Blood chemistry Hb Structure Functions Dr Vishnu Kumar

Blood chemistry Hb Structure & Functions Dr. Vishnu Kumar Awasthi Assistant Professor – In – Charge HLS, Department of Biochemistry 3/6/2021

Objectives § § § Structure of hemoglobin Transport of oxygen by Hb Oxygen dissociation curve (ODC) Factors affecting ODC Heme-heme interaction and co-operativity Effect of p. H and p. CO 2 The Bohr Effect The chloride shift Effect of temperature Effect of 2, 3 -BPG Transport of CO 2 3/6/2021

Haemoglobin structure Ø Haemoglobin (Hb) is the most abundant porphyrin – containing compound. Ø It is a tetramer made up of four subunits. Ø Each subunit contains a heme group and a polypeptide chain. 3/6/2021

Haemoglobin structure § Normal level of Hb in blood of males is 14 – 16 g/dl and in females, 13 – 15 g/dl. § Normal adult blood contains 97% Hb. A, about 2% Hb. A 2 and about 1% Hb. F. 3/6/2021

3/6/2021
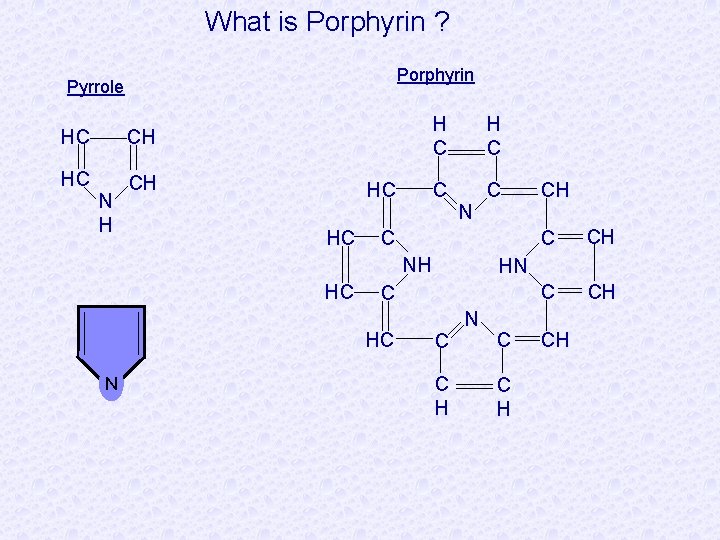
What is Porphyrin ? Porphyrin Pyrrole HC CH N H HC HC H C C C NH HC N C CH HN C HC CH C C H N C C H CH
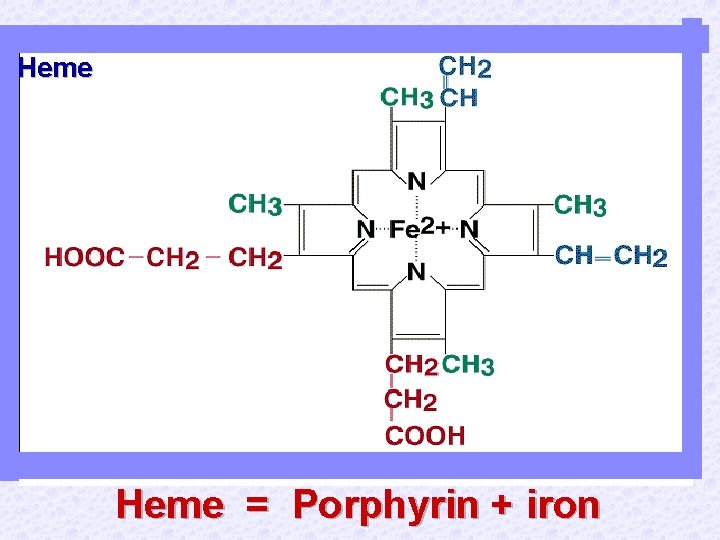
Heme = Porphyrin + iron

