Bivalirudin Monotherapy Improves 30 day Clinical Outcomes in

Bivalirudin Monotherapy Improves 30 -day Clinical Outcomes in Diabetics with Acute Coronary Syndrome: Report from the ACUITY Trial Frederick Feit, Steven Manoukian, Ramin Ebrahimi, Charles Pollack, Magnus Ohman, Michael Attubato, Roxana Mehran and Gregg Stone

Bivalirudin Monotherapy Improves 30 -day Clinical Outcomes in Diabetics with Acute Coronary Syndrome: Report from the ACUITY Trial Conflicts: Shareholder: Johnson and Johnson, Medicines Co. , Millenium Pharmaceuticals; Consultant: Medicines Co.

ACS in Diabetics: Metabolic Abnormalities § Increased blood glucose causes coronary artery inflammation and is prothrombotic § Increased generation of thrombin, CRP, fibrinogen, von Willebrand factor, factors VII and VIII, and platelet factor 4 § Increased expression of platelet activation markers including p-selectin, which mediates platelet-leukocyte interactions § Higher proportion of platelets expressing GPIIb/IIIa receptors

ACS in Diabetics: Background § Based on prior data including a metaanalysis of ACS trials current clinical guidelines recommend the use of GPIIb/IIIa inhibitors (GPI) in diabetic patients with ACS, especially those in whom PCI is planned 1 § In the ACUITY Trial 13, 819 pts, including 3852 diabetics, with moderate or high risk ACS, undergoing an early invasive strategy were randomly assigned to either the standard of care: Heparin (UFH or enoxaparin) + GPI; or, Bivalirudin + GPI; or Bivalirudin with provisional GPI 1. Roffi et al. Circulation. 2001; 104: 2767 -71

ACS in Diabetics: Methods § We compared adverse events: composite ischemia (death, nonfatal MI, unplanned ischemia driven revascularization), major bleeding and net clinical outcome (composite ischemia or bleeding) within the first 30 days in diabetic vs. nondiabetic pts § We compared the same 30 -day end points in diabetic pts by treatment group

