BIRADS CADRADS LIRADS LungRADS NIRADS ORADS PIRADS TIRADS

BI-RADS, CAD-RADS, LI-RADS, Lung-RADS, NI-RADS, O-RADS, PI-RADS, TI-RADS: Reporting and Data Systems Julie Y. An, MD Kyle M. L. Unsdorfer, MD Jeffrey C. Weinreb, MD An earlier incorrect version of this presentation appeared online. This presentation was corrected on September 12, 2019.

Author Affiliations From the Department of Medicine, Akron, Ohio 44304 (J. Y. A. ); Department of Radiology, Mayo Clinic, Rochester, Minn (K. M. L. U. ); and Department of Radiology, Yale University School of Medicine, New Haven, Conn (J. C. W. ). Address correspondence to: J. Y. A. Current address: Department of Radiology, University of California San Diego 200 W Arbor Drive, San Diego, CA 92103 (e-mail: j 1 an@ucsd. edu) 2018 RSNA Educational Exhibit: MS 101 Acknowledgments. —The authors would like to thank Philip Araoz, MD, Chi Wan Koo, MD, and Baris Turkbey, MD for their guidance and review of this presentation. Disclosures of Conflicts of Interest. —J. C. W. Activities related to the present article: member of the ACR LI-RADS and PI-RADS committees; received reimbursement for travel expenses for committee meetings from the ACR. Activities not related to the present article: disclosed no relevant relationships. Other activities: disclosed no relevant relationships.

Learning Objectives 1. Review the overview of the current American College of Radiology (______)* Reporting and Data Systems (RADS). 2. Recognize the systematic scoring and modifiers of RADS. 3. Review the general themes and key imaging components of each system. 4. Identify additional resources for more in-depth training and education. Note. —*Reprinted, with permission, from the ACR.

Why Standardized Reporting? • Unifies the language between radiologists and clinicians • Allows consistent data collection and improvement of diagnostic parameters • Helps to standardize care • Adds value by providing clinical recommendations and guidance

What is ______RADS? • RADS include systems with standardized terminology, assessment, and reporting endorsed by the ACR. • The predominant focus is on cancer imaging (breast, colon, head and neck, liver, lung, ovarian, prostate, and thyroid cancers), with additional systems focusing on coronary artery disease and head injuries (pending publication). • Systems were devised by expert consensus and may be updated periodically.

“The goal of the____ RADS is to reduce the variability of terminology in reports and to ease communication between radiologists and referring physicians. ” 1

ACR RADS Abbreviations BI-RADS = Breast Imaging Reporting and Data System C-RADS = CT Colonography Reporting and Data System CAD-RADS = Coronary Artery Disease Reporting and Data System LI-RADS = Liver Imaging Reporting and Data System NI-RADS = Neck Imaging Reporting and Data System O-RADS = Ovarian-Adnexal Reporting and Data System PI-RADS = Prostate Imaging Reporting and Data System TI-RADS =Thyroid Imaging Reporting and Data System
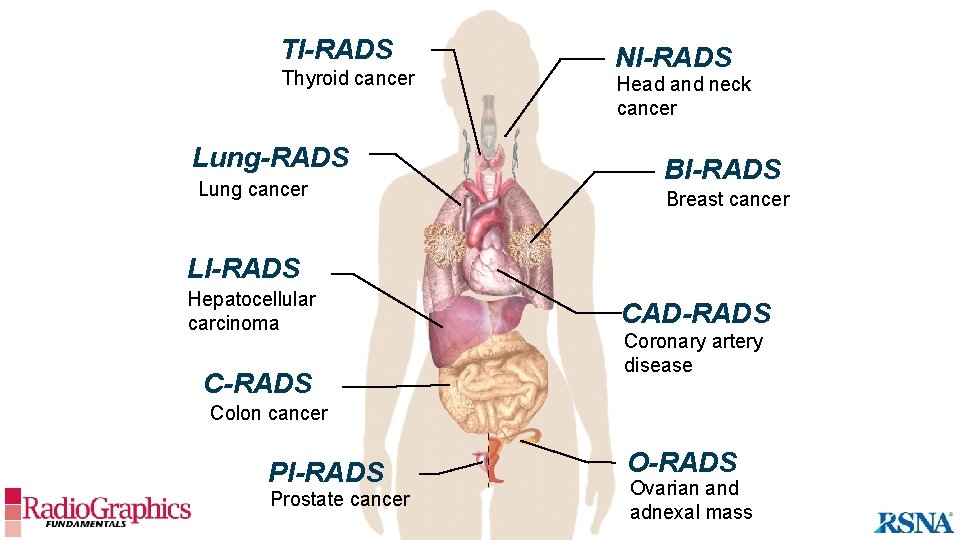
TI-RADS Thyroid cancer Lung-RADS Lung cancer NI-RADS Head and neck cancer BI-RADS Breast cancer LI-RADS Hepatocellular carcinoma C-RADS CAD-RADS Coronary artery disease Colon cancer PI-RADS Prostate cancer O-RADS Ovarian and adnexal mass
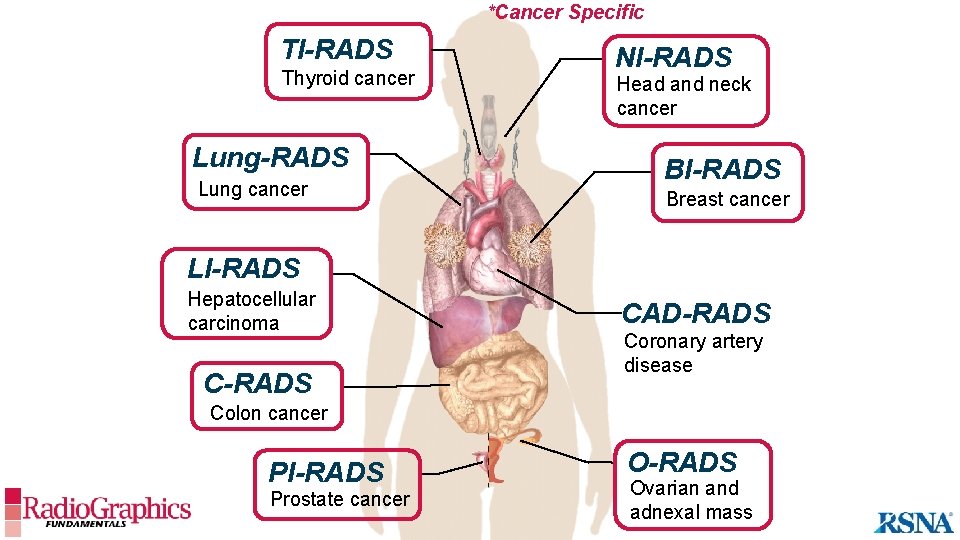
*Cancer Specific TI-RADS Thyroid cancer Lung-RADS Lung cancer NI-RADS Head and neck cancer BI-RADS Breast cancer LI-RADS Hepatocellular carcinoma C-RADS CAD-RADS Coronary artery disease Colon cancer PI-RADS Prostate cancer O-RADS Ovarian and adnexal mass
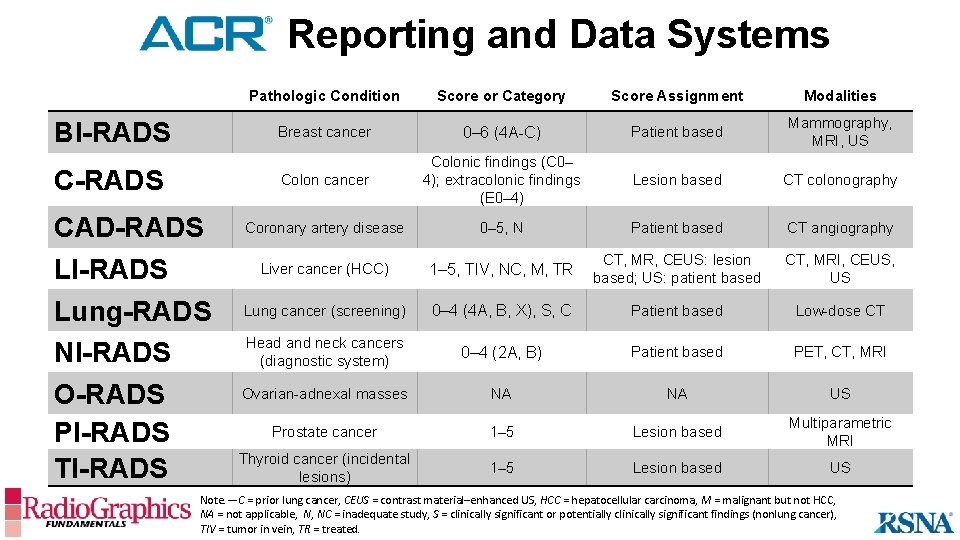
_______Reporting and Data Systems BI-RADS CAD-RADS LI-RADS Lung-RADS NI-RADS O-RADS PI-RADS TI-RADS Pathologic Condition Score or Category Score Assignment Modalities Breast cancer 0– 6 (4 A-C) Patient based Mammography, MRI, US Colon cancer Colonic findings (C 0– 4); extracolonic findings (E 0– 4) Lesion based CT colonography Coronary artery disease 0– 5, N Patient based CT angiography Liver cancer (HCC) 1– 5, TIV, NC, M, TR CT, MR, CEUS: lesion based; US: patient based CT, MRI, CEUS, US Lung cancer (screening) 0– 4 (4 A, B, X), S, C Patient based Low-dose CT Head and neck cancers (diagnostic system) 0– 4 (2 A, B) Patient based PET, CT, MRI Ovarian-adnexal masses NA NA US Prostate cancer 1– 5 Lesion based Multiparametric MRI Thyroid cancer (incidental lesions) 1– 5 Lesion based US Note. —C = prior lung cancer, CEUS = contrast material–enhanced US, HCC = hepatocellular carcinoma, M = malignant but not HCC, NA = not applicable, N, NC = inadequate study, S = clinically significant or potentially clinically significant findings (nonlung cancer), TIV = tumor in vein, TR = treated.