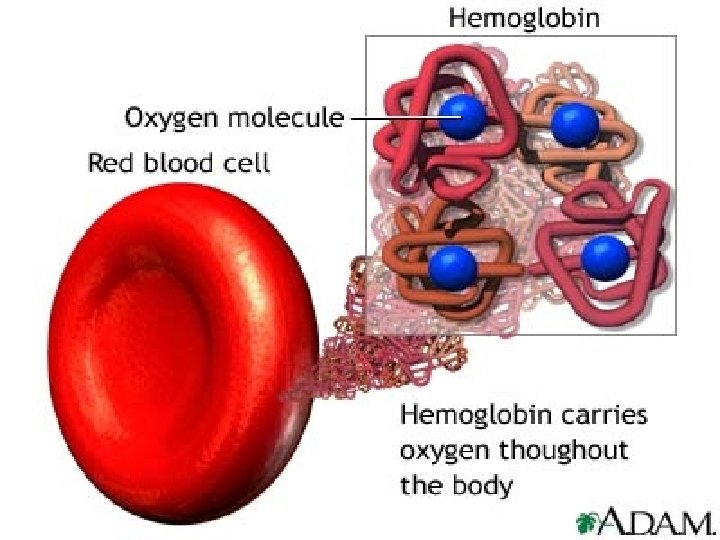
Ø Hemoglobin is a O 2 and CO 2 transport protein found in the RBCs Ø Hemoglobin is an oligomeric protein made up of 2 α β dimers, a total of 4 polypeptide chains: α 1 β 1 α 2 β 2. Ø Total Mr of hemoglobin is 64, 500. Ø The α (141 aa) and β (146 aa) subunits have < 50 % identity. 3/6/2021

§ • The 3 D- structures of α (141 aa) and β (146 aa) subunits of hemoglobin and the single polypeptide of myoglobin are very similar; all three are members of the globin family. § • Each subunit has a haem-binding pocket 3/6/2021
3/6/2021

§ The polypeptide chains are of five types viz. α, β, γ, δ and ε § The α chain is made up of 141 amino acids. § The β , γ, δ and ε chains are made up of 146 amino acid residues each. 3/6/2021

§ Normal adult haemoglobin (Hb. A) is made up of four haem groups, two α chains and two β chains, and is represented as α 2 β 2. § A small amount of Hb. A 2 is also found in adults which is α 2 δ 2. § Foetal haemoglobin (Hb. F) is α 2 γ 2 § Embryonic haemoglobin is α 2 ε 2 3/6/2021

§ The histidine residues linked to iron are present at positions 58 and 87 in α chains and at positions 63 and 92 in other chains. § The bond between iron and the distal histidine residue (His 87 or His 92) is unstable. 3/6/2021

§ The distal iron-histidine bond is broken when haemoglobin is exposed to high oxygen tension § This results in the formation of an ironoxygen bond § The binding of oxygen to haemoglobin changes the conformation of haemoglobin 3/6/2021

§ Two conformations have been described, T (taut) and R (relaxed). § Deoxygenated Hb exists in T form which is stabilised by 2, 3 -bisphoglycerate (2, 3 -BPG) which is formed from 1, 3 -BPG (an intermediate in glycolytic pathway) when there is a deficiency of oxygen in the tissues. 3/6/2021

§ T and R states of Hemoglobin § • Hemoglobin exists in two major conformational § states: Relaxed (R ) and Taut or Tense (T) § • R state has a higher affinity for O 2. § • In the absence of O 2, T state is more stable; when O 2 § binds, R state is more stable, so hemoglobin § undergoes a conformational change to the R state. § • The structural change involves readjustment of interactions between subunits 3/6/2021

§ There is a central cavity in the haemoglobin molecule surrounded by the four polypeptide chains. § 2, 3 -BPG enters this cavity and cross links the two β chains. § When oxygen tension increases, 2, 3 -BPG is displaced and the T form changes into R form. § During this transition, one pair of α and β subunits rotates by 15° relative to the other pair. 3/6/2021

§ Each subunit of haemoglobin can bind one oxygen molecule. § Since there are four subunits in a molecule of haemoglobin, one molecule can bind four oxygen molecules. 3/6/2021

Transport of oxygen by Hb Hb has all the requirements of an ideal respiratory pigment: § It can transport large quantities of oxygen § It has great solubility § It can take up an release oxygen at appropriate partial pressure § It is powerful buffer. 3/6/2021

