Black is the new Silver Silver Diamine Fluoride

Black is the new Silver ! Silver Diamine Fluoride: The Old and the New 2018 UK Community Based Faculty Annual Conference March 23, 2018 Jerry Caudill, DMD, Avēsis Incorporated, Kentucky State Dental Director Nikki Stone, DMD, UK North Fork Valley Community Health Center, Dental Director

Kentucky Oral Health “Network” Who are we? A loose-knit group of people interested in oral health in KY, most of whom are either affiliated with UK or the KPCA. When do we meet together? There are two main events each year: ▪ SPRING – this UK voluntary faculty conference ▪ FALL – the KPCA annual meeting What is our purpose? We are a “Community of Practice, ” sharing concern and passion for oral health, and trying to improve by interacting regularly and encouraging one another.

Community of Practice What is a Community of Practice? Identity defined by a shared domain of interest, shared commitment, competence, and expertise. • Community activities include engaging discussions, learning together, helping each other, sharing information, building relationships. • Made up of practitioners who develop a shared repertoire of resources, experiences, stories, tools, and ways of addressing recurring problems. • ESTABLISH – ELEVATE – EXPAND INTERPROFESSIONAL – INTEGRATED – INVOLVED – INFORMED STRONG – SUSTAINABLE – SUCCESSFUL

Objectives for Today �Review the history of silver diamine fluoride �Summarize the literature about SDF �Learn the uses for SDF �Hear the gold standard protocols for SDF �Review the Kentucky guidelines for SDF �Receive a toolkit for implementing SDF in your own dental office

The Old Nikki Stone, DMD, dental director, UKNFVCHC
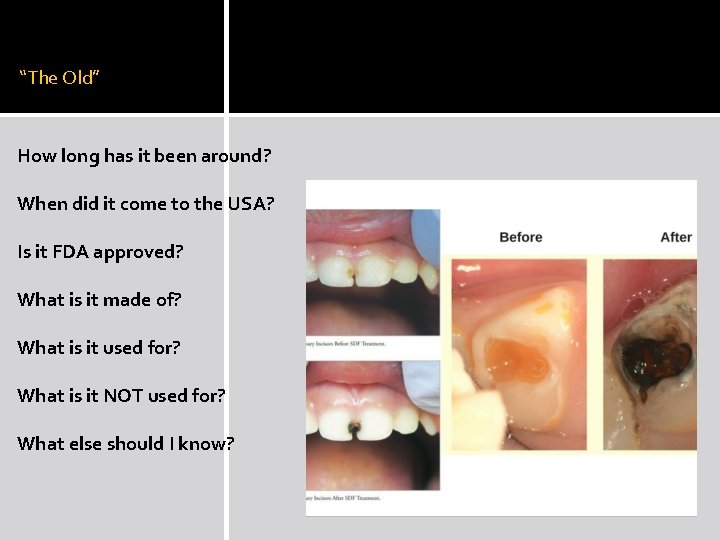
“The Old” How long has it been around? When did it come to the USA? Is it FDA approved? What is it made of? What is it used for? What is it NOT used for? What else should I know?

How long has it been around? �SDF has been used for decades outside the USA for various applications, including dentistry. “SDF…has been used by dental professionals outside the US for both the treatment of dentinal hypersensitivity and as a caries therapy for more than 45 years. ” “No adverse events using silver compounds have been reported in more than 80 years of use in dentistry. ”

When did it come to the USA? �In August 2014, SDF was cleared by the Food and Drug Administration (FDA) for use in the USA. �It is ONLY available from one company: www. elevateoralcare. com Advantage Arrest silver diamine fluoride ▪ Unidose 1 box of 30 ampules for $122. 50 ($4. 08 per use) ▪ Bottle 8 m. L for $162. 50

Is it FDA approved? �The FDA has approved SDF as a “desensitizing agent. ” Fluoride varnish was approved the same way 20 years ago, as a “Class II medical device for the treatment of tooth hypersensitivity. ” �BOTH silver diamine fluoride and fluoride varnish are used off-label to prevent and treat caries.

What is it made of? �SDF is a colorless liquid containing silver particles and 38% fluoride ion (44, 800 ppm). It is 25% silver 8% ammonia, 5% fluoride, and 62% water. This is referred to as 38% SDF.

What is it used for? �ARRESTS active carious lesions For people unable to access dental treatment For people unable to tolerate conventional dental care such as ▪ Very young “pre-cooperative” children ▪ Persons with intellectual/ developmental disabilities ▪ Older adults (root caries) �PREVENTS caries *

What is it NOT used for? �Silver allergy is the only known contraindication �Cannot be used in these situations: Pulpitis Pulpal necrosis Deep carious lesions where the dentin has been excavated (ammonia content and high p. H may create a pulpal reaction) People concerned about esthetics!