The Systematic Scoring 1 of______RADS

RADS Scoring Scale Pathologic Condition Score or Category Score Assignment Modalities Mammography, Breast cancer Patient based 0– 6 (4 A-C) BI-RADS MRI, US Colon cancer C 0 -4, E 0 -4 Lesion-based CT Colonography C-RADS 0 -5, N Patient-based CTA CAD-RADS Coronary artery disease CT, MR, CEUS: Lesion, CT, MRI, CEUS, Liver cancer: HCC 1 -5, TIV, NC, M, TR LI-RADS US: Patient-based US ____ Lung cancer: screening ____ Patient-based Low-dose CT 0 -4 (4 A, B, X), ____ S, C Lung-RADS Head & neck cancers: Patient-based PET, CT, MRI 0 -4 (2 A, B) NI-RADS diagnostic Increased likelihood of disease Ovarian-Adnexal masses N/A Ultrasound O-RADS Multiparametric Prostate cancer 1 -5 Lesion-based PI-RADS MRI Thyroid cancer: incidental 1 -5 Lesion-based Ultrasound TI-RADS lesions
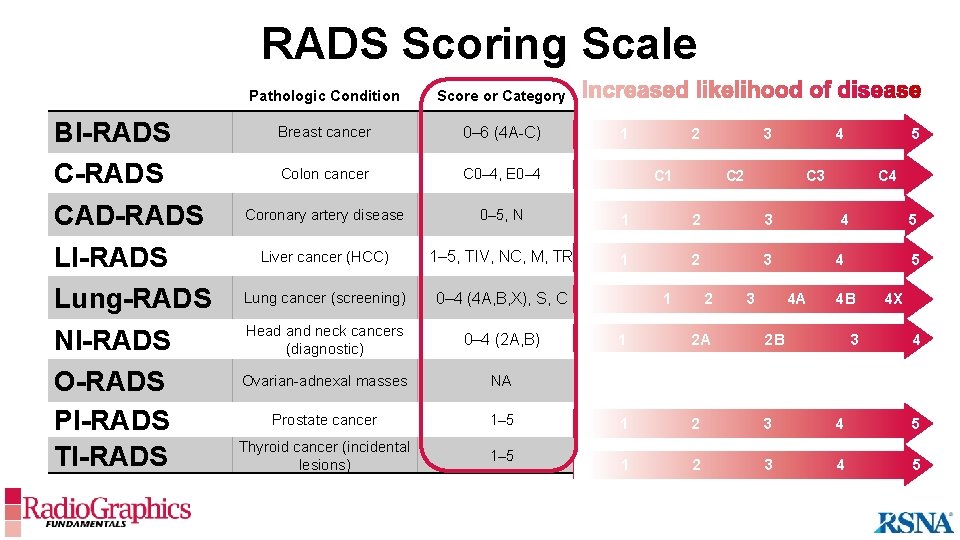
RADS Scoring Scale BI-RADS CAD-RADS LI-RADS Lung-RADS NI-RADS O-RADS PI-RADS TI-RADS Pathologic Condition Score or Category Breast cancer 0– 6 (4 A-C) Colon cancer C 0– 4, E 0– 4 Coronary artery disease 0– 5, N Liver cancer (HCC) 1– 5, TIV, NC, M, TR Lung cancer (screening) 0– 4 (4 A, B, X), S, C Head and neck cancers (diagnostic) 0– 4 (2 A, B) Ovarian-adnexal masses NA Prostate cancer 1– 5 Thyroid cancer (incidental lesions) 1– 5 Score Assignment Modalities Mammo, MRI, 3 Ultrasound 4 (US) 1 Patient-based 2 Lesion-based C 1 C 2 CT Colonography C 3 C 4 1 Patient-based 2 3 CTA 4 CT, MR, CEUS: Lesion, CT, MRI, CEUS, 1 2 3 4 US: Patient-based US Patient-based 1 2 1 Patient-based 2 A Lesion-based 2 1 5 5 Low-dose 4 A 4 B CT 4 X 3 2 B PET, CT, 3 MRI N/A 1 Lesion-based 2 5 4 Ultrasound 3 Multiparametric 4 MRI 5 3 Ultrasound 4 5

Nondiagnostic Scores BI-RADS CAD-RADS* LI-RADS Lung-RADS NI-RADS O-RADS PI-RADS TI-RADS Pathologic condition Score or Category Score Assignment Modalities Breast cancer 0– 6 (4 A-C) Patient-based Mammo, MRI, Ultrasound (US) Colon cancer C 0– 4, E 0 -4 Lesion-based CT Colonography Coronary artery disease 0– 5, N Liver cancer (HCC) 1– 5, TIV, NC, M, TR Lung cancer (screening) 0– 4 (4 A, B, X), S, C Patient-based Low-dose CT Head and neck cancers (diagnostic) 0– 4 (2 A, B) Patient-based PET, CT, MRI Ovarian-adnexal masses NA *CAD-RADS 0 = Exception, N/Aan inadequate. Ultrasound not study Prostate cancer 1– 5 Lesion-based Multiparametric MRI Thyroid cancer (incidental lesions) 1– 5 Lesion-based Ultrasound 0, N, NC Inadequate study, requires CTA Patient-based repeat or CT, MR, CEUS: Lesion, CT, MRI, CEUS, additional US: Patient-based US imaging
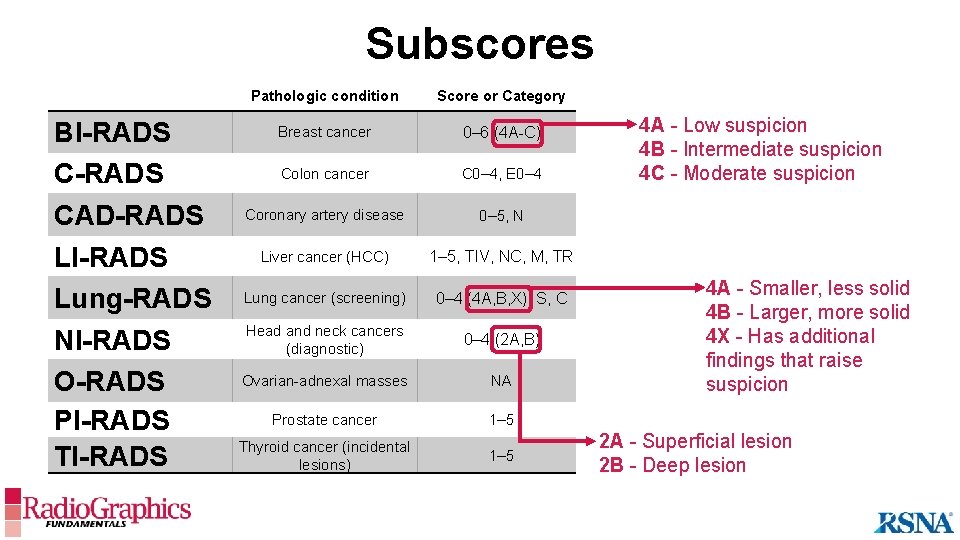
Subscores BI-RADS CAD-RADS LI-RADS Lung-RADS NI-RADS O-RADS PI-RADS TI-RADS Pathologic condition Score or Category Score Assignment Modalities Breast cancer 0– 6 (4 A-C) Colon cancer C 0– 4, E 0– 4 Coronary artery disease 0– 5, N Patient-based CTA Liver cancer (HCC) 1– 5, TIV, NC, M, TR CT, MR, CEUS: Lesion, US: Patient-based CT, MRI, CEUS, US Lung cancer (screening) 0– 4 (4 A, B, X), S, C Patient-based Head and neck cancers (diagnostic) 0– 4 (2 A, B) Ovarian-adnexal masses NA 4 A - Smaller, less solid Low-dose CT 4 B - Larger, more solid 4 X - Has additional Patient-based PET, CT, MRI findings that raise N/A suspicion Ultrasound Prostate cancer 1– 5 Lesion-based Thyroid cancer (incidental lesions) 1– 5 Mammo, MRI, 4 A - Low suspicion Patient-based Ultrasound (US) 4 B - Intermediate suspicion Lesion-based CT Colonography 4 C - Moderate suspicion Multiparametric MRI 2 A - Superficial lesion Lesion-based 2 B - Deep lesion Ultrasound
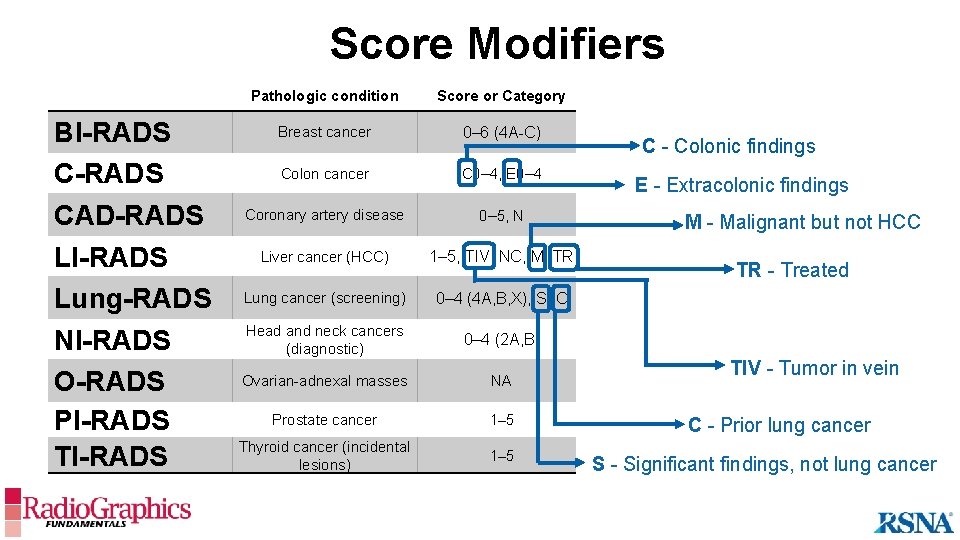
Score Modifiers BI-RADS CAD-RADS LI-RADS Lung-RADS NI-RADS O-RADS PI-RADS TI-RADS Pathologic condition Score or Category Score Assignment Breast cancer 0– 6 (4 A-C) Patient-based Colon cancer C 0– 4, E 0– 4 Lesion-based Coronary artery disease 0– 5, N Patient-based Liver cancer (HCC) 1– 5, TIV, NC, M, TR CT, MR, CEUS: Lesion, US: Patient-based. TR Lung cancer (screening) 0– 4 (4 A, B, X), S, C Patient-based Low-dose CT Head and neck cancers (diagnostic) 0– 4 (2 A, B) Patient-based PET, CT, MRI Ovarian-adnexal masses NA N/A Prostate cancer 1– 5 Thyroid cancer (incidental lesions) 1– 5 C - Colonic Modalities Mammo, MRI, Ultrasound (US) findings CT Colonography E - Extracolonic findings CTA M - Malignant but not HCC - CT, MRI, CEUS, US Treated TIV - Tumor in vein Lesion-based C - Prior Ultrasound Multiparametric lung cancer MRI Lesion-based Ultrasound S - Significant findings, not lung cancer