Oxygen dissociation curve § i. The ability of Hb to load and unload oxygen at physiological p. O 2 is shown by oxygen dissociation curve (ODC) 3/6/2021
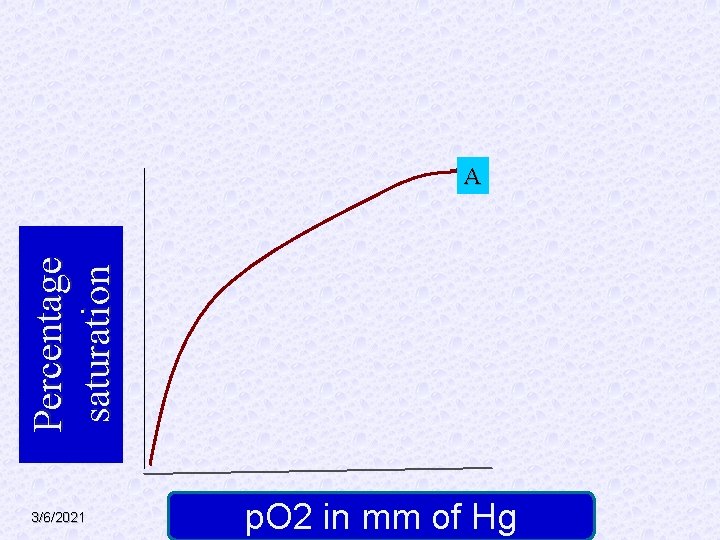
Percentage saturation A 3/6/2021 p. O 2 in mm of Hg
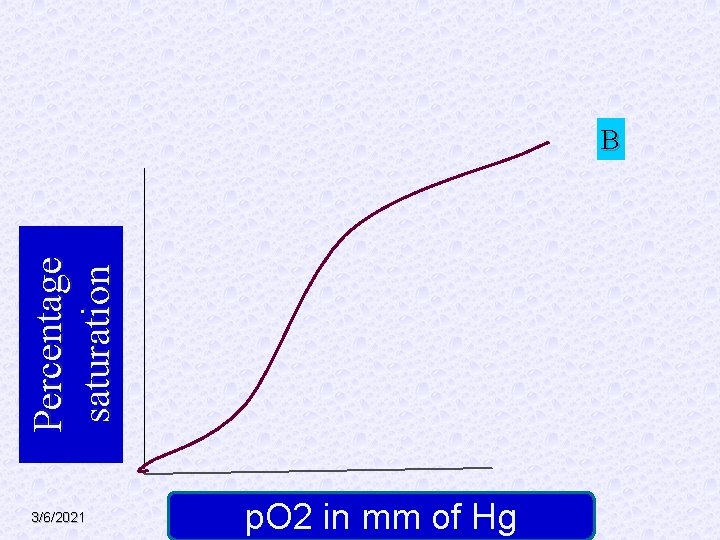
Percentage saturation B 3/6/2021 p. O 2 in mm of Hg
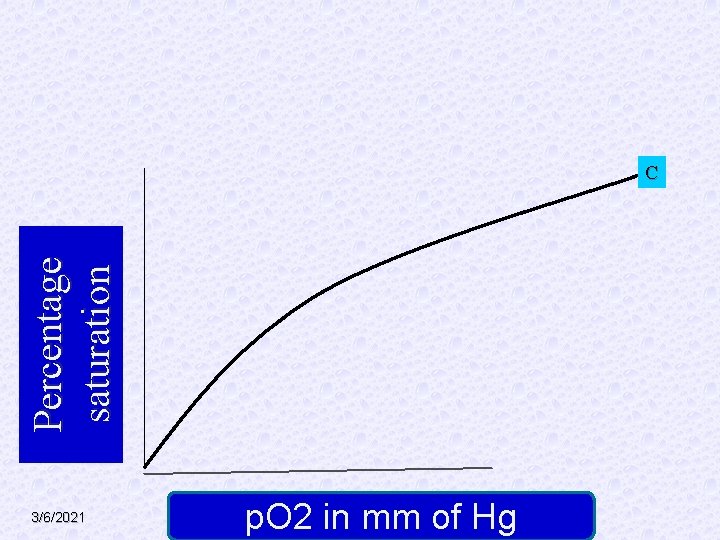
Percentage saturation C 3/6/2021 p. O 2 in mm of Hg
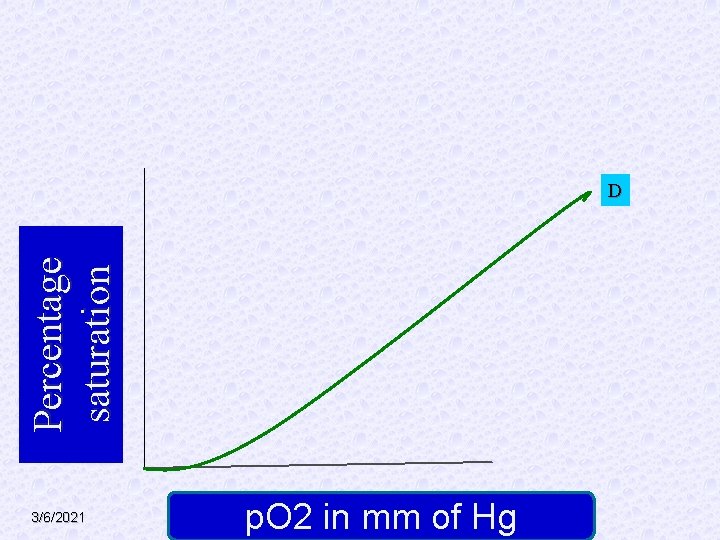
Percentage saturation D 3/6/2021 p. O 2 in mm of Hg