What else should I know? �SDF is not a complete solution to managing caries �Single application has been reported to be insufficient for sustained benefit �Unpleasant but transient metallic taste �Potential to irritate gingival and mucosal surfaces �Black staining is permanent to teeth (and to floors, clothing, and furniture!)

What else should I know? �SDF out-performed fluoride varnish when applied directly to cavitated lesions for the non-surgical arrest of caries in children and older adults �SDF demonstrated impressive caries prevention also

What else should I know? �At least 8 published reports of randomized clinical trials consistently demonstrated very high rates of caries arrest �A 2016 meta-analysis concluded SDF applied once a year effectively arrested more than 65% of active caries BUT there is still no consensus for the optimal number and frequency of applications

“The New” Jerry W. Caudill, DMD, FAGD, MAGD, CDC, FPFA, FICD KY State Dental Director, Avēsis Incorporated / A Guardian Company
“The New” Why am I a Medicaid Dentist? What Changed? How do I use SDF in my practice? What is the gold standard? What is the UCSF Protocol? Video Clip Kentucky Guidelines Toolkit Questions

Why take Medicaid? �Why am I a Medicaid Dentist? �https: //www. youtube. com/watch? v=JJUghr 1 a 4 z. Y

What Changed? �The Food and Drug Administration recently cleared silver diamine fluoride for reducing tooth sensitivity. Clinical trials document arrest and prevention of dental caries by silver diamine fluoride. This off-label use is now permissible and appropriate under U. S. law. A CDT code (D 1354) was approved for caries arresting medicaments for 2016 to facilitate documentation and billing.

How do I use SDF in my practice? �Silver diamine fluoride is an inexpensive topical medicament used extensively in other countries to treat dental caries across the age spectrum. �February 5, 2017 D 1354 was added to Kentucky Medicaid for reimbursement and is paid per quadrant. �Some commercial insurance is now covering SDF (e. g. Delta Dental of Wisconsin) �Avēsis encourages and pays for appropriate use of this material.

What is the gold standard? �As Dr. Stone said, there is no nationally agreed upon protocol. �Avēsis has corroborated with Dr. Jeremey Horst at the University of California at San Francisco in developing the KY Medicaid guidelines for our 4 partner Medicaid plans. �Dr. Horst is considered one of the leading researchers in the nation for SDF. He currently recommends applying every 6 months for 4 applications.

What is the gold standard? �That being said, the current Medicaid coverage is 1 application every 6 months for a maximum of 4 lifetime applications per quadrant per patient. �This is CHANGING! 2018 CDT now lists this as “per tooth”. Therefore, it my understanding DMS is changing the payment to per tooth and 2 applications per tooth every 6 months. �So, stay tuned for a future notice about this!

What is the UCSF Protocol? �UCSF Silver Diamine Fluoride Guidelines �https: //www. youtube. com/watch? v=z. UAJkqc. Itc o �https: //www. cda. org/Portals/0/journal_0 12016. pdf �Summary: � 38% silver diamine fluoride is effective and efficient in arresting and preventing carious lesions. �recommend twice-per-year application, only to carious lesions without excavation, for at least the first two years.

UCSF Protocol con’t �For patients without access to both sealants and monitoring, silver diamine fluoride is the agent of choice for prevention of caries in permanent molars — particularly as there is no margin to leak and thereby facilitate deep caries and it does not stain sound enamel.

UCSF Protocol con’t � � � Material: Advantage Silver Arrest (38% SDF, purified water) from Elevate Oral Care. Shelf life: three years unopened. Do not refrigerate. Avoid freezing or extreme heat. Indications: ▪ 1. Extreme caries risk (xerostomia or severe early childhood caries). ▪ 2. Treatment challenged by behavioral or medical management. ▪ 3. Patients with carious lesions that may not all be treated in one visit. ▪ 4. Difficult to treat dental carious lesions. ▪ 5. Patients without access to dental care. Maximum dose: 25 μL (1 drop) / 10 kg per treatment visit. SDF Contraindication: Silver allergy. SDF Relative Contraindications: Ulcerative gingivitis, stomatitis. SSKI Contraindications: Pregnancy, breastfeeding.