Imaging Examples 1 of ______RADS
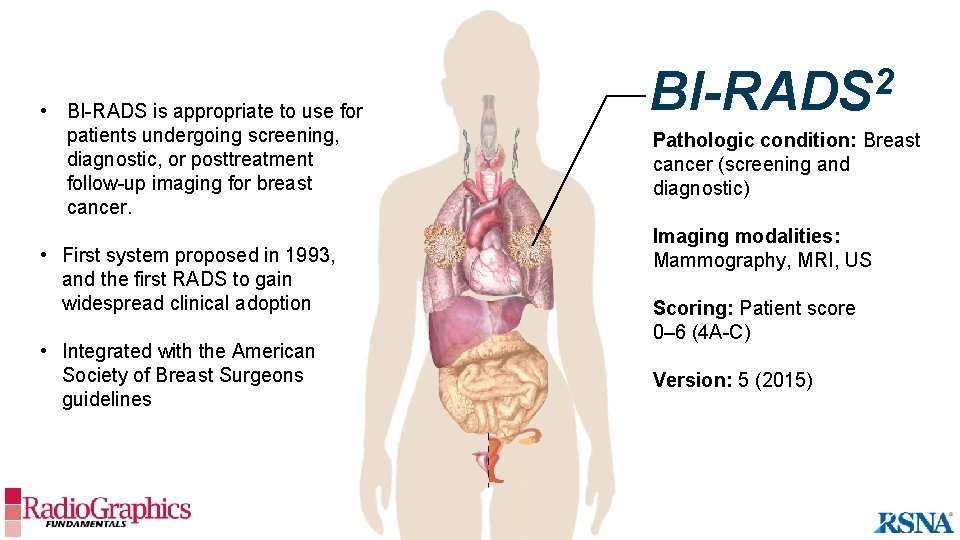
• BI-RADS is appropriate to use for patients undergoing screening, diagnostic, or posttreatment follow-up imaging for breast cancer. • First system proposed in 1993, and the first RADS to gain widespread clinical adoption • Integrated with the American Society of Breast Surgeons guidelines 2 BI-RADS Pathologic condition: Breast cancer (screening and diagnostic) Imaging modalities: Mammography, MRI, US Scoring: Patient score 0– 6 (4 A-C) Version: 5 (2015)
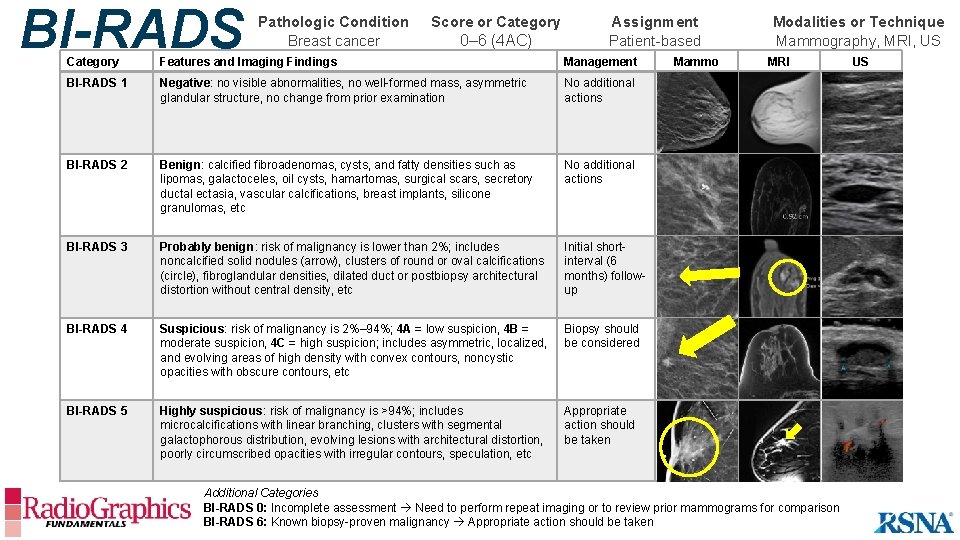
BI-RADS Pathologic Condition Breast cancer Score or Category 0– 6 (4 AC) Assignment Patient-based Category Features and Imaging Findings Management BI-RADS 1 Negative: no visible abnormalities, no well-formed mass, asymmetric glandular structure, no change from prior examination No additional actions BI-RADS 2 Benign: calcified fibroadenomas, cysts, and fatty densities such as lipomas, galactoceles, oil cysts, hamartomas, surgical scars, secretory ductal ectasia, vascular calcifications, breast implants, silicone granulomas, etc No additional actions BI-RADS 3 Probably benign: risk of malignancy is lower than 2%; includes noncalcified solid nodules (arrow), clusters of round or oval calcifications (circle), fibroglandular densities, dilated duct or postbiopsy architectural distortion without central density, etc Initial shortinterval (6 months) followup BI-RADS 4 Suspicious: risk of malignancy is 2%– 94%; 4 A = low suspicion, 4 B = moderate suspicion, 4 C = high suspicion; includes asymmetric, localized, and evolving areas of high density with convex contours, noncystic opacities with obscure contours, etc Biopsy should be considered BI-RADS 5 Highly suspicious: risk of malignancy is >94%; includes microcalcifications with linear branching, clusters with segmental galactophorous distribution, evolving lesions with architectural distortion, poorly circumscribed opacities with irregular contours, speculation, etc Appropriate action should be taken Mammo Modalities or Technique Mammography, MRI, US MRI Additional Categories BI-RADS 0: Incomplete assessment Need to perform repeat imaging or to review prior mammograms for comparison BI-RADS 6: Known biopsy-proven malignancy Appropriate action should be taken US
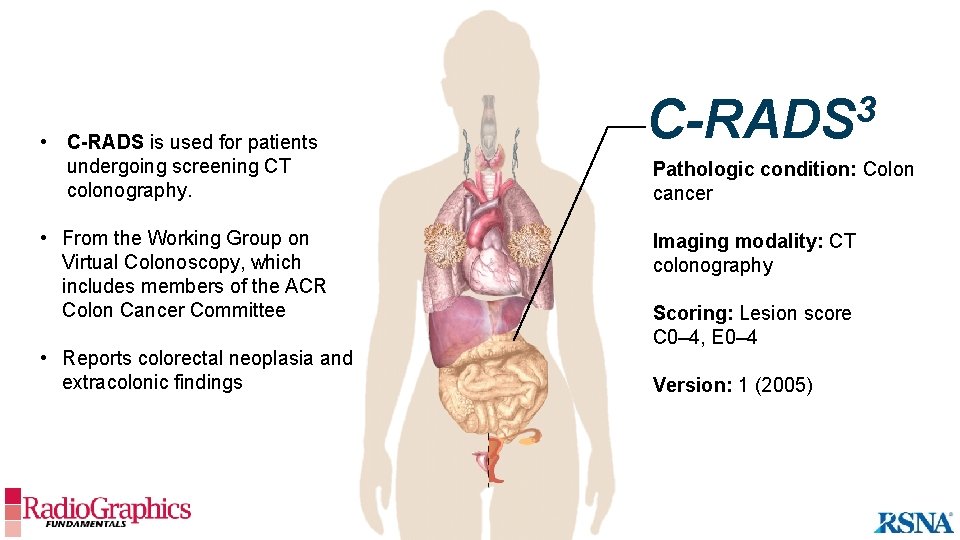
• C-RADS is used for patients undergoing screening CT colonography. • From the Working Group on Virtual Colonoscopy, which includes members of the ACR Colon Cancer Committee • Reports colorectal neoplasia and extracolonic findings 3 C-RADS Pathologic condition: Colon cancer Imaging modality: CT colonography Scoring: Lesion score C 0– 4, E 0– 4 Version: 1 (2005)
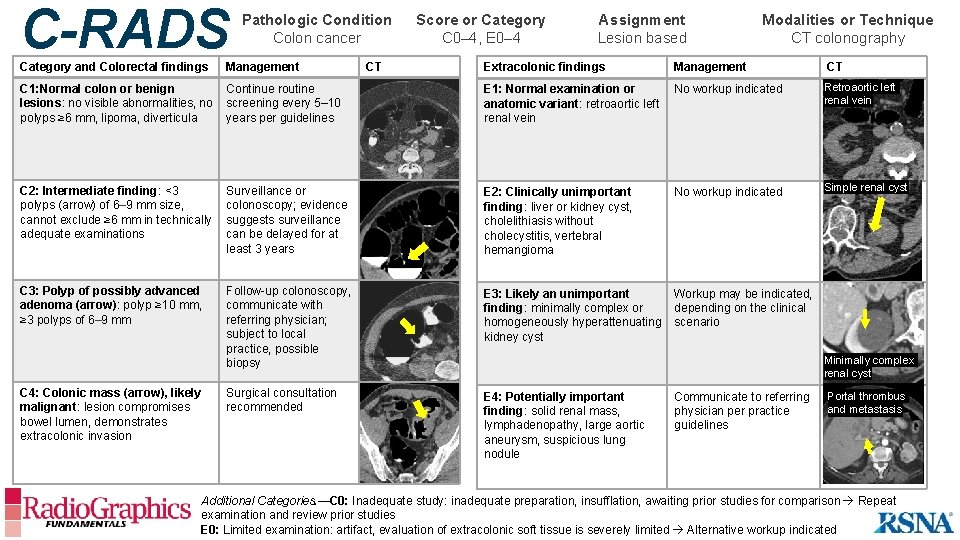
C-RADS Pathologic Condition Colon cancer Score or Category C 0– 4, E 0– 4 Assignment Lesion based Modalities or Technique CT colonography Extracolonic findings Management CT Continue routine screening every 5– 10 years per guidelines E 1: Normal examination or anatomic variant: retroaortic left renal vein No workup indicated Retroaortic left renal vein C 2: Intermediate finding: <3 polyps (arrow) of 6– 9 mm size, cannot exclude ≥ 6 mm in technically adequate examinations Surveillance or colonoscopy; evidence suggests surveillance can be delayed for at least 3 years E 2: Clinically unimportant finding: liver or kidney cyst, cholelithiasis without cholecystitis, vertebral hemangioma No workup indicated Simple renal cyst C 3: Polyp of possibly advanced adenoma (arrow): polyp ≥ 10 mm, ≥ 3 polyps of 6– 9 mm Follow-up colonoscopy, communicate with referring physician; subject to local practice, possible biopsy E 3: Likely an unimportant finding: minimally complex or homogeneously hyperattenuating kidney cyst Workup may be indicated, depending on the clinical scenario Surgical consultation recommended E 4: Potentially important finding: solid renal mass, lymphadenopathy, large aortic aneurysm, suspicious lung nodule Category and Colorectal findings Management C 1: Normal colon or benign lesions: no visible abnormalities, no polyps ≥ 6 mm, lipoma, diverticula C 4: Colonic mass (arrow), likely malignant: lesion compromises bowel lumen, demonstrates extracolonic invasion CT Minimally complex renal cyst Communicate to referring physician per practice guidelines Portal thrombus and metastasis Additional Categories. —C 0: Inadequate study: inadequate preparation, insufflation, awaiting prior studies for comparison Repeat examination and review prior studies E 0: Limited examination: artifact, evaluation of extracolonic soft tissue is severely limited Alternative workup indicated
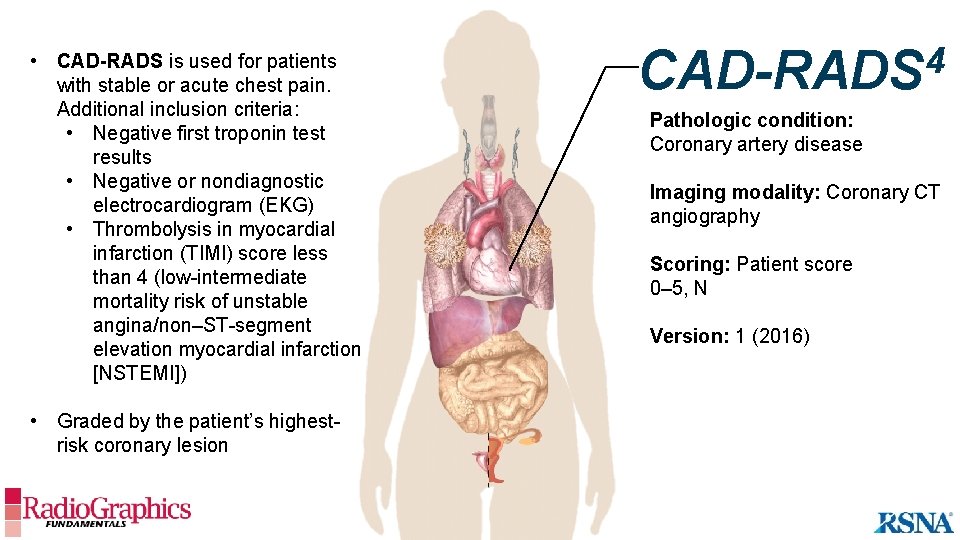
• CAD-RADS is used for patients with stable or acute chest pain. Additional inclusion criteria: • Negative first troponin test results • Negative or nondiagnostic electrocardiogram (EKG) • Thrombolysis in myocardial infarction (TIMI) score less than 4 (low-intermediate mortality risk of unstable angina/non–ST-segment elevation myocardial infarction [NSTEMI]) • Graded by the patient’s highestrisk coronary lesion 4 CAD-RADS Pathologic condition: Coronary artery disease Imaging modality: Coronary CT angiography Scoring: Patient score 0– 5, N Version: 1 (2016)