§ A. Theoretical curve as per mass action. § B. Sigmoid curve, due to heme-heme interaction (hill effect). § C. Further shift to right due to carbon dioxide (Bohr effect) and BPG. This curve represents the pattern under normal conditions. § D. further shift to right when temp is increased to 420 C. 3/6/2021

§ ii. At the oxygen tension in the pulmonary alveoli, the Hb is 97% saturated with oxygen. Normal blood with 15 gm/dl of Hb can carry 20 ml of oxygen /dl of blood. § iii. In the tissue capillaries, where the p. O 2 is only 40 mm. Hg, theoretically Hb saturation is 75%. Thus under STP conditions, blood can release only 22%. 3/6/2021

Factors affecting ODC § 1. Heme-heme interaction and cooperativity: § A. the sigmoid shape of ODC – due to allosteric effect, or co-operativity. § equilibrium of Hb=O 2 Hill equation (A V Hill, nobel prize, 1922) 3/6/2021
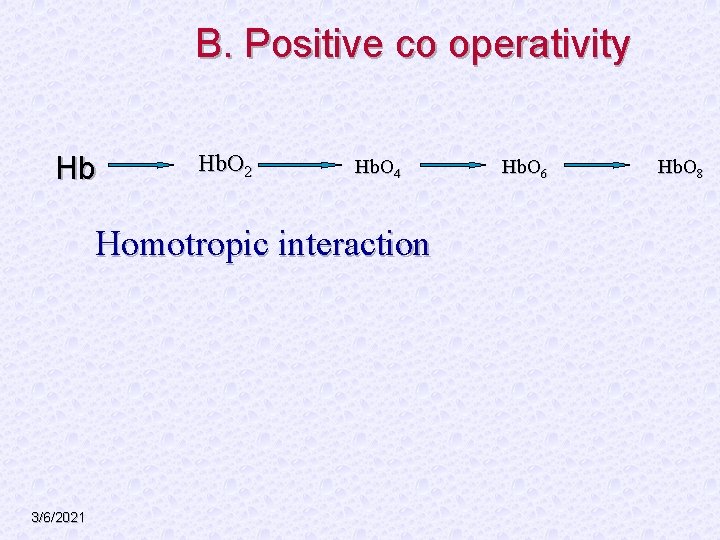
B. Positive co operativity Hb Hb. O 2 Hb. O 4 Homotropic interaction 3/6/2021 Hb. O 6 Hb. O 8

c. Each successive addition of O 2, increase the affinity of Hb to O 2 synergistically. § D. Similarly, binding of 2, 3 – BPG at a site other than the oxygen binding site, lowers the affinity for oxygen (heterotropic interaction). 3/6/2021
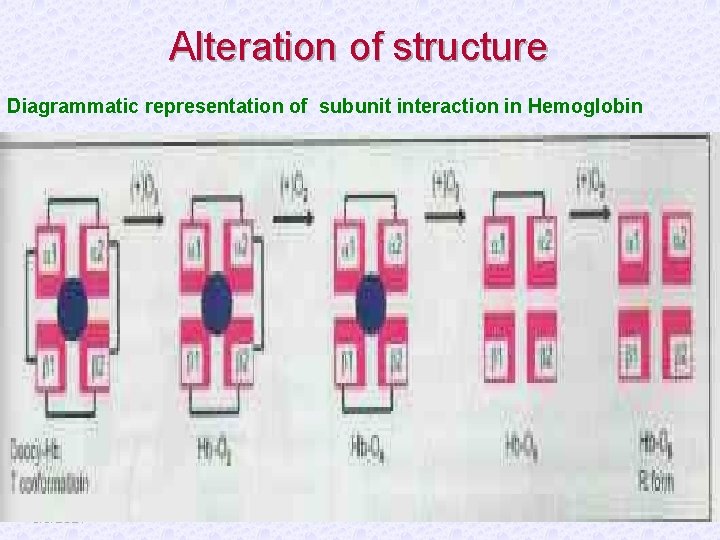
Alteration of structure Diagrammatic representation of subunit interaction in Hemoglobin 3/6/2021