ACUITY Design ACS: Unstable angina or NSTEMI, N=13, 819 Chest pain >10’ within 24 hours, plus Biomarker +, or Dynamic ECG changes, or Documented CAD or all other TIMI risk criteria ASA Clopidogrel per local practice Prior UFH, LMWH (1 dose), eptifibatide and tirofiban allowed Enoxaparin or UFH + IIb/IIIa inhibitor Bivalirudin + provisional IIb/IIIa Cath within 72 hours PCI, CABG or medical management 30 day endpoints Death, MI, IUR, ACUITY major bleeding (net clinical outcome) Stone et al. Presented 2006; ACC
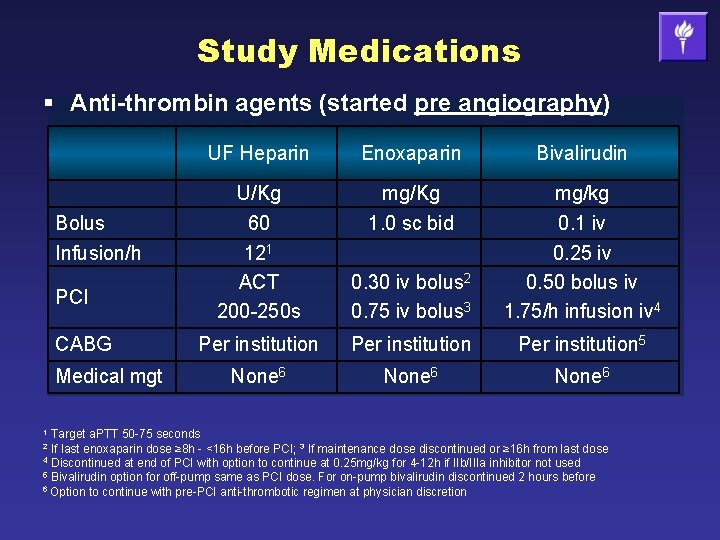
Study Medications § Anti-thrombin agents (started pre angiography) Bolus Infusion/h PCI CABG Medical mgt UF Heparin Enoxaparin Bivalirudin U/Kg 60 121 ACT 200 -250 s mg/Kg 1. 0 sc bid 0. 30 iv bolus 2 0. 75 iv bolus 3 mg/kg 0. 1 iv 0. 25 iv 0. 50 bolus iv 1. 75/h infusion iv 4 Per institution 5 None 6 Target a. PTT 50 -75 seconds If last enoxaparin dose ≥ 8 h - <16 h before PCI; 3 If maintenance dose discontinued or ≥ 16 h from last dose 4 Discontinued at end of PCI with option to continue at 0. 25 mg/kg for 4 -12 h if IIb/IIIa inhibitor not used 5 Bivalirudin option for off-pump same as PCI dose. For on-pump bivalirudin discontinued 2 hours before 6 Option to continue with pre-PCI anti-thrombotic regimen at physician discretion 1 2
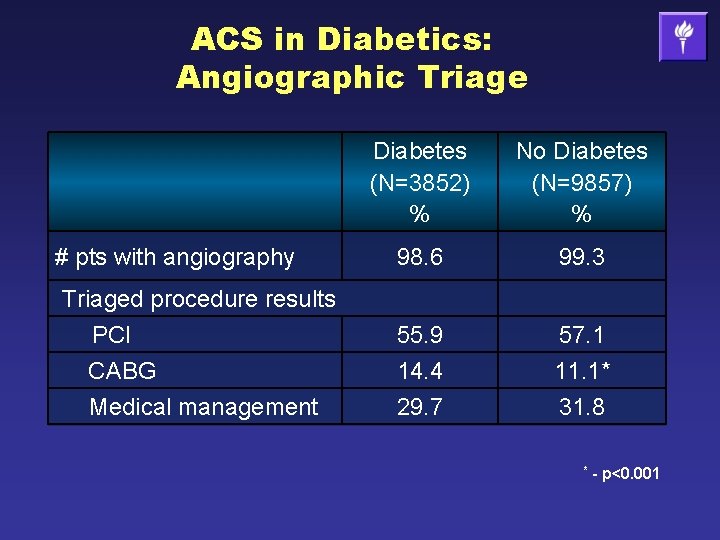
ACS in Diabetics: Angiographic Triage # pts with angiography Triaged procedure results PCI CABG Medical management Diabetes (N=3852) % No Diabetes (N=9857) % 98. 6 99. 3 55. 9 14. 4 29. 7 57. 1 11. 1* 31. 8 * - p<0. 001
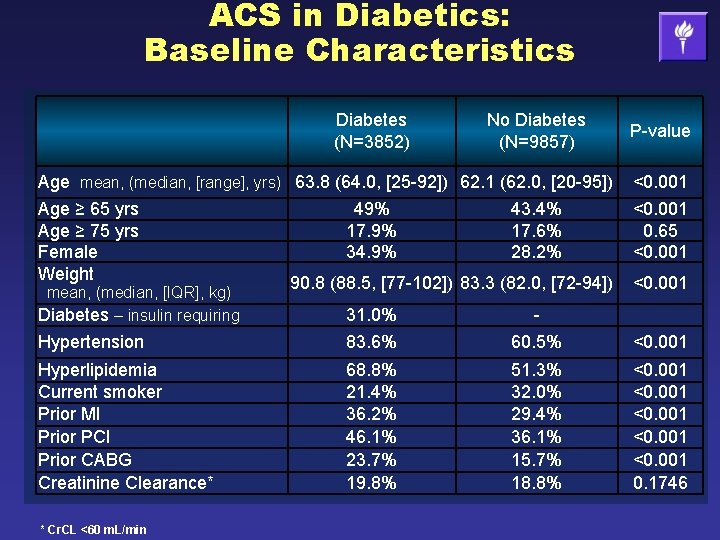
ACS in Diabetics: Baseline Characteristics Diabetes (N=3852) No Diabetes (N=9857) P-value Age mean, (median, [range], yrs) 63. 8 (64. 0, [25 -92]) 62. 1 (62. 0, [20 -95]) <0. 001 Age ≥ 65 yrs Age ≥ 75 yrs Female Weight <0. 001 0. 65 <0. 001 mean, (median, [IQR], kg) Diabetes – insulin requiring 49% 17. 9% 34. 9% 43. 4% 17. 6% 28. 2% 90. 8 (88. 5, [77 -102]) 83. 3 (82. 0, [72 -94]) <0. 001 Hypertension 31. 0% 83. 6% 60. 5% <0. 001 Hyperlipidemia Current smoker Prior MI Prior PCI Prior CABG Creatinine Clearance* 68. 8% 21. 4% 36. 2% 46. 1% 23. 7% 19. 8% 51. 3% 32. 0% 29. 4% 36. 1% 15. 7% 18. 8% <0. 001 0. 1746 * Cr. CL <60 m. L/min

