BISC 367 Plant Physiology Lab Spring 2009 Plant

BISC 367 - Plant Physiology Lab Spring 2009 Plant Biology Fall 2006 BISC 367 Notices: • Photosynthesis lab report due Feb. 09 • Lecture test Feb 10 • Please email water relations data to Doug Wilson & myself • Reading material (Taiz & Zeiger): • Chapter 12 assimilation of mineral nutrients

BISC 367 Water relations data Plant Treatment Pressure Bomb (Individual readings) Poplar dry wet 1. 80314 and 3. 039, 2. 3 1. 26625/1. 16495/0. 9117, 1. 25 Geranium dry wet 0. 75975/0. 82053/0. 72936. 38494/. 51663/. 48624. Bean Corn Leaf Press (MPa) Average 35, 39, 55 1. 1143 60, 85, 52, 29, 30 0. 86105 0. 35455 - Osomometer Osmometer Reading (mmol/Kg) Average 0. 2965 804, 1007 905. 5 0. 4528 632, 589, 588 603 Pressure Bomb (MPa) Leaf Press (psi) (average) Individual readings 0. 76988 73, 85, 60 0. 462603333 90, 58, 62, 45 0. 86105 42 0. 35455 37 1 psi=0. 00689476 MPa 35 39 55 The units are MPa The sign is NEGATIVE These readings look good Combine with data from other group 0. 5447 294, 293, 274, 298 0. 4826 280, 263, 299, 272 0. 2896 405 0. 2551 353 289. 75 278. 5 - 0. 2413 0. 2689 0. 3792 60 85 52 73 85 0. 4137 0. 5861 0. 3585 0. 5033 0. 5861 90 0. 6205 58 62 42 37 0. 3999 0. 4275 0. 2896 0. 2551 ? ? ? This data doesn't jive with the pressure bomb I agree! Convert to Ys using van’t Hoffs eqtn Poplar, bean Ys is lower for dry but not for gernanium – why?

Measuring Yw BISC 367 Relative water content Assesses the water content of plant tissues as a fraction of the fully turgid water content • relevant when considering metabolic / physiological aspects of water deficit stress Considered to be a better indicator of water status and physiological activity Captures effects of osmotic adjustment • Osmotic adjustment lowers the Yw at which a given RWC is reached Simple technique: • Leaf disks are excised, weighed (W) then allowed to reach full turgidity and re-weighed (TW). disks are dried to obtain their dry weight (DW). RWC (%) = [(W – DW) / (TW – DW)] X 100 Leaf

BISC 367 Water uptake by roots • Water crosses the roots using 3 possible pathways – Apoplastic pathway • Water moves via cell walls – Symplastic pathway • Water moves through the cells passing through the plasmodesmata – Transmembrane pathway • Water moves through cells but independently enters and exits each cell
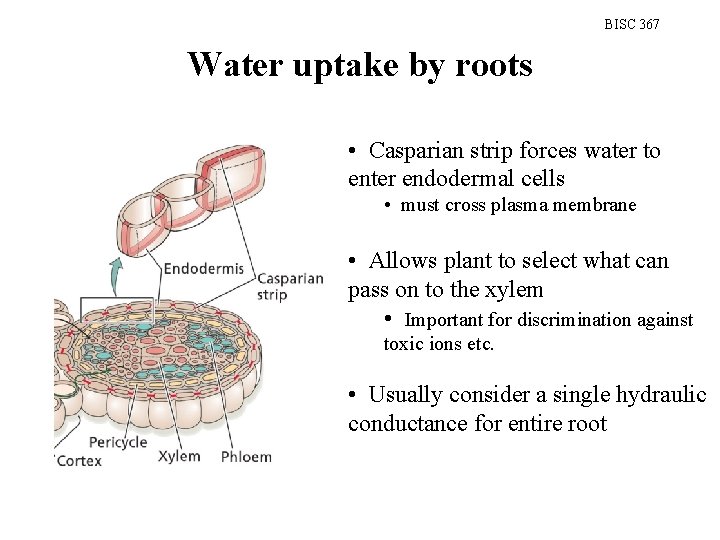
BISC 367 Water uptake by roots • Casparian strip forces water to enter endodermal cells • must cross plasma membrane • Allows plant to select what can pass on to the xylem • Important for discrimination against toxic ions etc. • Usually consider a single hydraulic conductance for entire root

