Black Body Radiation http www google comimages qblackbodyum1hlensafeactiverlscom


Black Body Radiation http: //www. google. com/images? q=black+body&um=1&hl=en&safe=active&rls=com. microsoft: *&tbs=isch: 1&ei=4 Aq. JTc 68 K 66 L 0 QHuj. Zn. ADg&sa=N&start=20&ndsp=20
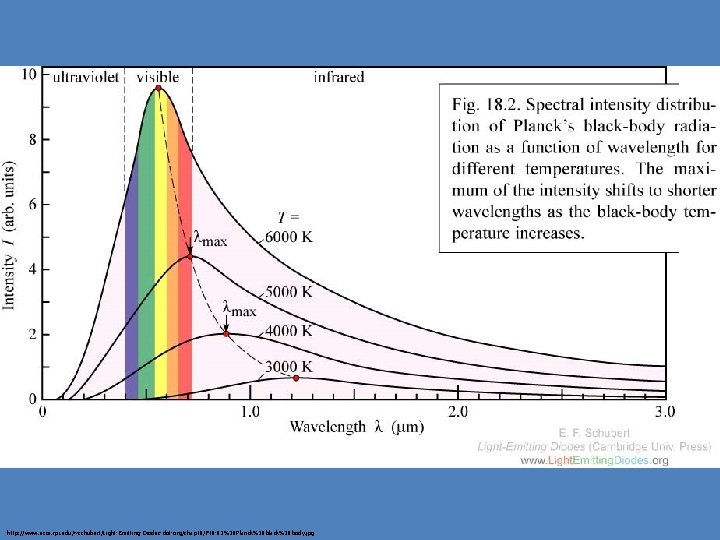
http: //www. ecse. rpi. edu/~schubert/Light-Emitting-Diodes-dot-org/chap 18/F 18 -02%20 Planck%20 black%20 body. jpg

Plank’s Constant Energy of a photon is proportional to the frequency of the photon. Eα f E = h·f -34 h = 6. 626 x 10 J·s
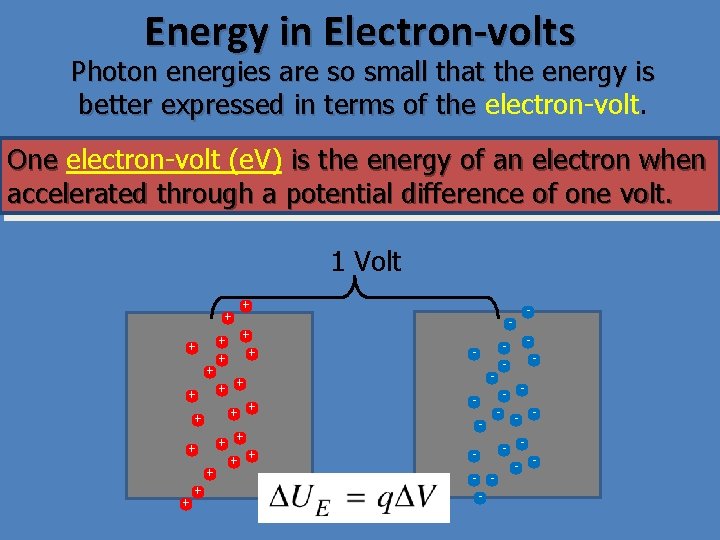
Energy in Electron-volts Photon energies are so small that the energy is better expressed in terms of the electron-volt. One electron-volt (e. V) is the energy of an electron when accelerated through a potential difference of one volt. 1 Volt - + - - - - - + + + + + +
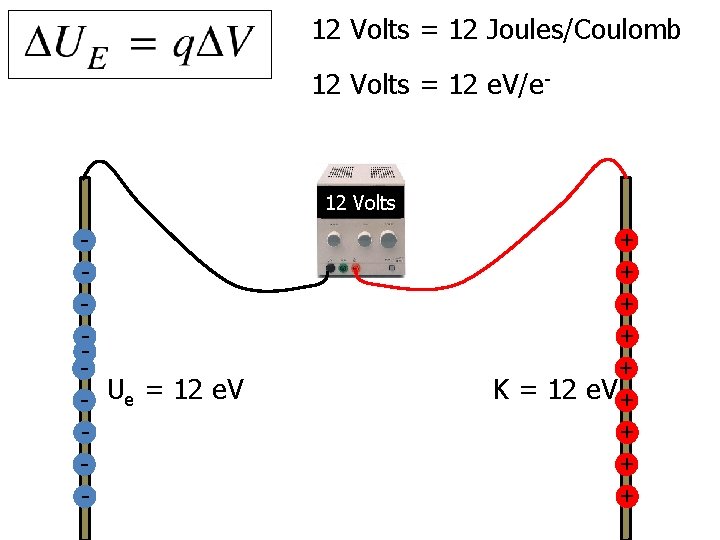
12 Volts = 12 Joules/Coulomb 12 Volts = 12 e. V/e- 12 Volts - Ue = 12 e. V - + + + K = 12 e. V + +