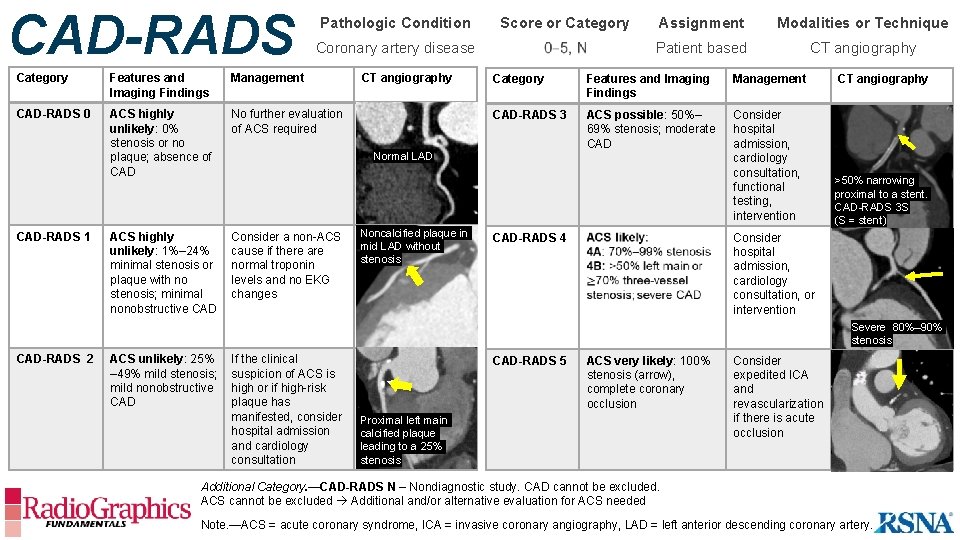
CAD-RADS Pathologic Condition Coronary artery disease Category Features and Imaging Findings Management CAD-RADS 0 ACS highly unlikely: 0% stenosis or no plaque; absence of CAD No further evaluation of ACS required ACS highly unlikely: 1%– 24% minimal stenosis or plaque with no stenosis; minimal nonobstructive CAD Consider a non-ACS cause if there are normal troponin levels and no EKG changes CAD-RADS 1 Score or Category CT angiography Modalities or Technique Patient based CT angiography Category Features and Imaging Findings Management CT angiography CAD-RADS 3 ACS possible: 50%– 69% stenosis; moderate CAD Consider hospital admission, cardiology consultation, functional testing, intervention >50% narrowing proximal to a stent. CAD-RADS 3 S (S = stent) Normal LAD Noncalcified plaque in mid LAD without stenosis Assignment CAD-RADS 4 Consider hospital admission, cardiology consultation, or intervention Severe 80%– 90% stenosis CAD-RADS 2 ACS unlikely: 25% – 49% mild stenosis; mild nonobstructive CAD If the clinical suspicion of ACS is high or if high-risk plaque has manifested, consider hospital admission and cardiology consultation CAD-RADS 5 ACS very likely: 100% stenosis (arrow), complete coronary occlusion Proximal left main calcified plaque leading to a 25% stenosis Consider expedited ICA and revascularization if there is acute occlusion Additional Category. —CAD-RADS N – Nondiagnostic study. CAD cannot be excluded. ACS cannot be excluded Additional and/or alternative evaluation for ACS needed Note. —ACS = acute coronary syndrome, ICA = invasive coronary angiography, LAD = left anterior descending coronary artery.
• LI-RADS applies to patients at high risk for HCC (eg, those patients with hepatitis B or C, cirrhosis from nonalcoholic steatohepatitis [NASH], alcohol, or prior HCC • Excludes patients younger than 18 years of age, those with no risk factors and/or cirrhosis, vasculopathies, or Budd-Chiari syndrome • Integrated with the American Association for the Study of Liver Diseases (AASLD) guidelines 5– 7 LI-RADS Pathologic condition: HCC Imaging modalities: CT, MRI, US, contrast-enhanced US Scoring: CT, MRI, contrast-enhanced US = lesion score, 1– 5, TIV, NC, M, T US = Patient score, 1– 3 Version: 3 (2017– 2018)
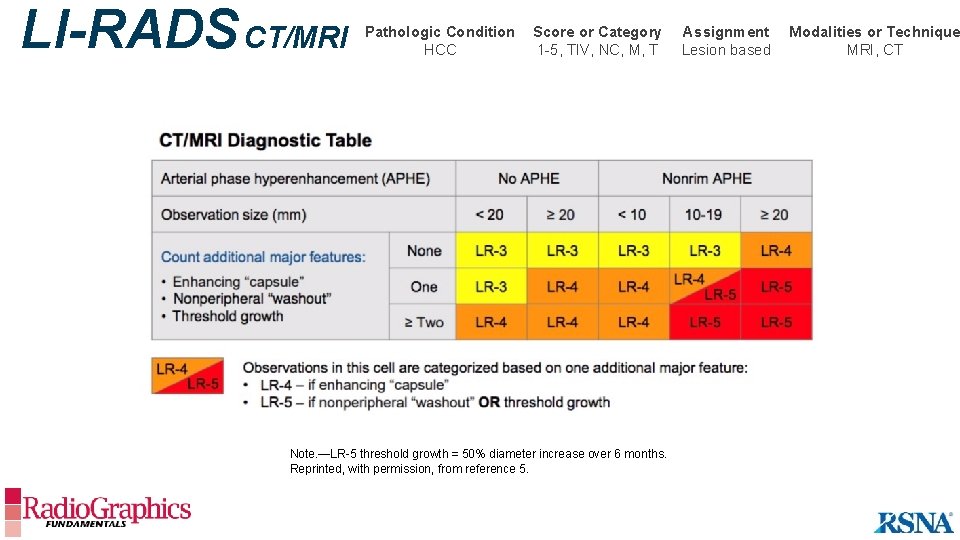
LI-RADS CT/MRI Pathologic Condition HCC Score or Category 1 -5, TIV, NC, M, T Note. —LR-5 threshold growth = 50% diameter increase over 6 months. Reprinted, with permission, from reference 5. Assignment Lesion based Modalities or Technique MRI, CT
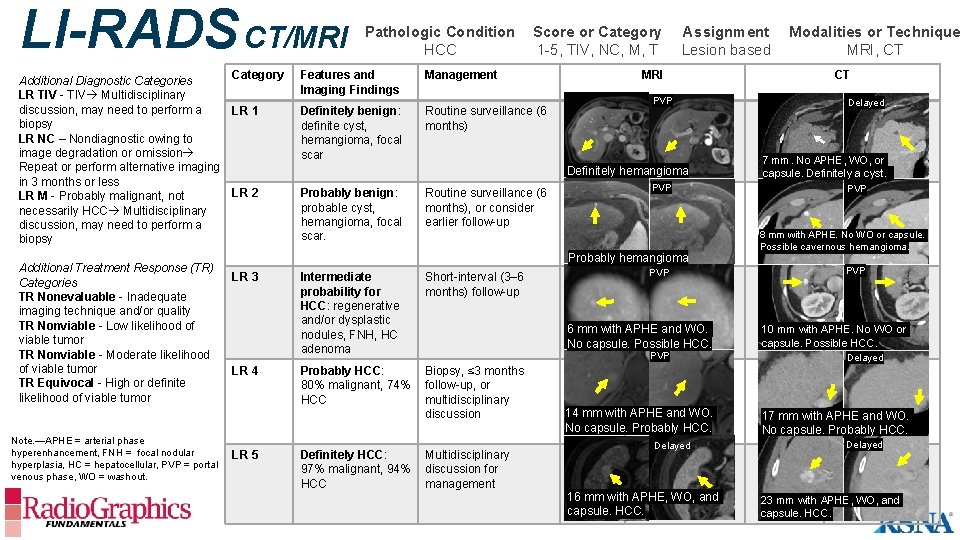
LI-RADS CT/MRI Additional Diagnostic Categories LR TIV - TIV Multidisciplinary discussion, may need to perform a biopsy LR NC – Nondiagnostic owing to image degradation or omission Repeat or perform alternative imaging in 3 months or less LR M - Probably malignant, not necessarily HCC Multidisciplinary discussion, may need to perform a biopsy Additional Treatment Response (TR) Categories TR Nonevaluable - Inadequate imaging technique and/or quality TR Nonviable - Low likelihood of viable tumor TR Nonviable - Moderate likelihood of viable tumor TR Equivocal - High or definite likelihood of viable tumor Note. —APHE = arterial phase hyperenhancement, FNH = focal nodular hyperplasia, HC = hepatocellular, PVP = portal venous phase, WO = washout. Pathologic Condition HCC Score or Category 1 -5, TIV, NC, M, T Category Features and Imaging Findings Management LR 1 Definitely benign: definite cyst, hemangioma, focal scar Routine surveillance (6 months) Assignment Lesion based MRI PVP Definitely hemangioma LR 2 Probably benign: probable cyst, hemangioma, focal scar. Routine surveillance (6 months), or consider earlier follow-up PVP Probably hemangioma LR 3 LR 4 LR 5 Intermediate probability for HCC: regenerative and/or dysplastic nodules, FNH, HC adenoma Short-interval (3– 6 months) follow-up Probably HCC: 80% malignant, 74% HCC Biopsy, ≤ 3 months follow-up, or multidisciplinary discussion Definitely HCC: 97% malignant, 94% HCC PVP 6 mm with APHE and WO. No capsule. Possible HCC. PVP Multidisciplinary discussion for management 14 mm with APHE and WO. No capsule. Probably HCC. Delayed PVP 16 mm with APHE, WO, and capsule. HCC. Modalities or Technique MRI, CT CT Delayed 7 mm. No APHE, WO, or capsule. Definitely a cyst. PVP 8 mm with APHE. No WO or capsule. Possible cavernous hemangioma. PVP 10 mm with APHE. No WO or capsule. Possible HCC. Delayed 17 mm with APHE and WO. No capsule. Probably HCC. Delayed 23 mm with APHE, WO, and capsule. HCC.
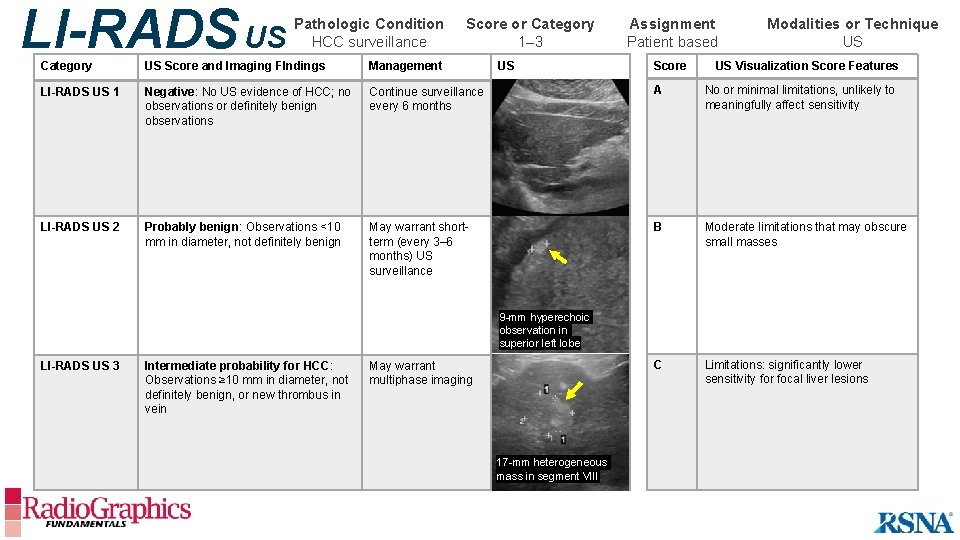