ACS in Diabetics: 30 -Day Outcomes Diabetes vs. No Diabetes P = 0. 0002 †Heparin=unfractionated or enoxaparin P = 0. 0037 P = 0. 0002
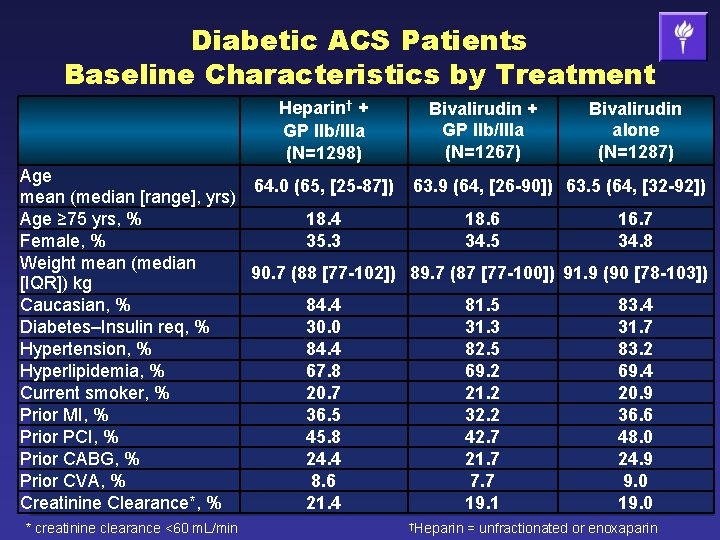
Diabetic ACS Patients Baseline Characteristics by Treatment Heparin† + GP IIb/IIIa (N=1298) Bivalirudin + GP IIb/IIIa (N=1267) Bivalirudin alone (N=1287) Age 64. 0 (65, [25 -87]) 63. 9 (64, [26 -90]) 63. 5 (64, [32 -92]) mean (median [range], yrs) Age ≥ 75 yrs, % 18. 4 18. 6 16. 7 Female, % 35. 3 34. 5 34. 8 Weight mean (median 90. 7 (88 [77 -102]) 89. 7 (87 [77 -100]) 91. 9 (90 [78 -103]) [IQR]) kg Caucasian, % 84. 4 81. 5 83. 4 Diabetes–Insulin req, % 30. 0 31. 3 31. 7 Hypertension, % 84. 4 82. 5 83. 2 Hyperlipidemia, % 67. 8 69. 2 69. 4 Current smoker, % 20. 7 21. 2 20. 9 Prior MI, % 36. 5 32. 2 36. 6 Prior PCI, % 45. 8 42. 7 48. 0 Prior CABG, % 24. 4 21. 7 24. 9 Prior CVA, % 8. 6 7. 7 9. 0 Creatinine Clearance*, % 21. 4 19. 1 19. 0 * creatinine clearance <60 m. L/min †Heparin = unfractionated or enoxaparin

Diabetic ACS Patients: 30 -Day Endpoints by Treatment Group Heparin* + GP IIb/IIIa vs. Bivalirudin + GP IIb/IIIa P = 0. 94 *Heparin = unfractionated or enoxaparin P = 0. 64 P = 0. 43

Diabetic ACS Patients: 30 -Day Endpoints Heparin* + GP IIb/IIIa vs. Bivalirudin alone P = 0. 02 *Heparin = unfractionated or enoxaparin P = 0. 39 P=0. 0002