Energy in Electron-volts Photon energies are so small that the energy is better expressed in terms of the electron-volt. One electron-volt (e. V) is the energy of an electron when accelerated through a potential difference of one volt. 1 Joule = 1 Coulomb· 1 Volt 1 e. V = 1. 6 x 10 -19 e · 1 Volt 1 e. V = 1. 60 x 10 -19 J 1 ke. V = 1. 6 x 10 -16 J 1 Me. V = 1. 6 x 10 -13 J
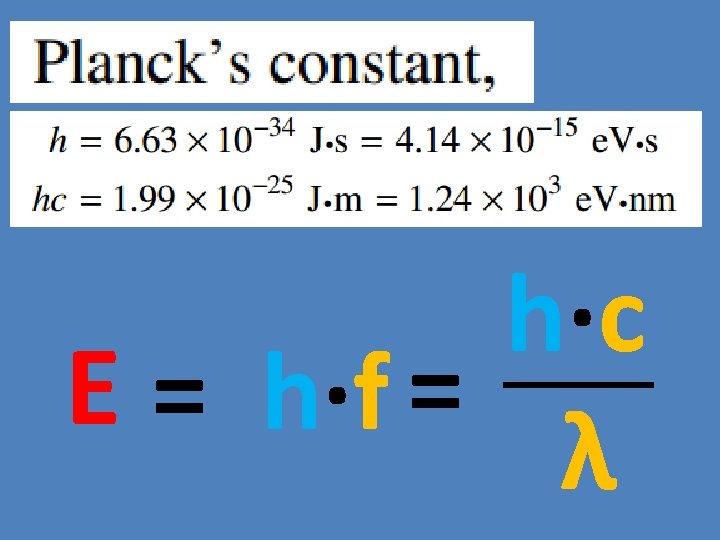
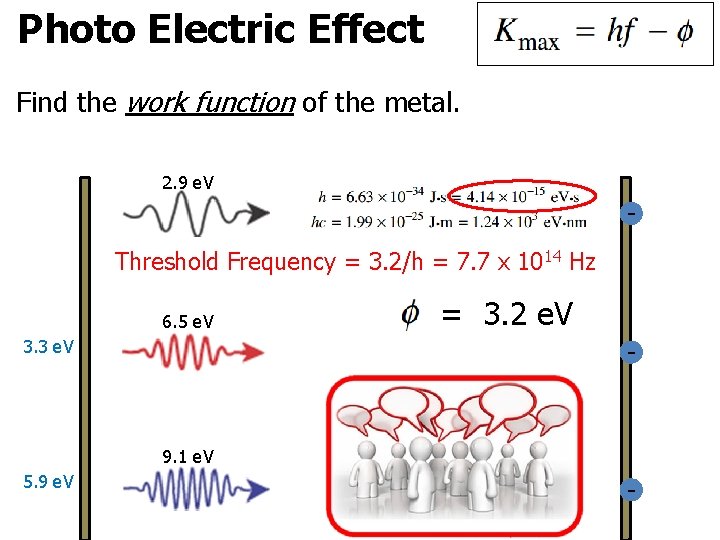
Photo Electric Effect Find the work function of the metal. 2. 9 e. V Threshold Frequency = 3. 2/h = 7. 7 x 1014 Hz 6. 5 e. V = 3. 2 e. V - 3. 3 e. V 9. 1 e. V 5. 9 e. V -