LI-RADS US Pathologic Condition HCC surveillance Score or Category 1– 3 US Assignment Patient based Score Modalities or Technique US US Visualization Score Features Category US Score and Imaging FIndings Management LI-RADS US 1 Negative: No US evidence of HCC; no observations or definitely benign observations Continue surveillance every 6 months A No or minimal limitations, unlikely to meaningfully affect sensitivity LI-RADS US 2 Probably benign: Observations <10 mm in diameter, not definitely benign May warrant shortterm (every 3– 6 months) US surveillance B Moderate limitations that may obscure small masses C Limitations: significantly lower sensitivity for focal liver lesions 9 -mm hyperechoic observation in superior left lobe LI-RADS US 3 Intermediate probability for HCC: Observations ≥ 10 mm in diameter, not definitely benign, or new thrombus in vein May warrant multiphase imaging 17 -mm heterogeneous mass in segment VIII
• Lung-RADS is used for screening examinations of adults ages 55– 80 (per the U. S. Preventive Services Task Force) with 30 pack -year (pkyr) smoking history and those who quit within 15 years. • Recommended annual repeat screening • Patient level score determined based on the nodule with the highest degree of suspicion 8 Lung-RADS Pathologic condition: Lung cancer (screening) Imaging modality: Low-dose CT Scoring: Patient score 0– 4 (4 A, B, X), S, C Version: 1 (2014)
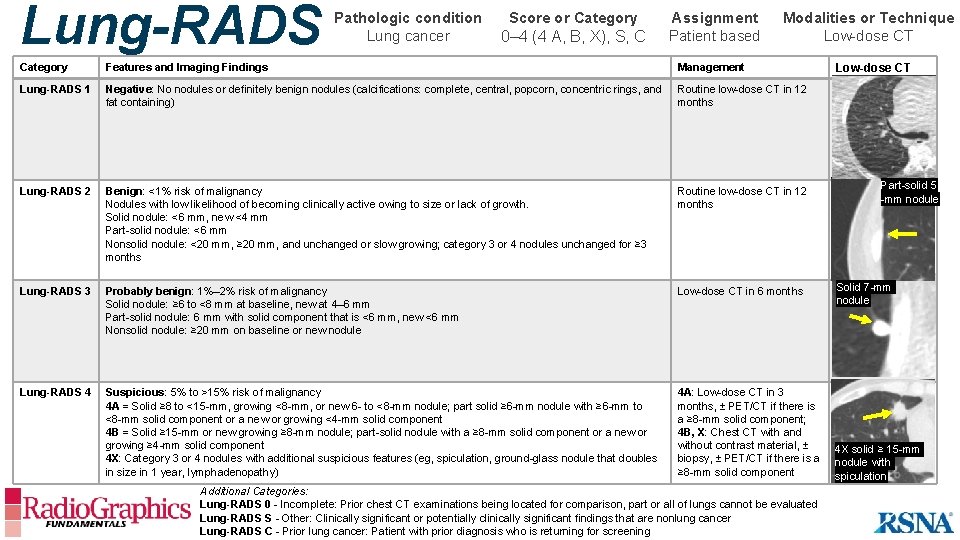
Lung-RADS Pathologic condition Lung cancer Score or Category 0– 4 (4 A, B, X), S, C Assignment Patient based Modalities or Technique Low-dose CT Category Features and Imaging Findings Management Lung-RADS 1 Negative: No nodules or definitely benign nodules (calcifications: complete, central, popcorn, concentric rings, and fat containing) Routine low-dose CT in 12 months Lung-RADS 2 Benign: <1% risk of malignancy Nodules with low likelihood of becoming clinically active owing to size or lack of growth. Solid nodule: <6 mm, new <4 mm Part-solid nodule: <6 mm Nonsolid nodule: <20 mm, ≥ 20 mm, and unchanged or slow growing; category 3 or 4 nodules unchanged for ≥ 3 months Routine low-dose CT in 12 months Lung-RADS 3 Probably benign: 1%– 2% risk of malignancy Solid nodule: ≥ 6 to <8 mm at baseline, new at 4– 6 mm Part-solid nodule: 6 mm with solid component that is <6 mm, new <6 mm Nonsolid nodule: ≥ 20 mm on baseline or new nodule Low-dose CT in 6 months Lung-RADS 4 Suspicious: 5% to >15% risk of malignancy 4 A = Solid ≥ 8 to <15 -mm, growing <8 -mm, or new 6 - to <8 -mm nodule; part solid ≥ 6 -mm nodule with ≥ 6 -mm to <8 -mm solid component or a new or growing <4 -mm solid component 4 B = Solid ≥ 15 -mm or new growing ≥ 8 -mm nodule; part-solid nodule with a ≥ 8 -mm solid component or a new or growing ≥ 4 -mm solid component 4 X: Category 3 or 4 nodules with additional suspicious features (eg, spiculation, ground-glass nodule that doubles in size in 1 year, lymphadenopathy) 4 A: Low-dose CT in 3 months, ± PET/CT if there is a ≥ 8 -mm solid component; 4 B, X: Chest CT with and without contrast material, ± biopsy, ± PET/CT if there is a ≥ 8 -mm solid component Additional Categories: Lung-RADS 0 - Incomplete: Prior chest CT examinations being located for comparison, part or all of lungs cannot be evaluated Lung-RADS S - Other: Clinically significant or potentially clinically significant findings that are nonlung cancer Lung-RADS C - Prior lung cancer: Patient with prior diagnosis who is returning for screening Low-dose CT Part-solid 5 -mm nodule Solid 7 -mm nodule 4 X solid ≥ 15 -mm nodule with spiculation
9 NI-RADS • NI-RADS is used for patients with a new neck mass and those with a known and/or treated mass undergoing surveillance • Provides recommendations regarding surveillance, direct inspection, short-term follow-up, additional imaging, and performing biopsy. Pathologic condition: Head and neck cancers (diagnostic and surveillance system) Imaging modalities: PET, CT, MRI Scoring: Patient score 0– 4 (2 A-B) Version: 1 (2016)
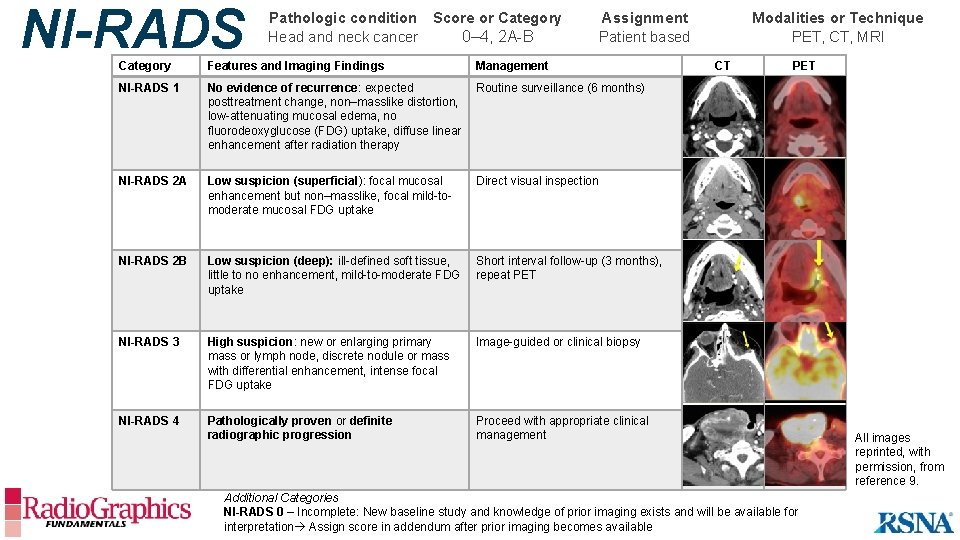
NI-RADS Pathologic condition Score or Category Head and neck cancer 0– 4, 2 A-B Assignment Patient based Category Features and Imaging Findings Management NI-RADS 1 No evidence of recurrence: expected posttreatment change, non–masslike distortion, low-attenuating mucosal edema, no fluorodeoxyglucose (FDG) uptake, diffuse linear enhancement after radiation therapy Routine surveillance (6 months) NI-RADS 2 A Low suspicion (superficial): focal mucosal enhancement but non–masslike, focal mild-tomoderate mucosal FDG uptake Direct visual inspection NI-RADS 2 B Low suspicion (deep): ill-defined soft tissue, little to no enhancement, mild-to-moderate FDG uptake Short interval follow-up (3 months), repeat PET NI-RADS 3 High suspicion: new or enlarging primary mass or lymph node, discrete nodule or mass with differential enhancement, intense focal FDG uptake Image-guided or clinical biopsy NI-RADS 4 Pathologically proven or definite radiographic progression Proceed with appropriate clinical management Modalities or Technique PET, CT, MRI CT PET Additional Categories NI-RADS 0 – Incomplete: New baseline study and knowledge of prior imaging exists and will be available for interpretation Assign score in addendum after prior imaging becomes available All images reprinted, with permission, from reference 9.