BISC 367 Water movement - an overview

Inorganic ions in the soil • Soil particles carry a negative charge – Bind cations Anions are not readily bound • NO 3 - is soluble • PO 42 - binds to Al 3+ or Fe 3+ and can be unavailable • SO 42 - reacts with Ca 2+ to form gypsum (Ca. SO 4)

Ion transport across the root • Ions can cross the root in the apoplast or symplast – All ions enter the symplast at the endodermis before entering the stele (vascular tissue) • To enter the cells of the xylem ions must move back to the apoplast Note: the casparian strip: – prevents outward movement of ions – Can allow a higher level of ions to build in the xylem relative to the soil

Ion uptake into a cell • Driving force for ion uptake is the electrochemical gradient – Conc. gradient across membrane – Electrical gradient across membrane • At eqm the conc. difference across the membrane is balanced by the electrical difference – Calculate electric potential for given ion using Nernst equation • All living cells have an electrical difference across the membrane - membrane potential

Ion uptake into a cell • Membrane potential is established by several ions coming to “eqm” – Ability to come to eqm (or steady state) is influenced by membrane transport processes • Only K+ is close to eqm. • Anions have a higher than predicted conc • Cations have a lower than predicted conc

Ion uptake into a cell • Membrane potential is set by: – Passive diffusion – Electrogenic pumping (primarily H+) • H+-ATPases – Located on PM (plasma membrane) - P-ATPases • Pump H+ into cell wall – and tonoplast (membrane surrounding vacuole) - V-ATPases • Pump H+ into vacuole

Ion uptake into a cell • H+ gradients drive 2 o transport across PM and tonoplast • In vacuole [H+] is high: – Anions move in to balance charge – Ys falls – Water moves in - turgor increases – H+-pyrophosphatase also moves H+ into vacuole • Utilize energy of PPi hydrolysis

Ion Composition • K+ acquired passively • Na+ actively pumped out to apoplast and vacuole • H+ actively pumped out to apoplast and vacuole – Acidic apoplast and vacuole, neutral cytoplasm (regulates cell p. H) • Anions are actively acquired • Ca 2+ is actively pumped out Passive transport Active transport
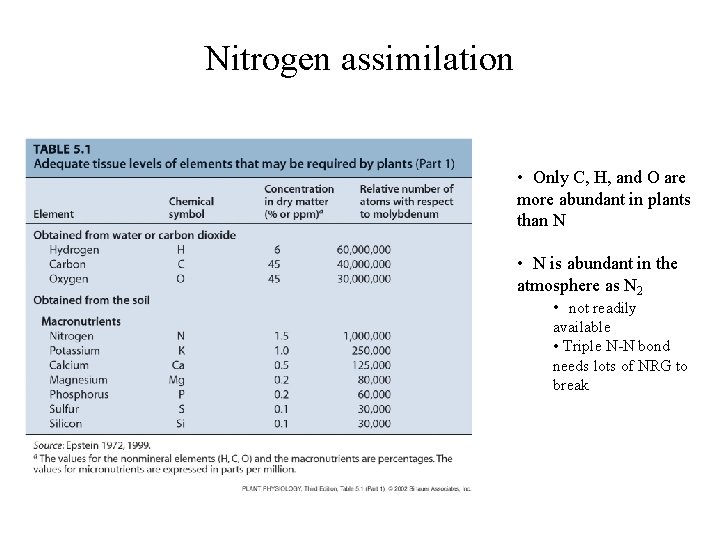
Nitrogen assimilation • • Only C, H, and O are more abundant in plants than N • N is abundant in the atmosphere as N 2 • not readily available • Triple N-N bond needs lots of NRG to break

Nutrient assimilation • Energetically costly! – NO 3 - reduction to NH 4+ utilizes 25% of a plants NRG requirements – Requires large amounts of reductant • Most occurs in stroma of chloroplast (cp) • Dependent on photosynthetic e- transport • photoassimilation