Photo Electric Effect Find the energy of the photon. = 3. 2 e. V Not enough information! -
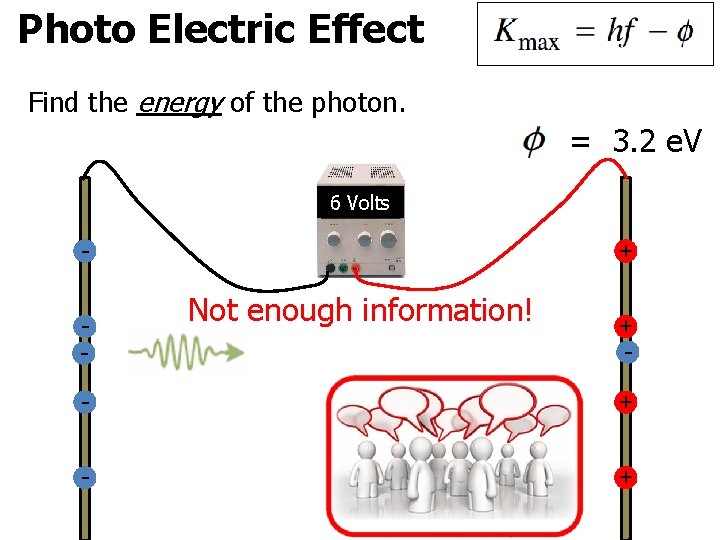
Photo Electric Effect Find the energy of the photon. = 3. 2 e. V 6 Volts - + Not enough information! + - - +
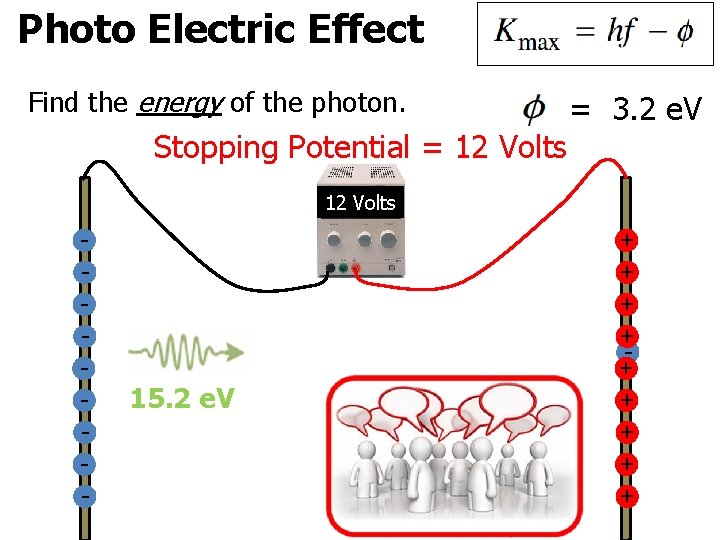
Photo Electric Effect Find the energy of the photon. Stopping Potential = 12 Volts = 3. 2 e. V 12 Volts - 15. 2 e. V + + + + +
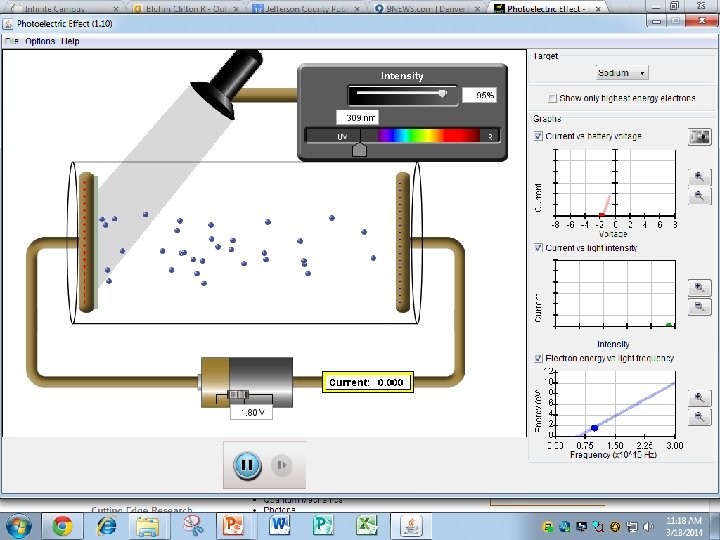
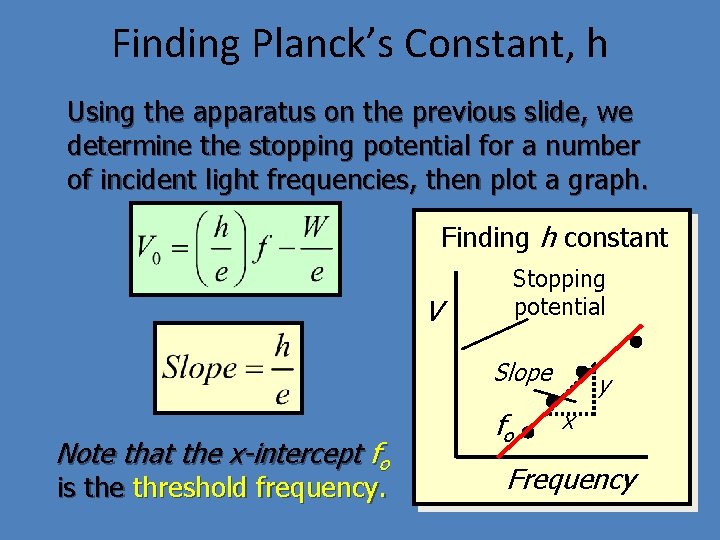
Finding Planck’s Constant, h Using the apparatus on the previous slide, we determine the stopping potential for a number of incident light frequencies, then plot a graph. Finding h constant V Stopping potential Slope Note that the x-intercept fo is the threshold frequency. fo y x Frequency

Momentum of a Photon p = m∙v m = p/v E = m∙c 2 p E= ∙ c 2 v p E= ∙ c 2 c E= p∙c h∙c = p∙c λ h= p λ