§ During oxygen uptake, the T form to the R form with disruption of the salt bridges. § The Hb subunits are moved relative to one another. § During oxygenation, the α 1 - β 2 interface shows movement. § The two subunits slip over each other. § The quaternary structure of oxy Hb is described as R form; and that of de-oxy Hb is T form. 3/6/2021

§ When oxygenation occurs the salt bonds are broken successively. Thus on oxygenation, the Hb molecule can form two similar dimers. (2 x alpha)+(2 x beta)→ 2 x(alpha-beta) (Deoxy-Hb) 3/6/2021 (oxy-Hb)

3/6/2021

3. The Bohr Effect i. The influence of p. H and p. CO 2 to facilitate oxygenation of Hb in the lungs and deoxygenation at the tissues is known as the Bohr effect (1904). ii. Binding of CO 2 forces the release of O 2 iii. When the p. CO 2 high, CO 2 diffuses into the RBCs CO 2 + H 2 O → H 2 CO 3 → H+ + HCO 3 Carbonic Anhydrase Iv. When carbonic acid is ionizes, the intracellular p. H falls. The affinity of Hb for oxygen is decreased and oxygen is unloaded 3/6/2021
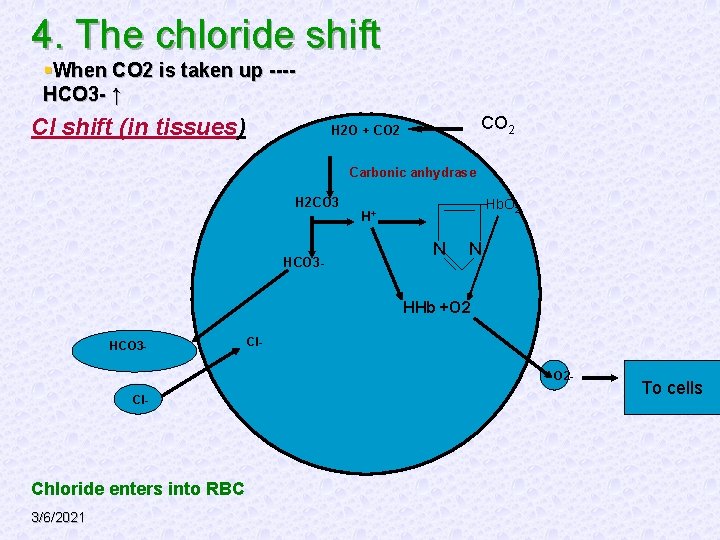
4. The chloride shift §When CO 2 is taken up ---HCO 3 - ↑ Cl shift (in tissues) CO 2 H 2 O + CO 2 Carbonic anhydrase H 2 CO 3 HCO 3 - Hb. O 2 H+ N N HHb +O 2 HCO 3 - Cl. O 2 - Cl- Chloride enters into RBC 3/6/2021 To cells

§ When the blood reaches the lungs, reverse reaction takes place 3/6/2021
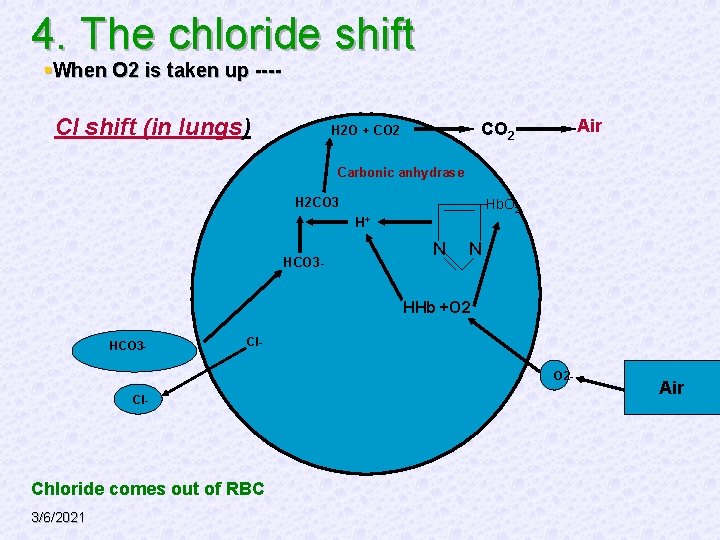
4. The chloride shift §When O 2 is taken up ---- Cl shift (in lungs) Air CO 2 H 2 O + CO 2 Carbonic anhydrase H 2 CO 3 Hb. O 2 H+ HCO 3 - N N HHb +O 2 HCO 3 - Cl. O 2 - Cl- Chloride comes out of RBC 3/6/2021 Air

