Blackbody Radiation All bodies at a temperature T

Blackbody Radiation ØAll bodies at a temperature T emit and absorb thermal electromagnetic radiation n n Blackbody radiation In thermal equilibrium, the power emitted equals the power absorbed ØHow is blackbody radiation absorbed and emitted? 1

Blackbody Radiation 2

Blackbody Radiation Ø A blackbody is a perfect absorber of radiation Ø A simple blackbody is given by a hole in a wall of some enclosure Ø Both absorption and can occur Ø The radiation properties of the cavity are independent of the enclosure material 3
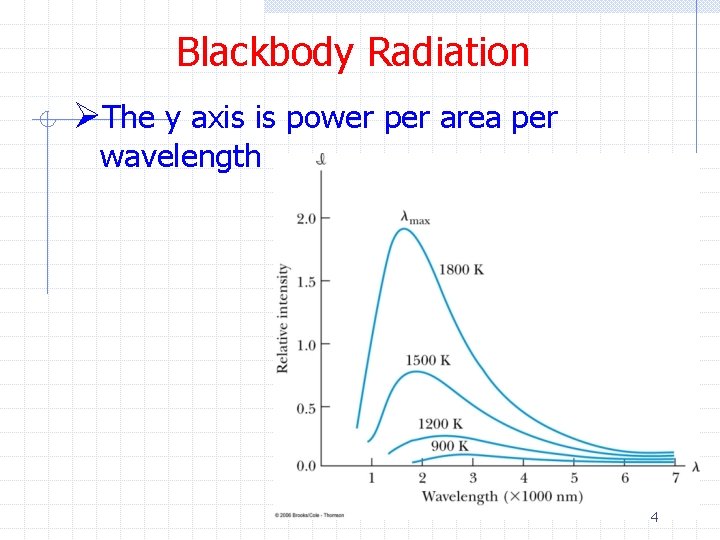
Blackbody Radiation ØThe y axis is power per area per wavelength 4
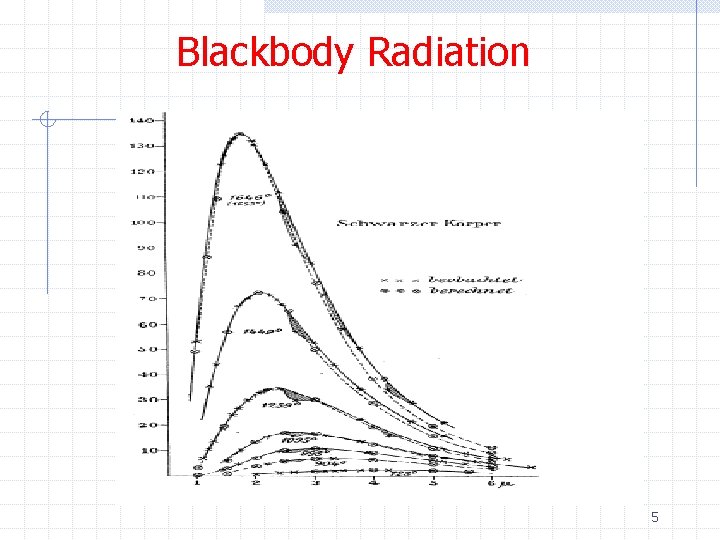
Blackbody Radiation 5

Blackbody Radiation ØWien’s displacement law n Wavelength decreases as T increases ØStefan-Boltzmann law n Total power / area radiated increases as T 4 6

Blackbody Radiation Ø Attempts to calculate the spectral distribution of blackbody radiation from first principles failed Ø The best description was given by the Rayleigh -Jeans formula Ø This described the distribution at long wavelengths but increased without limit as λ→ 0 n Ultraviolet catastrophe 7
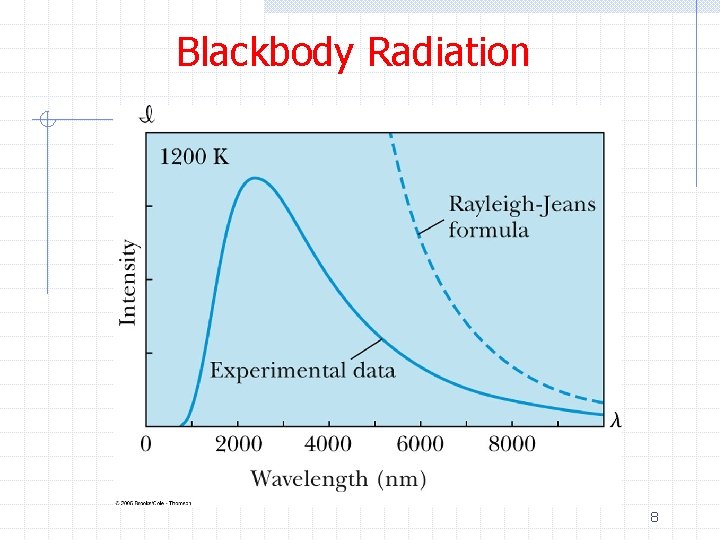
Blackbody Radiation 8