Waves and Particles We know that light behaves as both a wave and a particle. The rest mass of a photon is zero, and its wavelength can be found from momentum. Wavelength of a photon: All objects, not just EM waves, have wavelengths which can be found from their momentum de Broglie Wavelength:
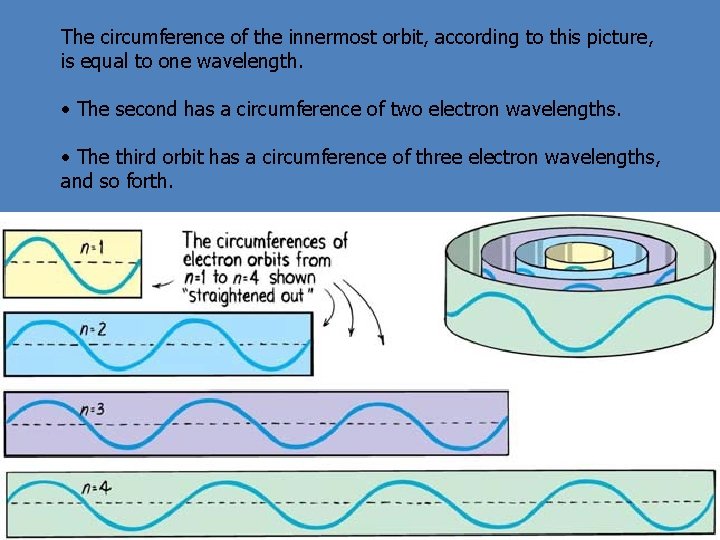
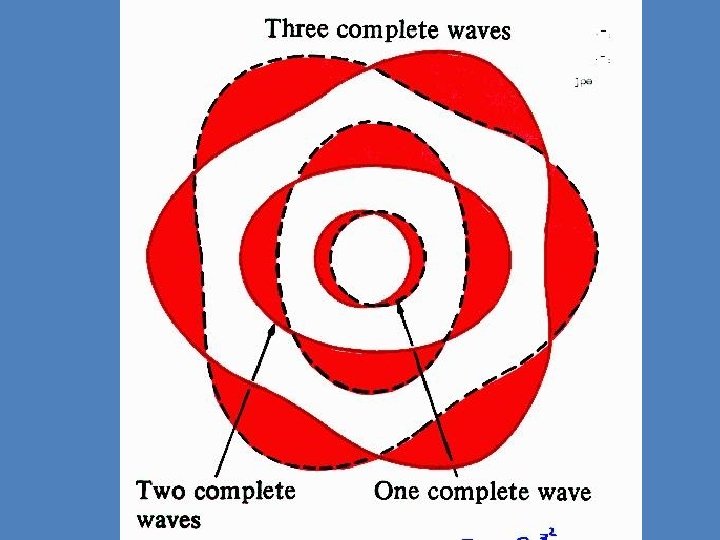
The circumference of the innermost orbit, according to this picture, is equal to one wavelength. • The second has a circumference of two electron wavelengths. • The third orbit has a circumference of three electron wavelengths, and so forth.
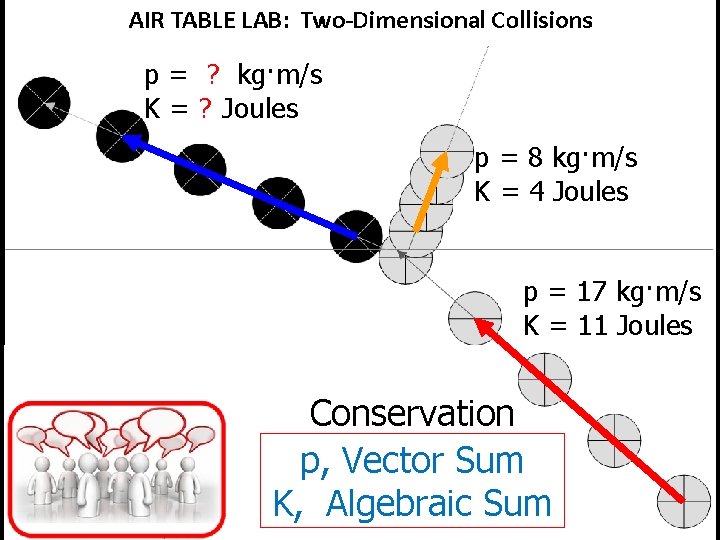
p = 15 ? kg·m/s K=7 ? Joules p = 8 kg·m/s K = 4 Joules p = 17 kg·m/s K = 11 Joules Conservation p, Vector Sum K, Algebraic Sum
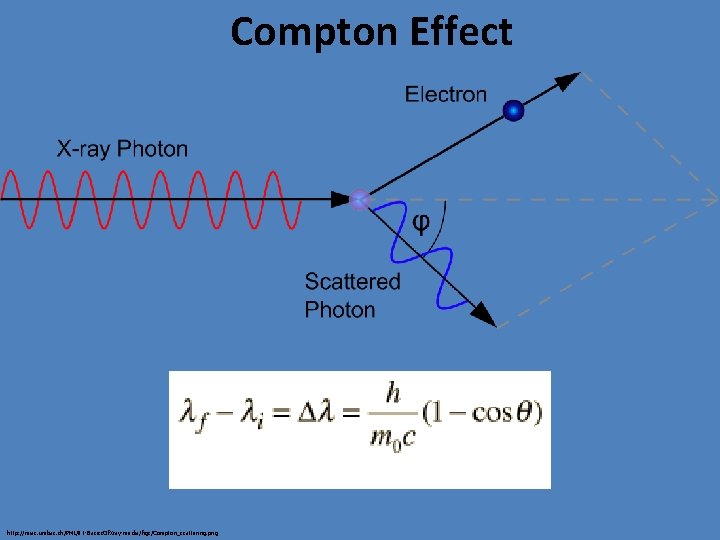
Compton Effect http: //miac. unibas. ch/PMI/01 -Basics. Of. Xray-media/figs/Compton_scattering. png
Compton Scattering
Photoelectric Effect

Chapter 38 C - Atomic Physics • A Power. Point Presentation by • Paul E. Tippens, Professor of Physics • Southern Polytechnic State University © 2007