UCSF Protocol con’t -Considerations � Considerations: � • Decayed dentin will darken as the caries lesions arrest. Most will be dark brown or black. � • SDF can stain the skin, which will clear in two to three weeks without treatment. � • SDF can permanently stain operatory surfaces and clothes. � • A control restoration (e. g. , GI via ART or other material) may be considered after SDF treatment. � • Saturated solution of potassium iodide (SSKI <saturated solution of potassium iodide>, Lugol’s Solution, various sources) can be used after SDF to decrease color changes. � • Re-application is usually recommended, biannually until the cavity is restored or arrested or the tooth exfoliates.

UCSF Protocol con’t Procedure � � � 1. Plastic-lined cover for counter, plastic-lined bib for patient. 2. Standard personal protective equipment (PPE) for provider and patient. 3. One drop of SDF into the deep end of a plastic dappen dish (also obtain one drop of SSKI in a separate dappen dish if selected). 4. Remove bulk saliva with saliva ejector. 5. Isolate tongue and cheek from affected teeth with 2 -inch by 2 -inch gauze or cotton rolls. 6. If near the gingiva, consider applying petroleum jelly with a cotton applicator for safety. 7. Dry affected tooth surfaces with triple syringe or if not feasible dry with cotton. 8. Bend microsponge, immerse into SDF, remove excess on side of dappen dish. 9. Apply directly onto the affected tooth surface(s) with microsponge. 10. Allow SDF to absorb for up to one minute if reasonable, then remove excess with gauze or cotton roll. (If using SSKI, apply with a different microsponge. Repeat one to three times until no further white precipitates are observed. Wait five to 10 seconds between applications. Remove excess with cotton. ) 11. Rinse with water. 12. Place gloves, cotton and microbrushes into plastic waste bags

UCSF Video Clip �Technique Video �https: //www. youtube. com/watch? v=i. Pgccl 9 q rs. A

Avēsis Kentucky Guidelines Special Consent Form
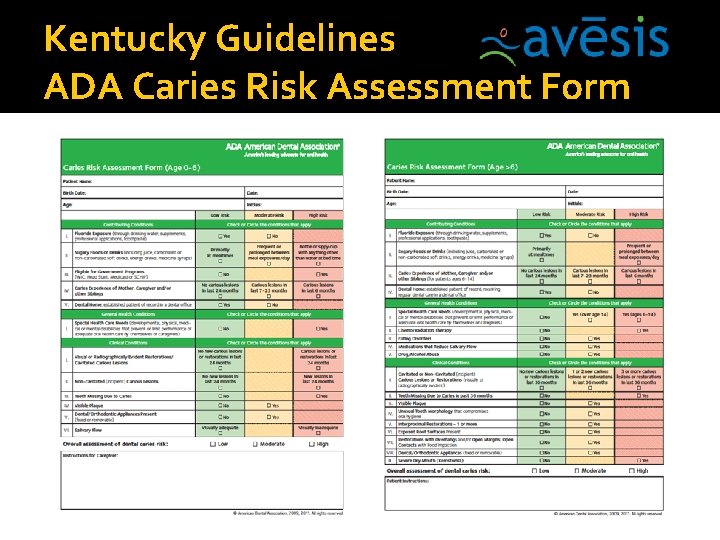
Kentucky Guidelines ADA Caries Risk Assessment Form

Avēsis Kentucky Guidelines �The consent form and ADA caries risk assessment forms are not required to be submitted for payment but must be kept in patient chart for audit purposes. �D 1354 (SDF) is for high caries risk patients. Procedure code D 0603, must be submitted on the same claim as the SDF treatment thus attesting the patient’s ADA CAMBRA (Caries management by risk assessment) score is “high caries risk. ”

Avēsis Kentucky Current Guidelines Benefit and Limitations �Tooth numbers and quadrants must be included on claims �D 1354 is one per six months per quadrant per dentist or dental group (will be changing to “two per six months” and to “per tooth”) �Limited to a total of four applications per lifetime per quadrant �For high risk caries patients, so code D 0603 must be submitted on the same claim form

Avēsis Kentucky Guidelines Benefit and Limitations �No age restriction �No extraction or restoration of a treated tooth within 90 days, except for special circumstances (pain, pre-auth, narrative, review) �At least one tooth in quadrant with decay into dentin �Should not apply to exposed pulps or teeth approaching exfoliation �Cannot bill fluoride on same day

Toolkit �Avesis notice mailed to all providers last year that included citations and resource page �SDF guidelines are available on the Avēsis portal �Informed consent form is available on the Avēsis portal �Caries risk assessment forms by age (0 -6 and >6) are available on the Avēsis portal

Questions?
- Slides: 35