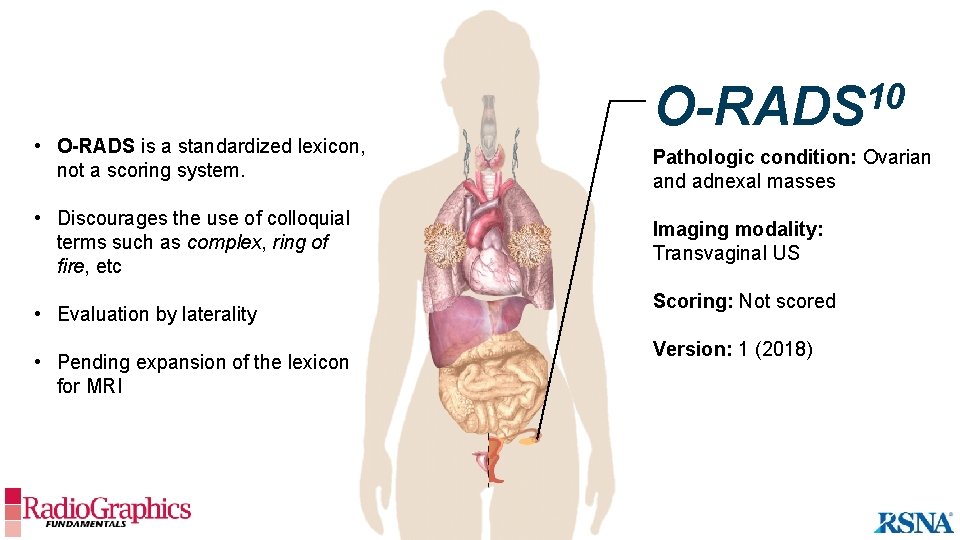
• O-RADS is a standardized lexicon, not a scoring system. • Discourages the use of colloquial terms such as complex, ring of fire, etc • Evaluation by laterality • Pending expansion of the lexicon for MRI 10 O-RADS Pathologic condition: Ovarian and adnexal masses Imaging modality: Transvaginal US Scoring: Not scored Version: 1 (2018)
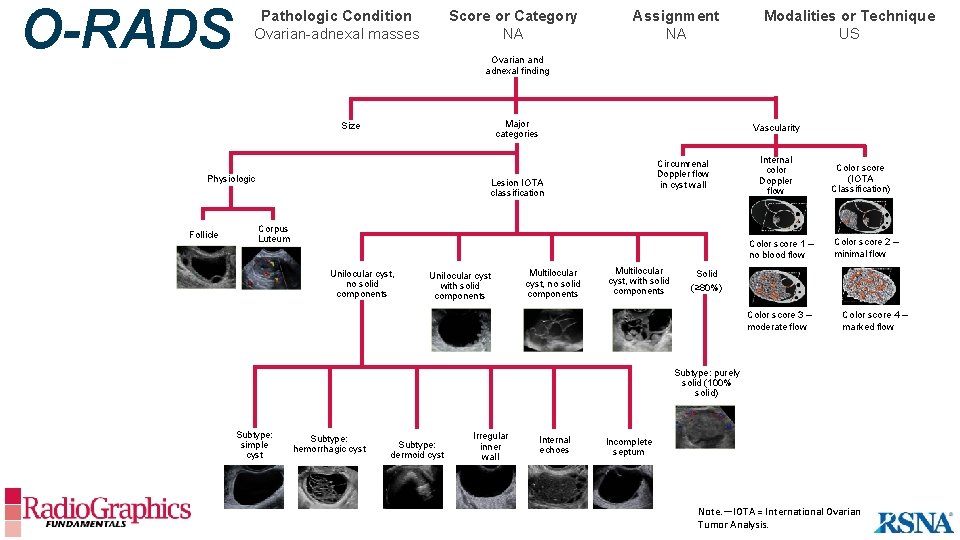
O-RADS Pathologic Condition Ovarian-adnexal masses Score or Category NA Modalities or Technique US Ovarian and adnexal finding Major categories Size Physiologic Follicle Assignment NA Vascularity Circumrenal Doppler flow in cyst wall Lesion IOTA classification Corpus Luteum Internal color Doppler flow Color score 1 – no blood flow Unilocular cyst, no solid components Unilocular cyst with solid components Multilocular cyst, no solid components Multilocular cyst, with solid components Color score (IOTA Classification) Color score 2 – minimal flow Solid (≥ 80%) Color score 3 – moderate flow Color score 4 – marked flow Subtype: purely solid (100% solid) Subtype: simple cyst Subtype: hemorrhagic cyst Subtype: dermoid cyst Irregular inner wall Internal echoes Incomplete septum Note. —IOTA = International Ovarian Tumor Analysis.
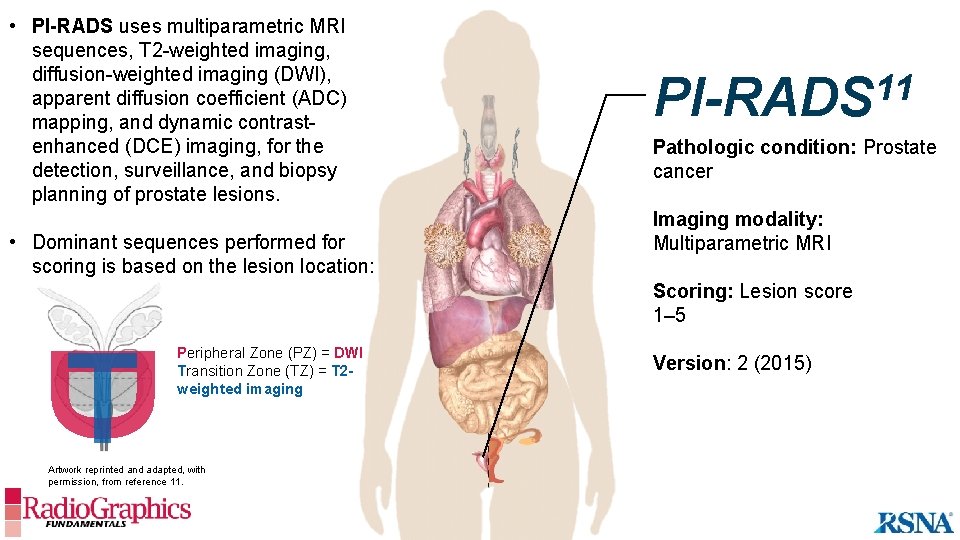
• PI-RADS uses multiparametric MRI sequences, T 2 -weighted imaging, diffusion-weighted imaging (DWI), apparent diffusion coefficient (ADC) mapping, and dynamic contrastenhanced (DCE) imaging, for the detection, surveillance, and biopsy planning of prostate lesions. • Dominant sequences performed for scoring is based on the lesion location: 11 PI-RADS Pathologic condition: Prostate cancer Imaging modality: Multiparametric MRI Scoring: Lesion score 1– 5 D T Peripheral Zone (PZ) = DWI Transition Zone (TZ) = T 2 weighted imaging Artwork reprinted and adapted, with permission, from reference 11. Version: 2 (2015)
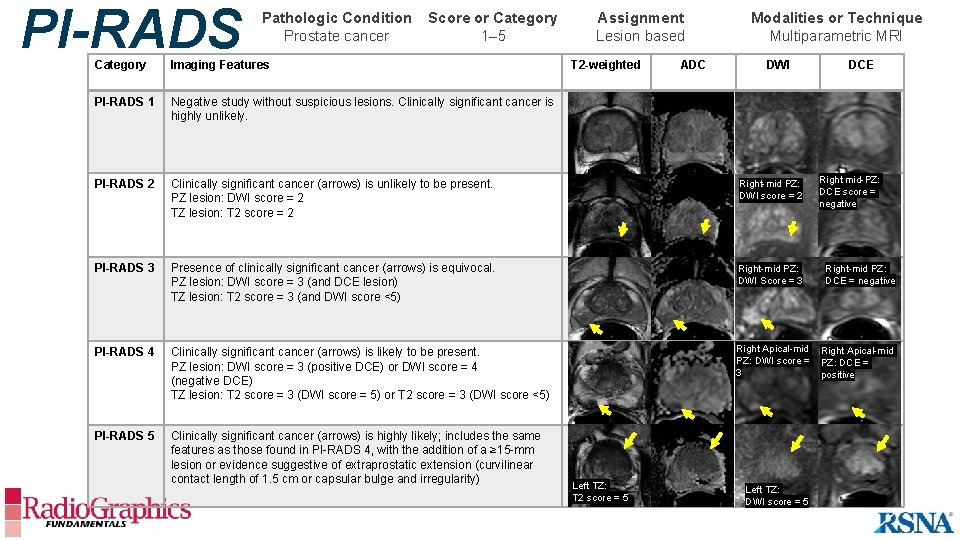
PI-RADS Pathologic Condition Prostate cancer Score or Category 1– 5 Assignment Lesion based T 2 -weighted Category Imaging Features PI-RADS 1 Negative study without suspicious lesions. Clinically significant cancer is highly unlikely. PI-RADS 2 Clinically significant cancer (arrows) is unlikely to be present. PZ lesion: DWI score = 2 TZ lesion: T 2 score = 2 Right-mid PZ: DWI score = 2 PI-RADS 3 Presence of clinically significant cancer (arrows) is equivocal. PZ lesion: DWI score = 3 (and DCE lesion) TZ lesion: T 2 score = 3 (and DWI score <5) Right-mid PZ: DWI Score = 3 PI-RADS 4 Clinically significant cancer (arrows) is likely to be present. PZ lesion: DWI score = 3 (positive DCE) or DWI score = 4 (negative DCE) TZ lesion: T 2 score = 3 (DWI score = 5) or T 2 score = 3 (DWI score <5) Right Apical-mid PZ: DWI score = 3 PI-RADS 5 Clinically significant cancer (arrows) is highly likely; includes the same features as those found in PI-RADS 4, with the addition of a ≥ 15 -mm lesion or evidence suggestive of extraprostatic extension (curvilinear contact length of 1. 5 cm or capsular bulge and irregularity) Left TZ: T 2 score = 5 ADC Modalities or Technique Multiparametric MRI DWI Left TZ: DWI score = 5 DCE Right mid-PZ: DCE score = negative Right-mid PZ: DCE = negative Right Apical-mid PZ: DCE = positive
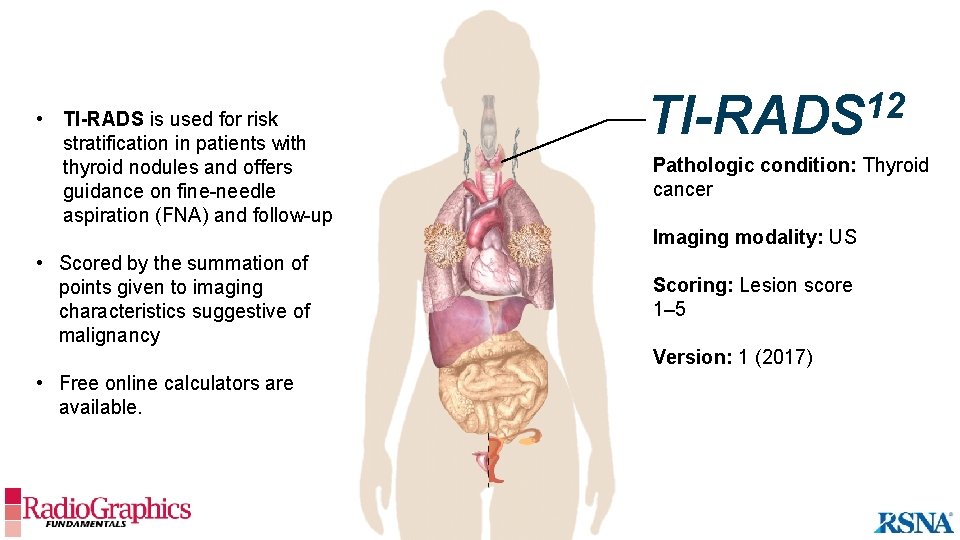
• TI-RADS is used for risk stratification in patients with thyroid nodules and offers guidance on fine-needle aspiration (FNA) and follow-up • Scored by the summation of points given to imaging characteristics suggestive of malignancy • Free online calculators are available. 12 TI-RADS Pathologic condition: Thyroid cancer Imaging modality: US Scoring: Lesion score 1– 5 Version: 1 (2017)
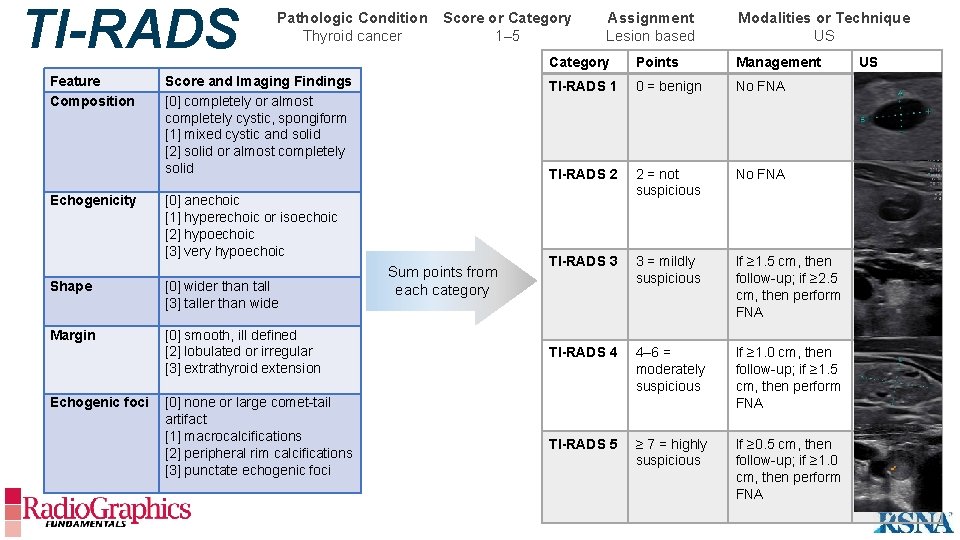
TI-RADS Feature Composition Echogenicity Pathologic Condition Score or Category Thyroid cancer 1– 5 Score and Imaging Findings [0] completely or almost completely cystic, spongiform [1] mixed cystic and solid [2] solid or almost completely solid [0] anechoic [1] hyperechoic or isoechoic [2] hypoechoic [3] very hypoechoic Shape [0] wider than tall [3] taller than wide Margin [0] smooth, ill defined [2] lobulated or irregular [3] extrathyroid extension Echogenic foci [0] none or large comet-tail artifact [1] macrocalcifications [2] peripheral rim calcifications [3] punctate echogenic foci Sum points from each category Assignment Lesion based Modalities or Technique US Category Points Management TI-RADS 1 0 = benign No FNA TI-RADS 2 2 = not suspicious No FNA TI-RADS 3 3 = mildly suspicious If ≥ 1. 5 cm, then follow-up; if ≥ 2. 5 cm, then perform FNA TI-RADS 4 4– 6 = moderately suspicious If ≥ 1. 0 cm, then follow-up; if ≥ 1. 5 cm, then perform FNA TI-RADS 5 ≥ 7 = highly suspicious If ≥ 0. 5 cm, then follow-up; if ≥ 1. 0 cm, then perform FNA US