JJ Thomson’s “Plum Pudding” Model 8 December 1856 – 30 August 1940 http: //2011 period 6 group 4. wikispaces. com/2. +Thomson's+Model
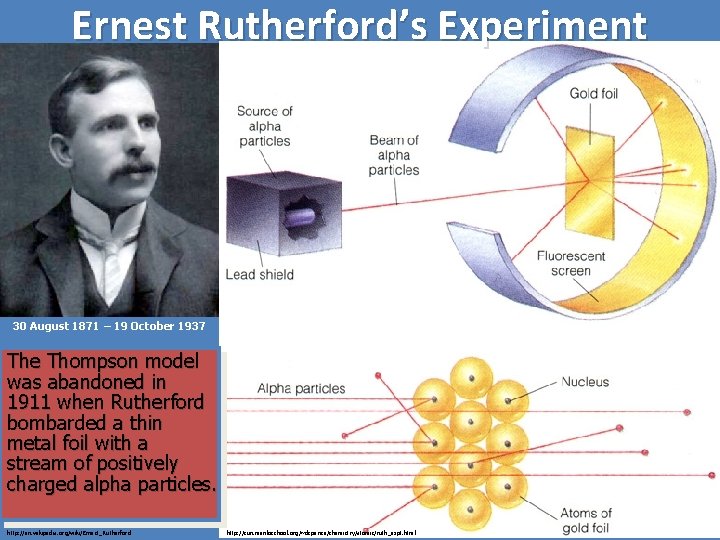
Ernest Rutherford’s Experiment 30 August 1871 – 19 October 1937 The Thompson model was abandoned in 1911 when Rutherford bombarded a thin metal foil with a stream of positively charged alpha particles. http: //en. wikipedia. org/wiki/Ernest_Rutherford http: //sun. menloschool. org/~dspence/chemistry/atomic/ruth_expt. html
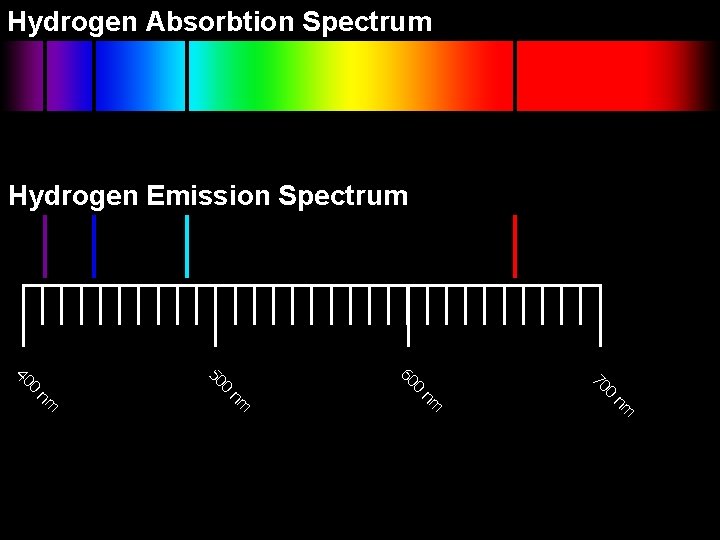
Hydrogen Absorbtion Spectrum Hydrogen Emission Spectrum nm nm 0 70 0 60 0 50 0 40
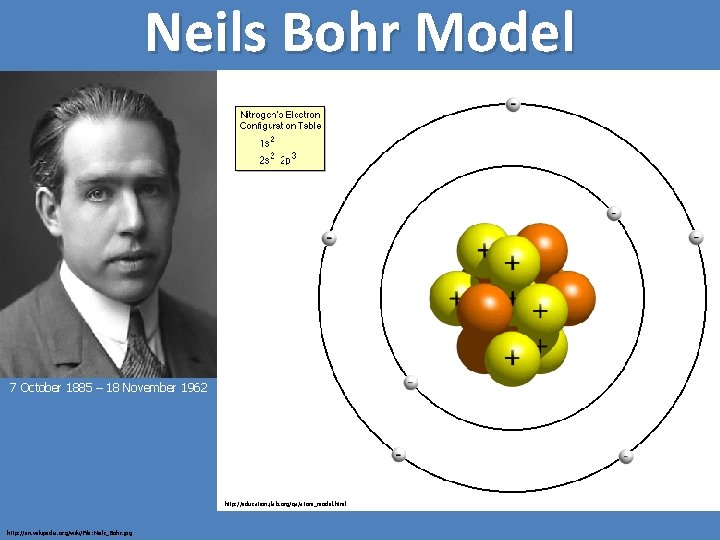
Neils Bohr Model 7 October 1885 – 18 November 1962 http: //education. jlab. org/qa/atom_model. html http: //en. wikipedia. org/wiki/File: Niels_Bohr. jpg

Bohr's starting point was to realize that classical mechanics by itself could never explain the atom's stability. A stable atom has a certain size so that any equation describing it must contain some fundamental constant or combination of constants with a dimension of length. The classical fundamental constants--namely, the charges and the masses of the electron and the nucleus--cannot be combined to make a length. Bohr noticed, however, that the quantum constant formulated by the German physicist Max Planck has dimensions which, when combined with the mass and charge of the electron, produce a measure of length. Numerically, the measure is close to the known size of atoms. This encouraged Bohr to use Planck's constant in searching for a theory of the atom