Nitrogen assimilation • Nitrate uptake is inducible: • Low and high affinity carriers exist • Carriers are synthesized in response to external NO 3 and is influenced by: • plant N status • form of N available in the soil • Sustained protein synthesis is necessary • NO 3 that enters root cells has 3 fates • Storage in the vacuole • Assimilation in root cells • Translocation in the xylem for assim. in leaf cells
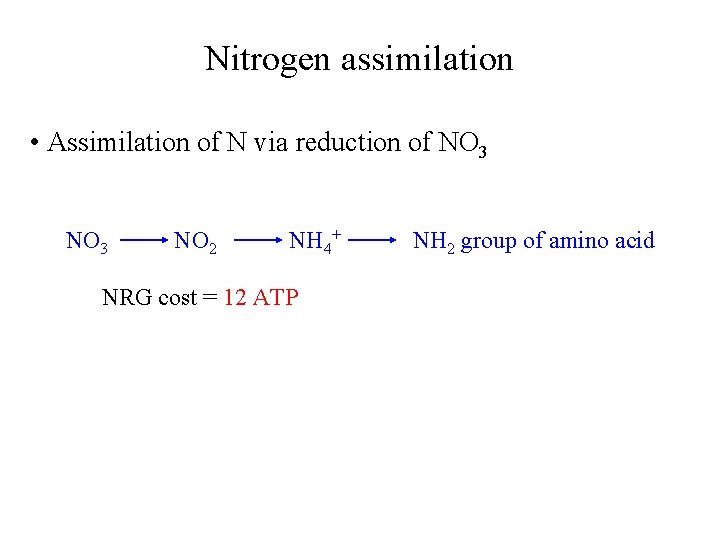
Nitrogen assimilation • Assimilation of N via reduction of NO 3 NO 2 NH 4+ NRG cost = 12 ATP NH 2 group of amino acid
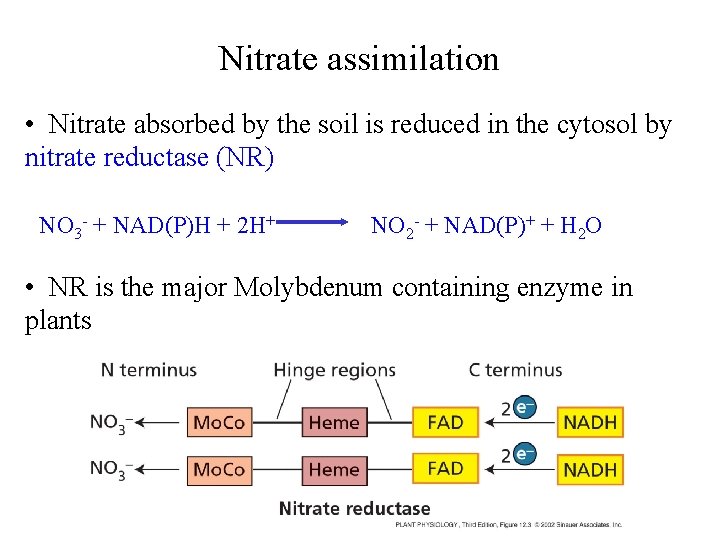
Nitrate assimilation • Nitrate absorbed by the soil is reduced in the cytosol by nitrate reductase (NR) NO 3 - + NAD(P)H + 2 H+ NO 2 - + NAD(P)+ + H 2 O • NR is the major Molybdenum containing enzyme in plants