Future Direction of 1 ______RADS

Current Limitations • Adoption varies from institution to May be improved by institution, as does experience among • Combining oncology RADS to five main radiologists types to avoid confusion. For example: • 1 = benign, 2 = likely benign, 3 = • Lack of consistency in terminology indeterminate, 4 = likely malignant, 5 and structure. For example: = malignant • lesion or observation • category, score, or level • Standardizing the terminology used for • category ranges: 0, 1– 4, 5, 6 nondiagnostic and negative studies • Nondiagnostic findings represented by 0, N, or NC • Limiting RADS to cancer imaging

Future Direction • ACR is currently changing the organization and approval process for RADS efforts in order to deal with growing demands. • Additional RADS are in varying stages of development. • The Myeloma Response Assessment and Diagnosis System (MY-RADS)13 (wholebody MRI for myeloma), Vesical Imaging-Reporting and Data System (VI-RADS)14 (MRI for bladder cancer), Metastasis Reporting and Data System for Prostate Cancer (MET-RADS)15 (whole-body MRI for metastatic prostate cancer) are recent non–ACR endorsed systems. • Incorporation of standardized templates and terminology with vendor-provided ACRassist • Integration with clinical society guidelines to increase adoption • For example, LI-RADS with the AASLD and BI-RADS with the American Society of Breast Surgeons guidelines
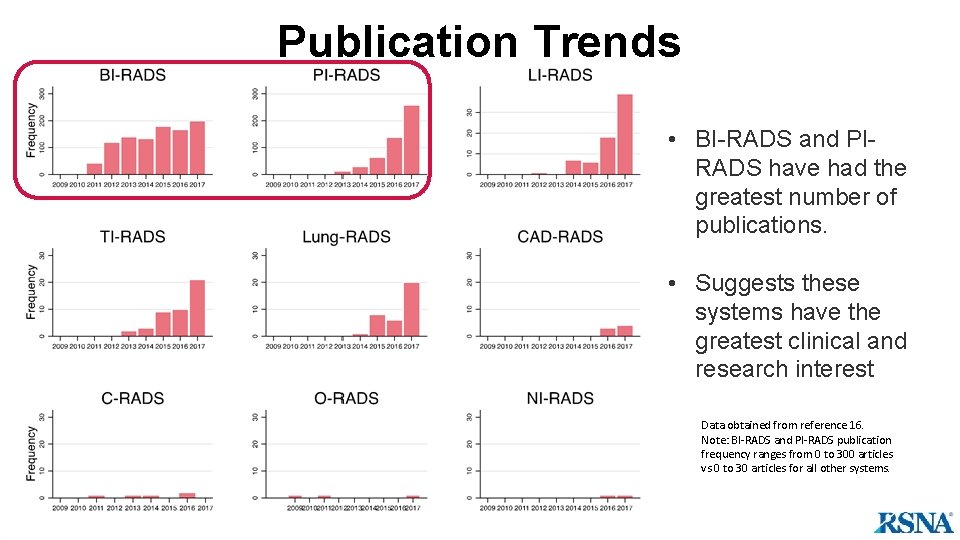
Publication Trends • BI-RADS and PIRADS have had the greatest number of publications. • Suggests these systems have the greatest clinical and research interest Data obtained from reference 16. Note: BI-RADS and PI-RADS publication frequency ranges from 0 to 300 articles vs 0 to 30 articles for all other systems.
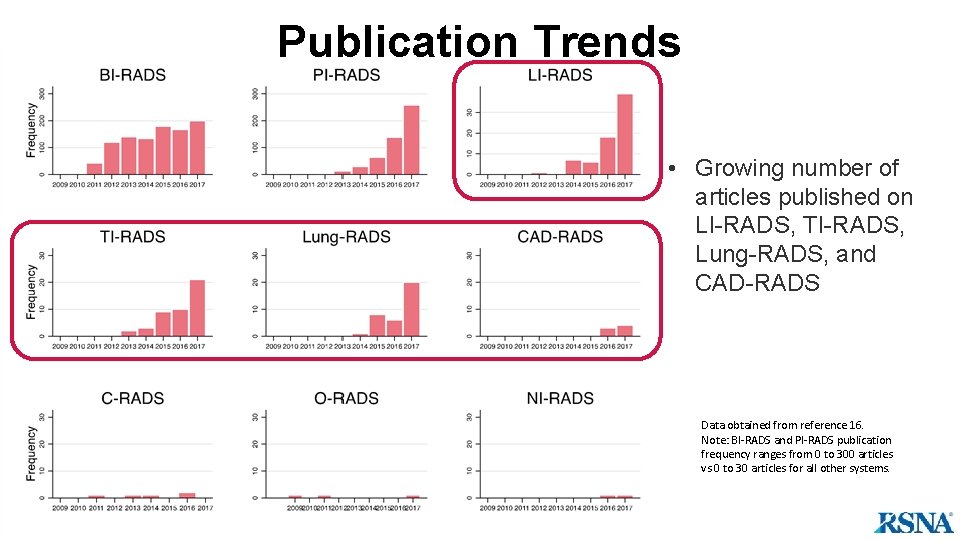
Publication Trends • Growing number of articles published on LI-RADS, TI-RADS, Lung-RADS, and CAD-RADS Data obtained from reference 16. Note: BI-RADS and PI-RADS publication frequency ranges from 0 to 300 articles vs 0 to 30 articles for all other systems.
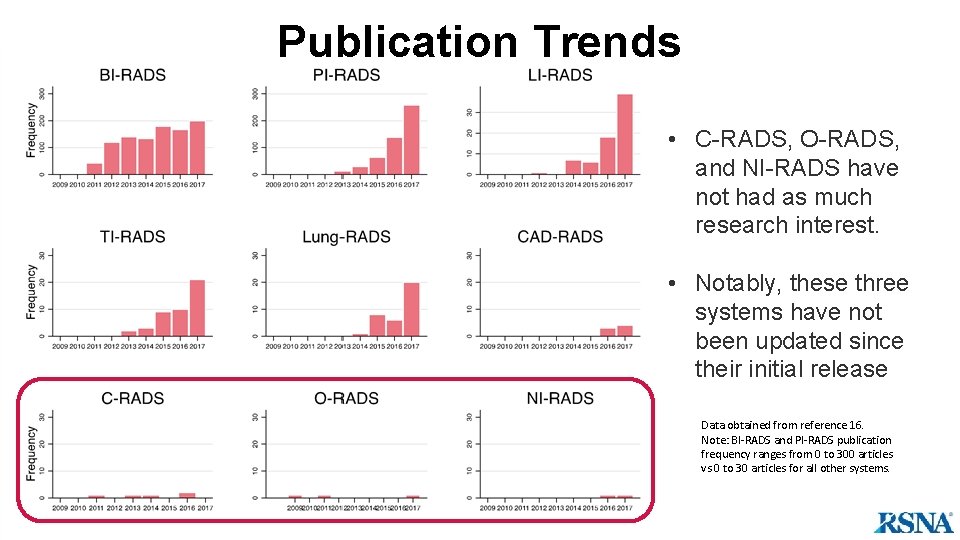
Publication Trends • C-RADS, O-RADS, and NI-RADS have not had as much research interest. • Notably, these three systems have not been updated since their initial release Data obtained from reference 16. Note: BI-RADS and PI-RADS publication frequency ranges from 0 to 300 articles vs 0 to 30 articles for all other systems.