Diabetic ACS Patients: Components of Ischemic Endpoint Heparin* + IIb/IIIa vs. Bivalirudin Alone P = 0. 39 P = 0. 32 P= 0. 27 P = 0. 37 *Heparin=unfractionated or enoxaparin 17
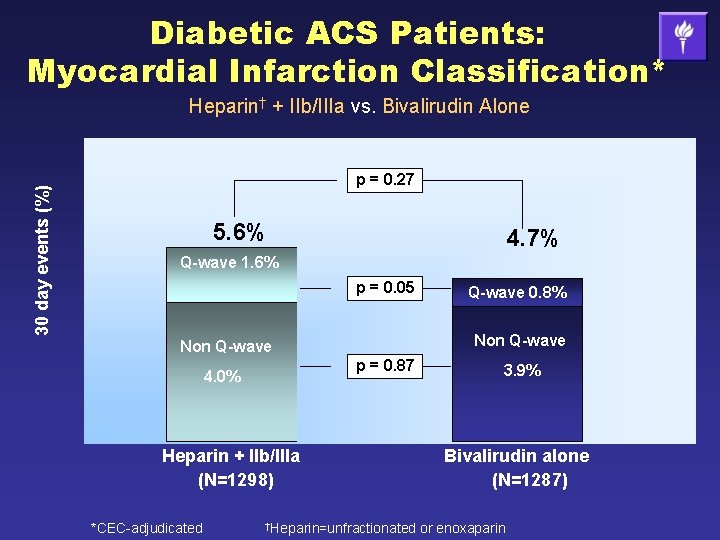
Diabetic ACS Patients: Myocardial Infarction Classification* 30 day events (%) Heparin† + IIb/IIIa vs. Bivalirudin Alone p = 0. 27 5. 6% 4. 7% Q-wave 1. 6% p = 0. 05 Non Q-wave p = 0. 87 4. 0% Heparin + IIb/IIIa (N=1298) *CEC-adjudicated Q-wave 0. 8% †Heparin=unfractionated 3. 9% Bivalirudin alone (N=1287) or enoxaparin
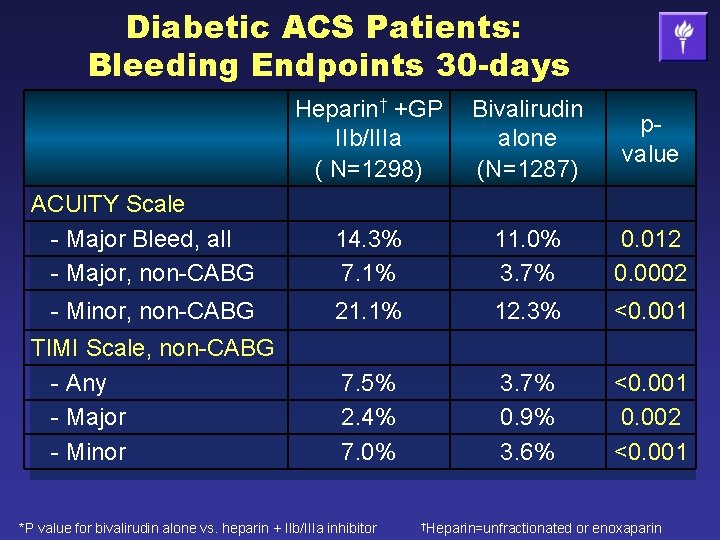
Diabetic ACS Patients: Bleeding Endpoints 30 -days Heparin† +GP IIb/IIIa ( N=1298) Bivalirudin alone (N=1287) pvalue ACUITY Scale - Major Bleed, all - Major, non-CABG 14. 3% 7. 1% 11. 0% 3. 7% 0. 012 0. 0002 - Minor, non-CABG 21. 1% 12. 3% <0. 001 TIMI Scale, non-CABG - Any - Major - Minor 7. 5% 2. 4% 7. 0% 3. 7% 0. 9% 3. 6% <0. 001 0. 002 <0. 001 *P value for bivalirudin alone vs. heparin + IIb/IIIa inhibitor †Heparin=unfractionated or enoxaparin

Insulin-dependent Diabetic ACS Patients: 30 -Day Endpoints by Treatment Group Heparin† + GP IIb/IIIa vs. Bivalirudin alone P = 0. 23 †Heparin=unfractionated or enoxaparin P = 0. 74 P = 0. 04

Diabetic Patients with ACS : Conclusions § Compared with non-diabetics, diabetic patients have worse net clinical outcomes at 30 days (12. 8% vs. 10. 5%; p=0. 0002), resulting from significantly higher rates of the composite ischemic end point (8. 6% vs. 7. 2%; p=0. 0037) and non-CABG major bleeding (5. 7% vs. 4. 2%; p=0. 0002) § In diabetic patients, compared with the standard of care, heparin (UFH or enoxaparin) + GPIIb/IIIa, bivalirudin + GPIIb/IIIa was not better for protection from ischemic events or bleeding and resulted in similar net clinical outcome

Diabetic Patients with ACS: Conclusions § Compared to those receiving the reference standard, diabetics receiving bivalirudin monotherapy, with provisional GPIIb/IIIa in 7. 9%, had similar protection from ischemic events (7. 8% vs. 8. 8%; p=0. 39) and a marked reduction in major bleeding (3. 7% vs. 7. 1%; p=0. 0002) with improved net clinical outcome (10. 8% vs. 13. 7%; p=0. 02) § These 30 -day outcomes suggest that bivalirudin monotherapy is safe and effective for diabetic patients with ACS, including those requiring insulin § One-year clinical and economic data will determine whether this regimen will become the standard of care for these patients.
- Slides: 19