↓, combined PCO 2↑ Alveolar disease ¨ Reduced alveolar area ¨ Thickened alveolar membrane ¨ V/Q mismatch ¨ Shunt

Alveolar-arterial Oxygen Gradient PAO 2 = Fi. O 2 (PB-PH 2 O) – PCO 2/R = 0. 21(760 -47) – 40/0. 8 = 100 R: respiratory quotient P(A-a)O 2 = PAO 2 – Pa. O 2 (= Age x 0. 4)

Oxygen Content and Saturation O 2 content = 1. 34 x Hb x Saturation + 0. 0031 x. PO 2

Pulse Oximeters n n n Percentage of oxygenated hemoglobin in blood Absorption of light in the red and infra-red spectra Continuous monitor Accurate ( 3%) at high saturation, less below 80% Insensitive around the normal PO 2 COHb and Met. Hb

Clinical Example 1 72 y/o male, COPD with acute exacerbation n Under O 2 2 L/min p. H 7. 44, PCO 2 54, PO 2 60, HCO 3 36 n Metabolic alkalosis with respiratory compensation n Mixed respiratory acidosis n

Clinical Example 2 30 y/o male, sudden onset dyspnea n Room air n 7. 33/24/111/12 n Metabolic acidosis n Respiratory compensation n Normal A-a O 2 gradient n O 2↑: hyperventilation n

Clinical Example 3 70 y/o male, acute hemoptysis and dyspnea n Room air n 7. 50/31/88/24 n Respiratory alkalosis n Not been renal compensated yet n Normal PO 2, but A-a O 2 gradient↑ n

Clinical Example 4 n n n n 18 y/o female, chest tightness and dyspnea for 4 hrs RR 28/min, distressed, widespread wheezing O 2 mask 6 L/min 7. 31/49/115/26 Respiratory acidosis Normal bicarbonate acute May have problems with oxygenation

Clinical Example 5 n n n n 37 y/o female, mild asthma history Wheezes for 3 weeks, increasing chest tightness and dyspnea for 24 hrs, call for ambulance with Oxygen use RR 18/min, anxious and distressed Room air 7. 37/43/97/27 Normal? r/o CO 2 retention Low A-a O 2: Oxygen use in the ambulance

Clinical Example 6 n n n n 19 y/o male, Duchenne muscular dystrophy on wheelchair for 7 yrs No previous respiratory problems but frequent UTI Room air 7. 21/81/44/36 Respiratory acidosis Metabolic compensation Normal A-a O 2 pure ventilatory failure

Clinical Example 7 n n n n 57 y/o male, smoker, one week URI then 36 hrs productive cough, fever and dyspnea RR 36/min, distressed, CXR: RLL pneumonia 7. 33/27/51/22, 2 L/min 7. 34/32/58/24, 10 L/min mask Early metabolic acidosis Severe hypoxemic respiratory failure Intra-pulmonary shunting

Thank you for your attention
- Slides: 55