Nitrate assimilation • NR is tightly regulated: • Gene transcription and enzyme activation are stimulated by: • NO 3 • Light • enhances activation by NO 3 • links NO 3 assimilation with NRG • CHO • Inactivation of NR is stimulated by: • Dark • Mg 2+ • NR is regulated by a NR kinase • phosphorylated and non-phosphorylated states are active • if the phosphorylated form is transferred to darkness an inhibitor switches NR off • activity is restored in the light by: • inhibitor release • phosphatase
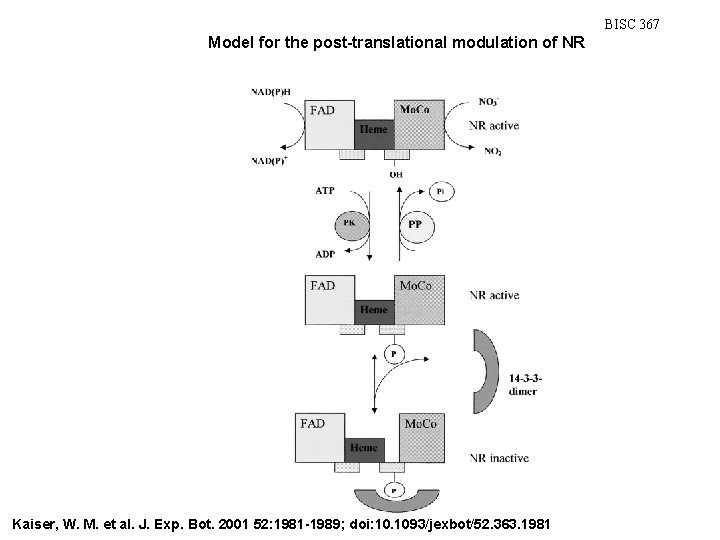
BISC 367 Model for the post-translational modulation of NR Kaiser, W. M. et al. J. Exp. Bot. 2001 52: 1981 -1989; doi: 10. 1093/jexbot/52. 363. 1981
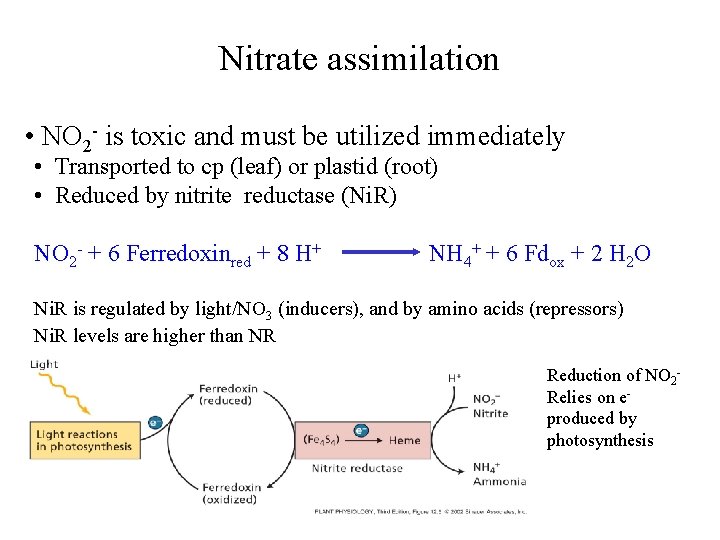
Nitrate assimilation • NO 2 - is toxic and must be utilized immediately • Transported to cp (leaf) or plastid (root) • Reduced by nitrite reductase (Ni. R) NO 2 - + 6 Ferredoxinred + 8 H+ NH 4+ + 6 Fdox + 2 H 2 O Ni. R is regulated by light/NO 3 (inducers), and by amino acids (repressors) Ni. R levels are higher than NR Reduction of NO 2 Relies on eproduced by photosynthesis
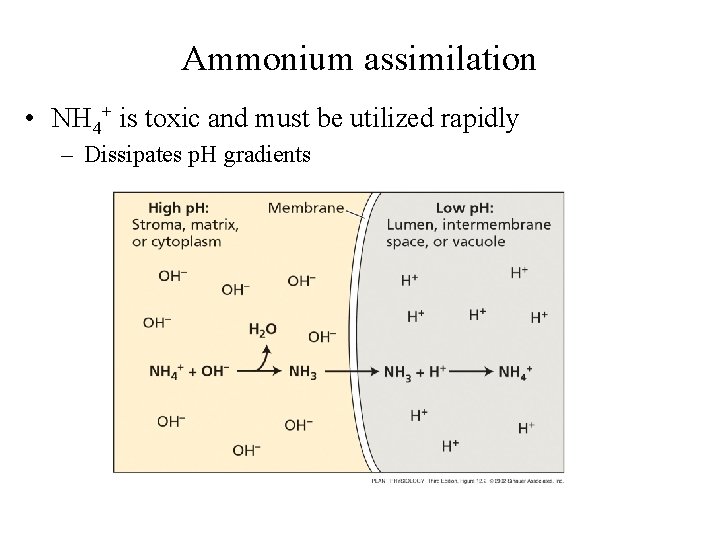
Ammonium assimilation • NH 4+ is toxic and must be utilized rapidly – Dissipates p. H gradients

Ammonium assimilation • Glutamine synthetase (GS) combines NH 4+ and glutamate Glu + NH 4+ + ATP Glutamine + ADP + Pi • Glutamate synthase (GOGAT) transfers the amide of glutamine to 2 oxoglutarate Glutamine + 2 -oxoglutarate + Fdred/NADH 2 glutamate + Fdox • Transamination rxns transfer amide N to other amino acids Glu + oxaloacetate aspartate and 2 -oxoglutarate
- Slides: 23