Energy Levels We can now visualize the hydrogen atom with an electron at many possible energy levels. Emission Absorption The energy of the atom increases on absorption (nf > ni) and decreases on emission (nf < ni). Energy of nth level: The change in energy of the atom can be given in terms of initial ni and final nf levels:
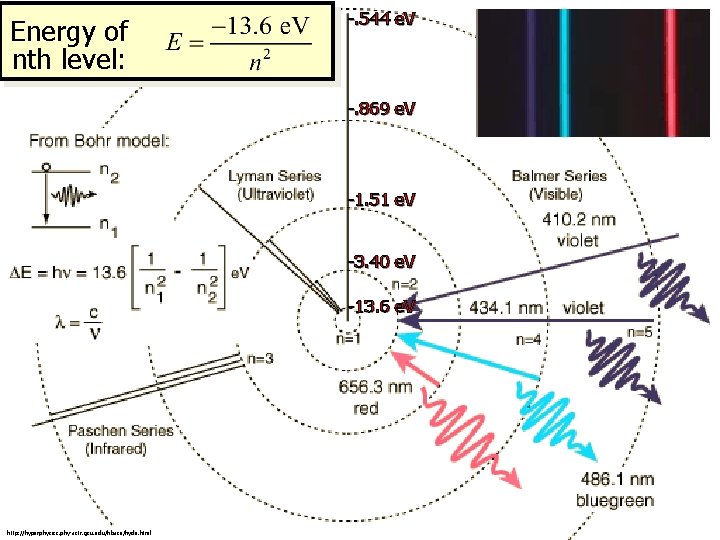
Energy of nth level: -. 544 e. V -. 869 e. V -1. 51 e. V -3. 40 e. V -13. 6 e. V http: //hyperphysics. phy-astr. gsu. edu/hbase/hyde. html

Definitions A nucleon is a general term to denote a nuclear particle - that is, either a proton or a neutron. The atomic number Z of an element is equal to the number of protons in the nucleus of that element. The mass number A of an element is equal to the total number of nucleons (protons + neutrons). The mass number A of any element is equal to the sum of the atomic number Z and the number of neutrons N : A=N+Z
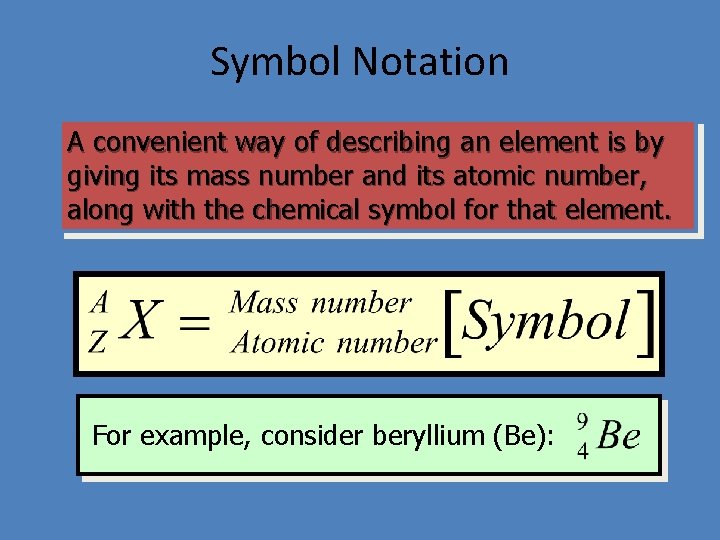
Symbol Notation A convenient way of describing an element is by giving its mass number and its atomic number, along with the chemical symbol for that element. For example, consider beryllium (Be):
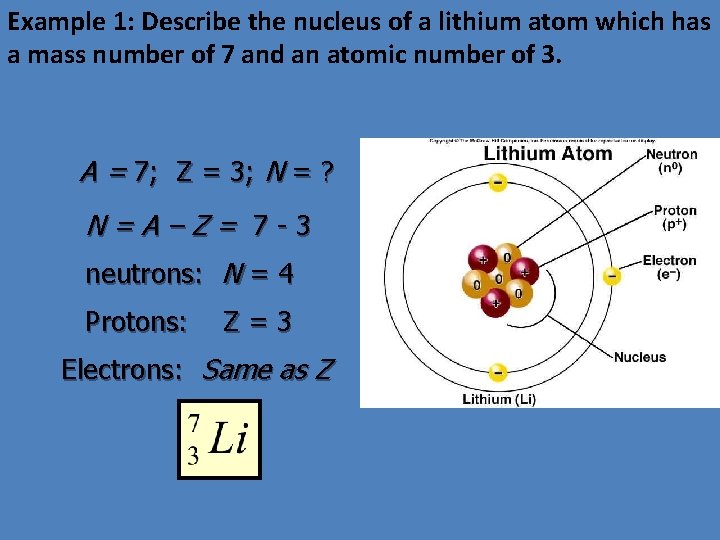
Example 1: Describe the nucleus of a lithium atom which has a mass number of 7 and an atomic number of 3. A = 7; Z = 3; N = ? N=A–Z= 7 -3 neutrons: N = 4 Protons: Z=3 Electrons: Same as Z
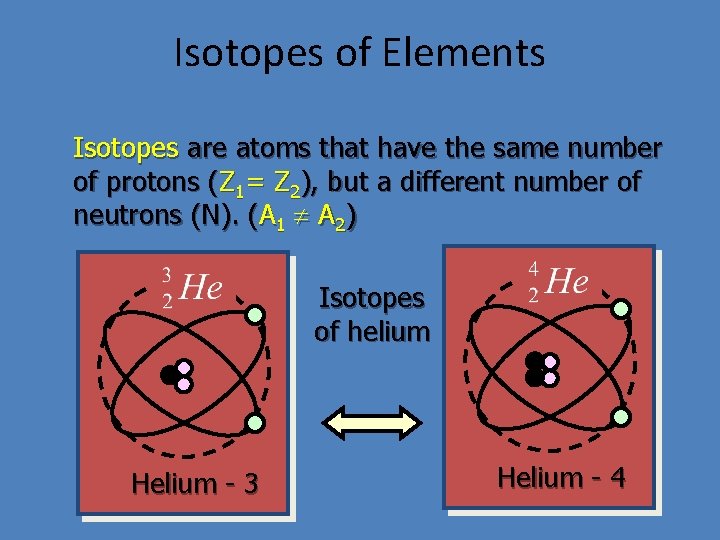
Isotopes of Elements Isotopes are atoms that have the same number of protons (Z 1= Z 2), but a different number of neutrons (N). (A 1 A 2) Isotopes of helium Helium - 3 Helium - 4

