Blood Gas Analysis Teguh Triyono Bagian Patologi Klinik

Blood Gas Analysis Teguh Triyono Bagian Patologi Klinik FKUGM/ RSUP Dr Sardjito Yogyakarta

Normal acid-base balance • Maintained through a number of buffer system but the most important is the bicarbonate system. The organs responsible are the kidneys and the lungs • The body maintain p. H or the hydrogen ion concentration (H+) of the blood 7. 35 -7. 45

• Assessment of acid-base balance is obtained by measuring the components of bicarbonate buffer system which can be expressed as the following modified Hendersen-Hasselbalch equation: p. H= (HCO 3)/ p. CO 2

Components of a Blood Gas HCO 3 p. O 2 Oxygenation: lungs/ECMO Base: metabolic p. CO 2 Acid: lungs/ECMO p. H The sum total of the acid/base balance, on a log scale (p. H=-log[H+])

Normal Values p. H p. CO 2 p. O 2 HCO 3 BE Arterial 7. 35 -7. 45 35 -45 80 -100 22 -26 -2 to +2 Venous 7. 30 -7. 40 43 -50 ~45 22 -26 -2 to +2

Aims of Blood Gas Analysis 1. Determine if p. H is acidotic or alkalotic 2. Determine cause: 1. Respiratory 2. Metabolic 3. Mixed 3. Check oxygenation

Why Arterial Blood? Firmly establish the severity of an oxygenation abnormality To evaluate hyper- or hypoventilation – Currently no convenient noninvasive way of evaluating p. CO 2 To determine acid-base status, particularly in patients with metabolic acidosis (e. g. , diabetic ketoacidosis) To monitor the application of mechanical ventilation

• The p. H defines the (H+) of blood: increased p. H = alkalaemia decreased p. H = acidemia • The (HCO 3) defines metabolic component: increased (HCO 3) = metabolic alkalosis decreased (HCO 3) = metabolic acidosis • The p. CO 2 defines respiratory component: increased p. CO 2 = respiratory acidosis decreased p. CO 2 = respiratory alkalosis

• Acidaemia : p. H < 7. 35 metabolic acidosis : (HCO 3) < 23 mmol/L respiratory acidosis : p. CO 2 > 45 mm. Hg • Alkalaemia : p. H > 7. 45 metabolic alkalosis : (HCO 3) > 33 mmol/L respiratory alkalosis : p. CO 2 < 35 mm. Hg

Simple metabolic acidosis • Low blood bicarbonate • The primary event is either an increased in hydrogen ion production or a decreased in its rate of excretion. Loss of bicarbonate via the gut (diarrhea) or the kidney is equivalent to increased H+ production • Causes: renal failure, diarrhea, lactic acidosis, diabetic ketoacidosis

Simple metabolic alkalosis • high blood bicarbonate • The primary event is either an decreased in hydrogen ion production or a increased in its rate of excretion. Loss of hydrogen ions via the gut (vomiting) or the kidney (diuretic therapy) is equivalent to increased bicarbonate production • Causes: diuretic therapy, vomiting, mineralocorticoid excess

Respiratory acidosis • high blood p. CO 2 • The primary event is retention of CO 2 which can be due to lung disease or hypoventilation by a subject with a normal respiratory tract • Causes: CNS depression(trauma, drugs), neuromuscular disorders(poliomyelitis, GBS, MG), thoracic disorder(hydrothorax, pneumothorax), lung disorders(bronchial obstruction, emphysema, oedema), mechanical ventilation

Respiratory alkalosis • low blood p. CO 2 • The primary event is hyperventilation resulting in a low p. CO 2 • Causes: CNS disturbances (hypoxamia, trauma, infection), pulmonary disorders(embolus, oedema, asthma, pneumonia), mechanical ventilation

Acid-Base Regulation • Three mechanisms to maintain p. H – Respiratory (CO 2) – Buffer (in the blood: carbonic acid/bicarbonate, phosphate buffers, Hgb) – Renal (HCO 3 -)

Compensation of acid-base disorders • Occurs in response to simple acid-base disturbances whereby homeostatic mechanism attempt to shift an abnormal p. H back towards normal. • A metabolic acidosis (low HCO 3) is compensated by a respiratory alkalosis (low p. CO 2)

Practical-1 • Every change in CO 2 of 10 m. Eq/L causes p. H to change by 0. 08 (or Δ 1 = 0. 007) • Increased CO 2 causes a decreases in p. H • Decreased CO 2 causes an increase in p. H

Respiratory Acidosis • Hypercarbia from hypoventilation • Findings: – p. CO 2 increased therefore… p. H decreases • Example: ABG : 7. 32/50/ /25