Blackbody Radiation Ø Planck was able to calculate the correct distribution by assuming energy was quantized (he was desperate) n n Microscopic (atomic) oscillators can only have certain discrete energies The oscillators can only absorb or emit energy in multiples of 9

Blackbody Radiation Ø Planck’s radiation law agreed with data Ø It leads directly to Wien’s displacement law and the Stefan-Boltzmann law Ø It agrees with Rayleigh-Jeans formula for large wavelengths n See derivations for both in Thornton and Rex 10

Blackbody Radiation Ø We may derive the radiation law later in the course but for now consider n n The electromagnetic energy inside the (metallic) cavity must exist in the form of standing waves with nodes at the surfaces Classically, the equipartition theorem gives the average energy of a standing wave w Note the same value is predicted for all standing waves independent of their frequency n The amount of radiation coming out of the cavity is then the number of different wave modes X the energy per mode 11

Blackbody Radiation Ø Let E=nhv, then the average energy is 12

Kinetic Theory of Gases ØBased on “atomic” theory of matter ØResults include n Speed of a molecule in a gas w vrms = (<v 2>)1/2 = (3 k. T/m)1/2 n Equipartion theorem w n Internal energy U = f/2 Nk. T = f/2 n. RT Heat capacity w CV = (d. U/d. T)V = f/2 R n Maxwell speed distribution 13

Blackbody Radiation ØTo evaluate this we use standard “tricks” from statistical mechanics 14

Blackbody Radiation ØAnd note 15

Blackbody Radiation ØPutting these together we have 16

Blackbody Radiation ØTo finish the calculation we’d have to multiply the average energy of a wave of frequency f x the number of waves with frequency f ØThis would give us the Planck distribution 17

Blackbody Radiation ØYou can how Planck avoided the ultraviolet catastrophe n Because the energy is proportional to the frequency w The average energy is k. T when the possible energies are small compared to k. T w The average energy is extremely small when the possible energies are large compared to k. T (because P(ε) is extremely small) 18

Blackbody Radiation ØPlanck’s paper is generally considered to be the birthplace of quantum mechanics n n Revisionist history? Planck did not pay too much attention to energy quantization Neither did anyone else There is controversy of whether he even intended the energy of an oscillator to be nhf 19
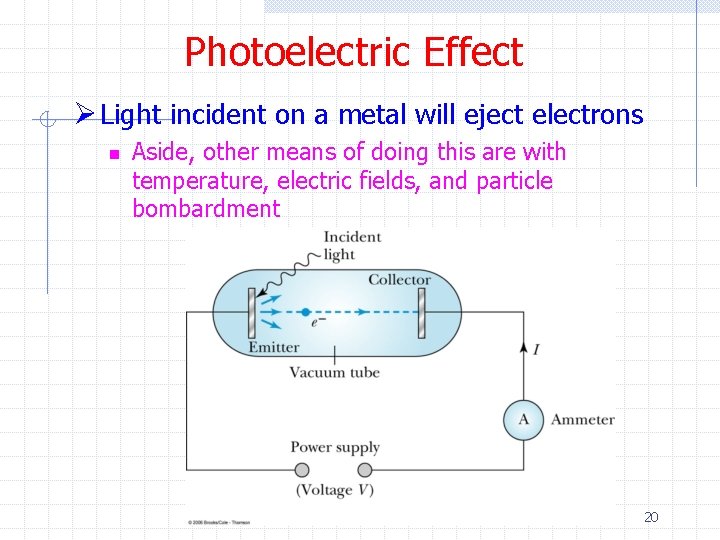
Photoelectric Effect Ø Light incident on a metal will eject electrons n Aside, other means of doing this are with temperature, electric fields, and particle bombardment 20