Proton = 1. 673 x 10 -27 kg Neutron = 1. 675 x 10 -27 kg Electron = 9. 11 x 10 -31 kg One atomic mass unit (1 u) is equal to one-twelfth of the mass of the most abundant form of the carbon atom-carbon-12. 6(1. 673 x 10 -27 ) + 6(1. 675 x 10 -27) = 2. 0088 x 10 -26 kg 6( = 1. 9927 x 10 -26 kg 1 u= = 1. 6606 x 10 -27 kg 12 Proton = 1. 673 x 10 -27 kg 1 u = 1. 007276 u 1. 6606 x 10 -27 kg Neutron = 1. 675 x 10 -27 kg 1 u = 1. 008665 u 1. 6606 x 10 -27 kg Electron = = 9. 11 x 10 -31 kg 1 u = 0. 00055 u -27 1. 6606 x 10 kg

Mass and Energy Recall Einstein’s equivalency formula for m and E: The energy of a mass of 1 u can be found: E = (1 u)c 2 = (1. 66 x 10 -27 kg)(3 x 108 m/s)2 E = 1. 49 x 10 -10 J When converting amu to energy: Or E = 931. 5 Me. V

The Mass Defect The mass defect is the difference between the rest mass of a nucleus and the sum of the rest masses of its constituent nucleons. The whole is LESS than the sum of the parts! Consider the carbon-12 atom (12. 00000 u): Nuclear mass = Mass of atom – Electron masses = 12. 00000 u – 6(0. 00055 u) = 11. 996706 u The nucleus of the carbon-12 atom has this mass. (Continued. . . )

Mass Defect (Continued) Mass of carbon-12 nucleus: 11. 996706 Proton: 1. 007276 u Neutron: 1. 008665 u The nucleus contains 6 protons and 6 neutrons: 6 p = 6(1. 007276 u) = 6. 043656 u 6 n = 6(1. 008665 u) = 6. 051990 u Total mass of parts: = 12. 095646 u Mass defect m. D = 12. 095646 u – 11. 996706 u m. D = 0. 098940 u
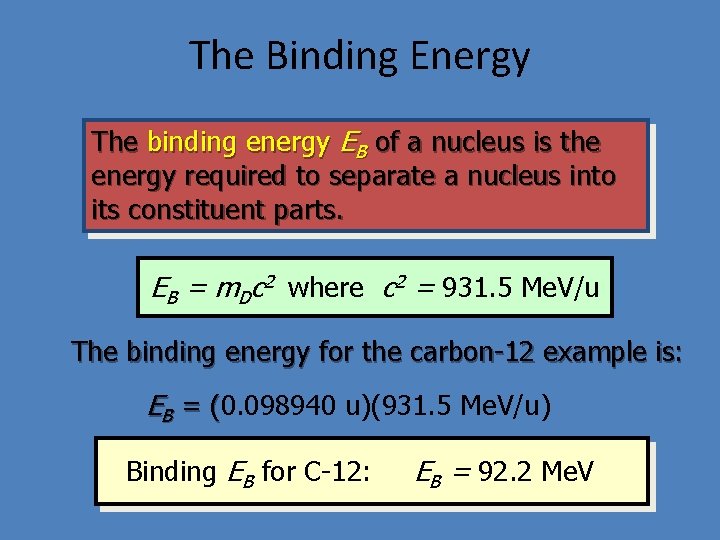
The Binding Energy The binding energy EB of a nucleus is the energy required to separate a nucleus into its constituent parts. EB = m. Dc 2 where c 2 = 931. 5 Me. V/u The binding energy for the carbon-12 example is: EB = (0. 098940 u)(931. 5 Me. V/u) ( Binding EB for C-12: EB = 92. 2 Me. V

Binding Energy per Nucleon An important way of comparing the nuclei of atoms is finding their binding energy per nucleon: Binding energy per nucleon For our C-12 example A = 12 and:

Formula for Mass Defect The following formula is useful for mass defect: Mass defect m. D m. H = 1. 007825 u; mn = 1. 008665 u Z is atomic number; N is neutron number; M is mass of atom (including electrons). By using the mass of the hydrogen atom, you avoid the necessity of subtracting electron masses.

Radioactivity As the heavier atoms become more unstable, particles and photons are emitted from the nucleus and it is said to be radioactive. All elements with A > 82 are radioactive. a bb+ g Examples are: Alpha particles a b- particles (electrons) Gamma rays g b+ particles (positrons)

Conservation Laws For any nuclear reaction, there are three conservation laws which must be obeyed: Conservation of Charge: The total charge of a system can neither be increased nor decreased. Conservation of Nucleons: The total number of nucleons in a reaction must be unchanged. Conservation of Mass Energy: The total massenergy of a system must not change in a nuclear reaction.