Respiratory Alkalosis • Hypocarbia from hyperventilation • Findings: – p. CO 2 decreased… therefore p. H increases • Example: ABG – 7. 45/32/ /25

Metabolic Changes • Remember normal HCO 3 - is 22 -26

Practical-2 • Every change in HCO 3 - of 10 m. Eq/L causes p. H to change by 0. 15 • Increased HCO 3 - causes an increase in p. H • Decreased HCO 3 - causes a decrease in p. H

Metabolic Acidosis • Inability to excrete acid – e. g. renal tubular acidosis • Loss of base – e. g. diarrhea • Example: – ABG – 7. 25/40/ /15

Metabolic Alkalosis • Loss of acid – e. g. vomiting (low Cl and kidney retains HCO 3 -) • Gain of base – e. g. contraction alkalosis (lasix) • Example: – ABG – 7. 55/40/ /35

Mixed • p. H depends on the type, severity, and acuity of each disorder • Over-correction of the p. H does not occur

Practical Application 1. Check p. H 2. Check p. CO 2 3. Every change in CO 2 of 10 m. Eq/L causes p. H to change by 0. 08 4. Every change in HCO 3 - of 10 m. Eq/L causes p. H to change by 0. 15

Example -1 • • ABG- 7. 30/48/ /22 Acidotic or Alkalotic? p. CO 2 High or Low? p. H change = p. CO 2 change?

Example -1 • • ABG- 7. 30/48/ /22 Acidotic or Alkalotic? p. CO 2 High or Low? p. H change = p. CO 2 change? Combined respiratory and metabolic acidosis

Example -2 • • ABG- 7. 42/50/ /32 Acidotic or Alkalotic? p. CO 2 High or Low? p. H change = p. CO 2 change?

Example -2 • • ABG- 7. 42/50/ /32 Acidotic or Alkalotic? p. CO 2 High or Low? p. H change = p. CO 2 change? Metabolic alkalosis with respiratory compensation

For respiratory abnormalities, is the condition acute or chronic? Acute respiratory disturbances change p. H 0. 08 units for every 10 mm. Hg deviation from normal Therefore, in acute respiratory acidosis, the p. H will fall by 0. 08 x [(PCO 2 - 40)/10] In acute respiratory alkalosis, the p. H will rise by 0. 08 x [(40 PCO 2)/10]

For respiratory abnormalities, is the condition acute or chronic? Chronic respiratory disturbances only change p. H 0. 03 units for every 10 mm. Hg deviation from normal Therefore, in chronic respiratory acidosis, p. H will fall by 0. 03 x [(PCO 2 - 40)/10] In chronic respiratory alkalosis, the p. H will rise by 0. 03 x [(40 PCO 2)/10]

Limitations of ABGs • ABGs measure gas partial pressures (tensions) – Remember: PO 2 is not the same as content! A severely anemic patient may have an oxygen content reduced by half while maintaining perfectly acceptable gas exchange and therefore maintaining p. O 2 • Technical issues – They hurt – Sampling from a vein by mistake – Finding an arterial pulse can be difficult in very hypotensive patients – Complications such as arterial thrombosis are possible, but awfully rare
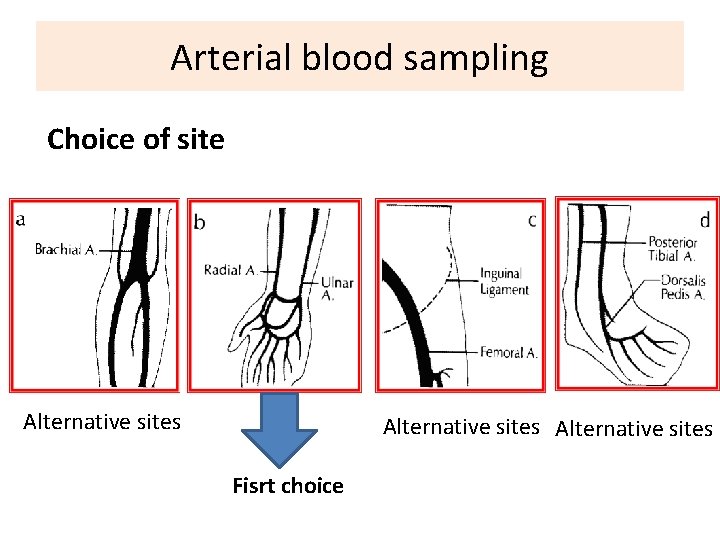
Arterial blood sampling Choice of site Alternative sites Fisrt choice

Terimakasih
- Slides: 33