5. Effect of temperature § § § p 50 = the p. O 2 at which Hb is half saturated p 50 of normal Hb = 26 mm. Hg (at 37 o. C) Elevation of temp. causes 88 % increase in p 50 § ODC shifts to left at low temp. § Under febrile conditions , increased needs of oxygen met by a shift in ODC to right. 3/6/2021

6. Effect of 2, 3 -BPG § § § Normal 2, 3 -BPG level=15 ± 1. 5 mg/g Hb. 2, 3 -BPG == high in children 2, 3 -BPG is produced from 1, 3 -BPG, an intermediate of glycolytic pathway. 2, 3 -BPG, preferentially binds to deoxy. Hb and stabilizes T form When T form reverts to R, 2, 3 -BPG ejected During oxygenation, BPG released 3/6/2021
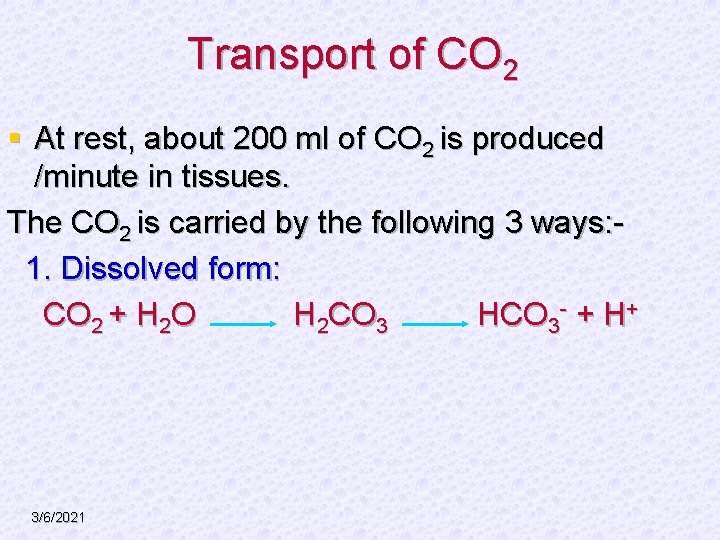
Transport of CO 2 § At rest, about 200 ml of CO 2 is produced /minute in tissues. The CO 2 is carried by the following 3 ways: 1. Dissolved form: CO 2 + H 2 O H 2 CO 3 HCO 3 - + H+ 3/6/2021

2. Isohydric transport of CO 2 1. Haldane effect: The H+ ions are buffered by the deoxyhemoglobin. 2. In tissue 3. Oxy-Hb is more – (negatively) charged than deoxy-Hb 4. In the Lungs H - Hb + 4 O 2 3/6/2021 + Hb (O 2)4 + H

5. The proton released in the RBC combine with HCO 3 - forming H 2 CO 3 which would dissociate to CO 2, that is expelled through pulmonary capillaries. 6. As the HCO 3 - level inside the erythrocytes falls, more and HCO 3 - gets into the RBC, and chloride diffuse out. 3/6/2021

3. Carriage as carbamino- Hb R-NH 2 + CO 2 R-NH-COOH 4. Clinical Applications. 1 - Hypoxic states, O 2 affinity decreased. - ODC shift right. - increased in 2, 3 -BPG increased in RBC. 2. In anemia, increased oxygen unloading will ensure proper oxygenation of tissues. 3. 2, 3 -BPG level varies as Hb conc. 3/6/2021

4. The Red cell 2, 3 -BPG level is decreased in acidosis and increased in alkalosis, ODC shift to right. 5. Transfusion of large vol. of stored blood which has a low level of 2, 3 -BPG can lead sudden hypoxia and a left shifted ODC. 3/6/2021
- Slides: 44