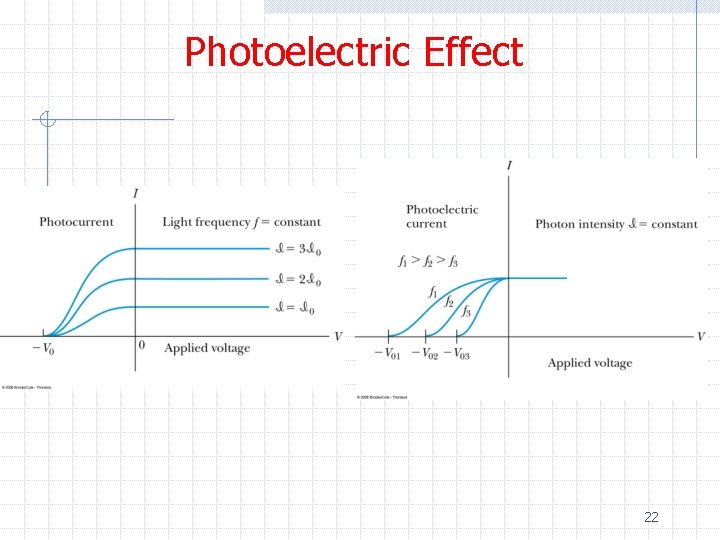
Photoelectric Effect ØExperiments showed n n Kinetic energy of the photoelectrons are independent of the light intensity The maximum kinetic energy of the photoelectrons depends on the light frequency The smaller the work function φ the smaller the threshold frequency to produce photoelectrons The number of photoelectrons is proportional to light intensity 21
Photoelectric Effect 22
Photoelectric Effect 23

Photoelectric Effect ØExplained by Einstein in one of his annus mirabilis papers ØIn his paper he assumed n n Electromagnetic field was quantized Light quanta were localized in space (like particles) == photons Energy E = hf In the photoelectric process, the energy quanta (photons) are completely absorbed 24

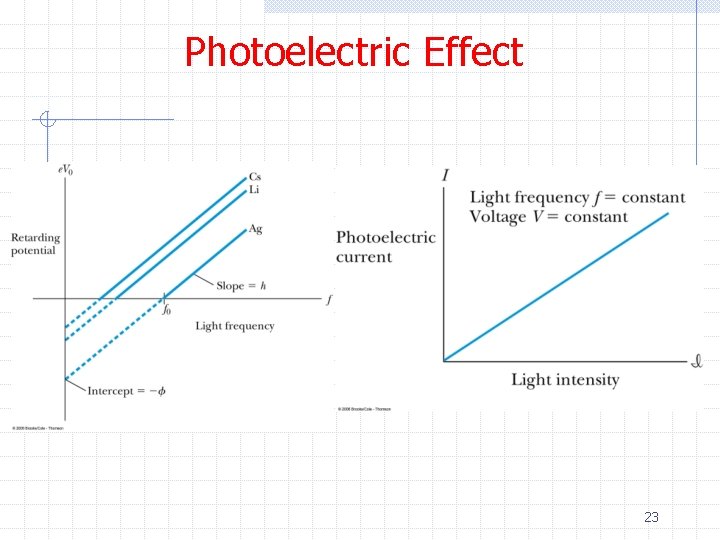
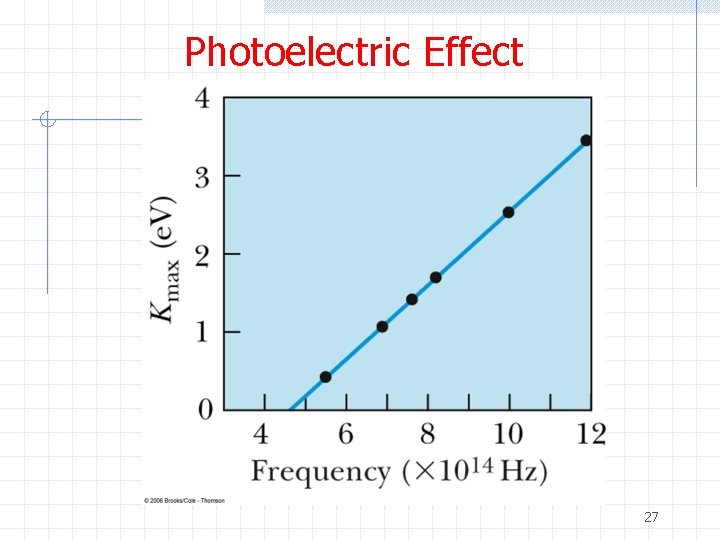
Photoelectric Effect ØThus photons penetrate the surface of the metal and are absorbed by electrons ØThe electrons overcome attractive forces that normally hold them in the material and escape ØConservation of energy gives n hf = 1/2 mv 2 max + φ ØAnd consequently he predicted n n 1/2 mv 2 max= e. V 0 = hf – φ Note h/e can be measured from the slope 25

Photoelectric Effect ØThis was strange since it involved Planck’s constant h ØThis was a difficult experiment to carry out n n It took almost a decade to verify Millikan was the principle experimenter (who tried to prove Einstein’s theory wrong) ØThe end result was proof that light energy is quantized and E=hf 26
Photoelectric Effect 27
- Slides: 27