Radioactive Decay As discussed, when the ratio of N/Z gets very large, the nucleus becomes unstable and often particles and/or photons are emitted. Alpha decay results in the loss of two protons and two neutrons from the nucleus. X is parent atom and Y is daughter atom The energy is carried away primarily by the K. E. of the alpha particle.

Beta-minus Decay Beta-minus b- decay results when a neutron decays into a proton and an electron. Thus, the Z-number increases by one. X is parent atom and Y is daughter atom The energy is carried away primarily by the K. E. of the electron. -

Beta-plus Decay Beta-plus b+ decay results when a proton decays into a neutron and a positron. Thus, the Z-number decreases by one. X is parent atom and Y is daughter atom The energy is carried away primarily by the K. E. of the positron. +
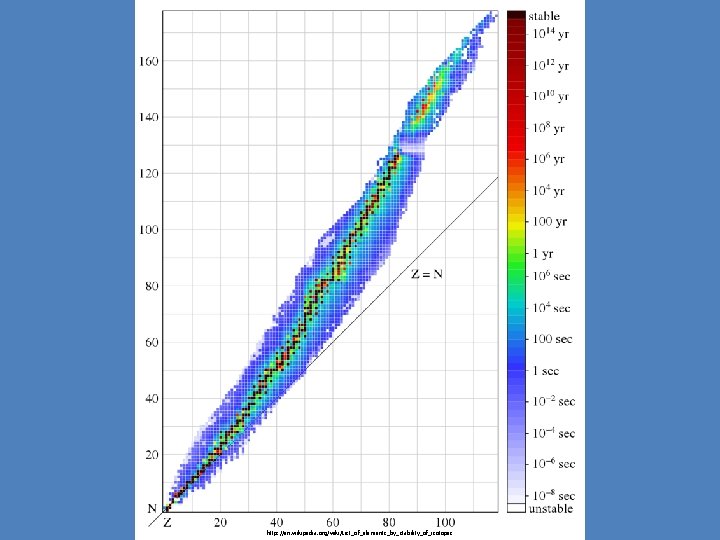
http: //en. wikipedia. org/wiki/List_of_elements_by_stability_of_isotopes
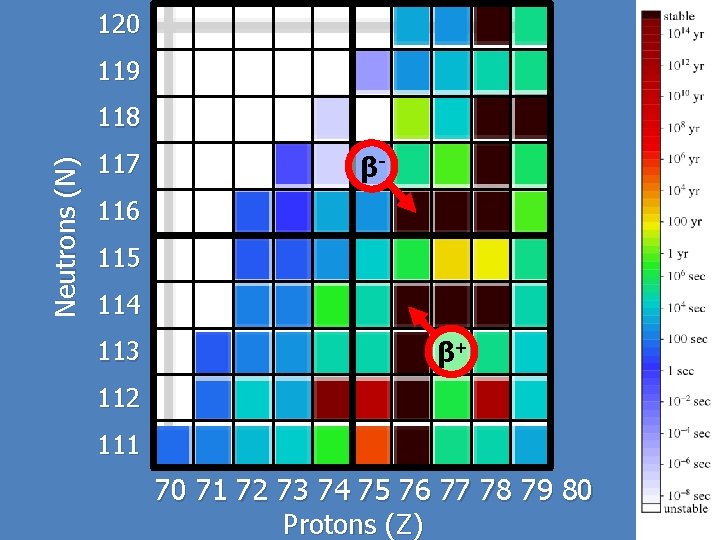
120 119 Neutrons (N) 118 117 β- 116 115 114 113 β+ 112 111 70 71 72 73 74 75 76 77 78 79 80 Protons (Z)
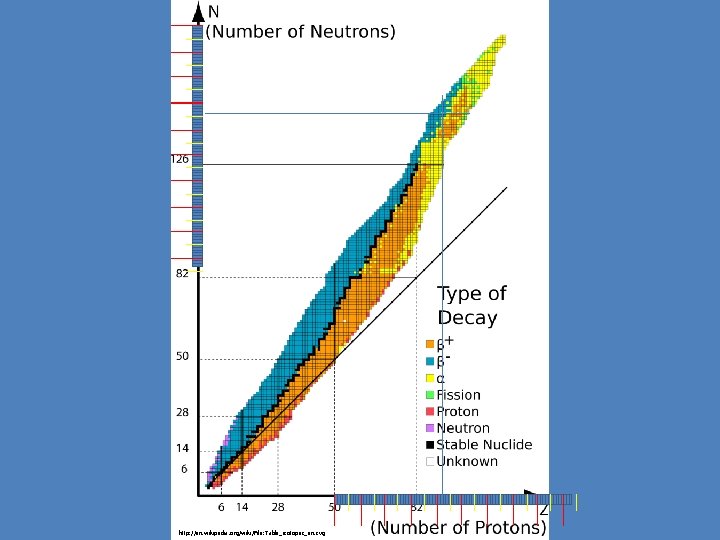
http: //en. wikipedia. org/wiki/File: Table_isotopes_en. svg
- Slides: 49