BLACKS PRINCIPLE At the end of the chapter

BLACK’S PRINCIPLE At the end of the chapter, you should be able : To apply formula Q=m. c. ∆t; Q=m. U; Q=m. L for solving simple problem.

Black’s Principle The amount of heat energy given by a warm substance is the same as the amount of heat energy received by cooler substance

Suppose there are two substances having different temperatures Substance 1 Mass = m 1 Specific heat = c 1 Temperature = t 1 l Substance 2 Mass = m 2 Specific heat = c 2 Temperature = t 2 l If t 1>t 2 , Then substance 1 will give an amount of heat energy to substance 2, so
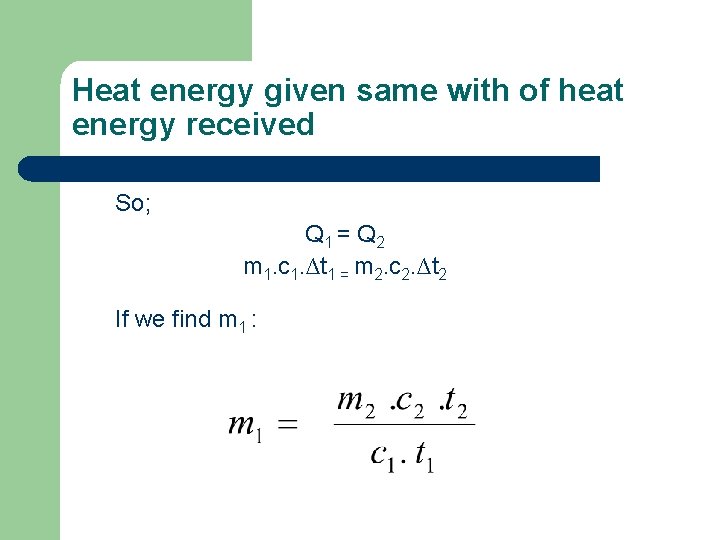
Heat energy given same with of heat energy received So; Q 1 = Q 2 m 1. c 1. ∆t 1 = m 2. c 2. ∆t 2 If we find m 1 :

Example 1. 2 kg of water have 20 o. C is immersed in the container. Which is have 1 kg with 60 o. C. What is the final temperature ? Solution 1= 2 kg m = 1 kg 2 t 1=20 o. C t 2=60 o. C Unknown : t(final) ? Given : m

Solution : Q 1 = Q 2 m 1. c 1. ∆t 1 = m 2. c 2. ∆t 2 o o 2 kg. {t(final)-20 c} = 1 kg. {60 c-(tfinal)} o o 2. t(final)-40 c= 60 c- t(final) o 100 C o t(final)=33. 3 C 3. t(final)=

Exercise 100 g of iron is heated up to 80 o. C. The iron is immersed in water at 30 o. C; then the temperature of the water increases to 32 o. C. What is the mass of water ? c iron = 450 j/kgo. C, c water = 4200 j/kgo. C 2. Kedalam bak mandi dicampurkan 150 kg air panas bersuhu 90 o. C dengan air dingin bersuhu 10 o. C sehingga menjadi air hangat bersuhu 40 o. C. Hitung massa air dingin yang di campurkan ? 1.
- Slides: 7