Additional Resources for 1 ______RADS
______Website • Free, detailed, and up-todate resources for new systems and recent changes 1 • Ex: Head Injury Imaging Reporting and Data System (HI-RADS) is currently being developed for imaging traumatic brain injury Reprinted, with permission, from reference 1.

Video Resources TI-RADS Three-part ACR TI-RADS Webinar ACR TI-RADS Steering Committee https: //www. youtube. com/watch? v=s 0 f 1 c. U 7 rx. XA&t=113 s 17 https: //www. youtube. com/watch? v=Y 9 JU 2 i 4 IF-M&t=111 s 18 https: //www. youtube. com/watch? v=fs. WAle 6 u-u. E&t=333 s 19 Images reprinted, with permission, from references 17– 19. LI-RADS Introduction to LI-RADS, Claude Sirlin, MD https: //www. youtube. com/wat ch? v=Fml 7 w. H 5 ps 90&t=1 s 23 Image reprinted, with permission, from reference 23. ACR TI-RADS Series Jenny Hoang, MD https: //www. youtube. com/watch ? v=l. Aeyb-Qr. Xm 4&t=7 s 20 https: //www. youtube. com/watch ? v=P 7 GHt. G_9 Gv. Q 21 https: //www. youtube. com/watch ? v=w 4 o. ZYkw 8 wfw&t=2 s 22 Images reprinted, with permission, from references 20– 22. 2017 Version of LI-RADS for CT and MR Imaging: An Update Radio. Graphics https: //www. youtube. com/watch ? v=q-4 BEMvhr. Ng 24

Video Resources BI-RADS Breast Cancer Review Series: BI-RADS 5 th Edition Leonardo Valentin, MD https: //www. youtube. com/watch? v=AYZ cnh. Rwdxo&t=1 s 25 Image reprinted, with permission, from reference 25. PI-RADS Prostate MRI Using PIRADS Andrew Rosenkrantz, MD https: //www. youtube. com /watch? v=GJPQ 1 a 4 xj. As &t=25 s 26 Prostate MRI: Common Imaging Pitfalls Art Rastinehad, DO https: //www. youtube. com/ watch? v=7 L 0 VC 9 JY 2 ak 27 Image reprinted, with permission, from reference 26. Image reprinted, with permission, from reference 27.
Interactive Cases Lung-RADS Lung Cancer Screening e. Learning Program (ACR login required) https: //www. acr. org/Lifelong. Learning-and-CME/Learning. Activities/Lung-Cancer-Screening. Education 28 Image reprinted, with permission, from reference 28. BI-RADS LI-RADS Mammography Case Review (ACR MC 7 forthcoming) https: //www. acr. org/Lifelong. Learning-and-CME/Learning. Activities/Mammo-Case. Review 29 Image reprinted, with permission, from reference 29. LI-RADS teaching and case-based module, Montefiore/Albert Einstein, (ACR login required) https: //cortex. acr. org/Presenters/Case. Scri pt/Case. View? CDId=b. Sa. Qzu 9 Qe. R 0%3 d 30 PI-RADS Learn Prostate MRI Andrew Rosenkrantz, MD http: //learnprostatemri. com/interactivecases/31 Image reprinted, with permission, from reference 31. Image reprinted, with permission, from reference 30.

Summary • The ACR-endorsed RADS are a set of guidelines for the evaluation and interpretation of disease-oriented imaging studies. • Systems may be periodically updated to improve diagnostic parameters, and new systems are in various stages of development. • Clinical adoption of ACR RADS varies. • BI-RADS, followed by PI-RADS and LI-RADS, have the greatest acceptance. • Integration of RADS into multidisciplinary guidelines has the potential to increase the field of radiology’s impact on clinical care.

Suggested Readings Citation Ver Yr BI-RADS D’Orsi CJ, Sickles EA, Mendelson EB et al. ACR BI-RADS atlas, Breast Imaging Reporting and Data System. Reston, Va: American College of Radiology, 2013. v 5 2015 C-RADS Zalis ME, Barish MA, Choi JR et-al. CT colonography reporting and data system: a consensus proposal. Radiology. 2005; 236 (1): 3– 9. v 1 2005 CAD-RADS Cury RC, Abbara S, Achenbach S, et al. CAD-RADS Coronary Artery Disease: Reporting and Data System—An expert consensus document of the Society of Cardiovascular Computed Tomography (SCCT), the American College of Radiology (ACR) and the North American Society for Cardiovascular Imaging (NASCI): Endorsed by the American College of Cardiology J Cardiovasc Comput Tomogr. 2016; 10(4): 269– 281. v 1 2016 LI-RADS American College of Radiology. Liver Reporting and Data Systems (LI-RADS). https: //www. acr. org/Clinical-Resources/Reporting-and-Data -Systems/LI-RADS. Accessed June 4, 2019. v 3 2017/8 Lung-RADS Lung Imaging Reporting and Data System (Lung-RADS). Amer College of Radiology. CC BY-NC-ND 4. 0. v 1 2014 NI-RADS Cavazuti B, Hudgins P, Rath T, et al. Neck Imaging Reporting and Data System: An Atlas of NI-RADS Categories for Head and Neck Cancer. American College of Radiology. Accessed June 4, 2019. v 1 2014 O-RADS Andreotti RF, Timmerman D, Benacerraf BR. , et al. (2018). Ovarian-Adnexal Reporting Lexicon for Ultrasound: A White Paper of the ACR Ovarian-Adnexal Reporting and Data System Committee. Journal of the American College of Radiology. 15 (10), 1415– 1429. v 1 2018 PI-RADS Weinreb JC, Barentsz JO, Choyke PL, Cornud F, Haider MA, Macura KJ, Margolis D, Schnall MD, Shtern F, Tempany CM, et al. PI-RADS Prostate Imaging: Reporting and Data System: 2015, Version 2. Eur Urol. 2016; 69(1): 16– 40. v 2 2015 TI-RADS Tessler FN, Middleton WD, Grant EG, Hoang JK, Berland LL, et al. ACR Thyroid Imaging, Reporting and Data System (TI-RADS): White Paper of the ACR TI-RADS Committee. Journal of the American College of Radiology. 2017; 14 (5): 587– 595. v 1 2017

References 1. ACR. Reporting and Data Systems. https: //www. acr. org/Clinical-Resources/Reporting-and-Data-Systems. Accessed June 4, 2019. 2. D’Orsi CJ, Sickles EA, Mendelson EB et al. ACR BI-RADS atlas, Breast Imaging Reporting and Data System. Reston, Va: American College of Radiology, 2013. 3. Zalis ME, Barish MA, Choi JR et-al. CT colonography reporting and data system: a consensus proposal. Radiology. 2005; 236 (1): 3– 9. 4. Cury RC, Abbara S, Achenbach S, et al. CAD-RADS Coronary Artery Disease: Reporting and Data System—An expert consensus document of the Society of Cardiovascular Computed Tomography (SCCT), the American College of Radiology (ACR) and the North American Society for Cardiovascular Imaging (NASCI): Endorsed by the American College of Cardiology J Cardiovasc Comput Tomogr. 2016; 10(4): 269– 281. 5. American College of Radiology. CT/MRI LI-RADS v 2018. https: //www. acr. org/-/media/ACR/Files/RADS/LI-RADS 2018 -Core. pdf? la=en. Published July 2018. Accessed June 4, 2019. 6. American College of Radiology. Ultrasound LI-RADS v 2017. https: //www. acr. org/Clinical-Resources/Reporting-and-Data. Systems/LI-RADS/Ultrasound-LI-RADS-v 2017. Published 2017. Accessed June 4, 2019. 7. American College of Radiology. CEUS LI-RADS v 2017. https: //www. acr. org/Clinical-Resources/Reporting-and-Data. Systems/LI-RADS/CEUS-LI-RADS-v 2017. Published 2017. Accessed June 4, 2019. 8. American College of Radiology. Lung Imaging Reporting and Data System (Lung-RADS). https: //www. acr. org/Clinical. Resources/Reporting-and-Data-Systems/Lung-Rads. Accessed June 4, 2019.

References 9. Cavazuti B, Hudgins P, Rath T, et al. Neck Imaging Reporting and Data System: An Atlas of NI-RADS Categories for Head and Neck Cancer. American College of Radiology. Accessed June 4, 2019. 10. Andreotti RF, Timmerman D, Benacerraf BR. , et al. Ovarian-Adnexal Reporting Lexicon for Ultrasound: A White Paper of the ACR Ovarian-Adnexal Reporting and Data System Committee. J Am Coll Radiol. 2018; 15(10): 1415– 1429. 11. Weinreb JC, Barentsz JO, Choyke PL, Cornud F, Haider MA, Macura KJ, Margolis D, Schnall MD, Shtern F, Tempany CM, et al. PI-RADS Prostate Imaging: Reporting and Data System: 2015, Version 2. Eur Urol. 2016; 69(1): 16– 40. 12. Tessler FN, Middleton WD, Grant EG, Hoang JK, Berland LL, et al. ACR Thyroid Imaging, Reporting and Data System (TIRADS): White Paper of the ACR TI-RADS Committee. Journal of the American College of Radiology. 2017; 14(5): 587– 595. 13. Messiou C, Hillengass J, Delorme S, et al. Guidelines for Acquisition, Interpretation, and Reporting of Whole-Body MRI in Myeloma: Myeloma Response Assessment and Diagnosis System (MY-RADS). Radiology. 2019; 291(1): 5– 13. 14. Panebianco V, Narumi Y, Altun E et al. Multiparametric magnetic resonance imaging for bladder cancer: development of VI -RADS (Vesical Imaging-Reporting and Data System). Eur Urol 2018; 74(3): 294– 306. 15. Padhani AR, Lecouvet FE, Tunariu N. METastasis Reporting and Data System for Prostate Cancer: Practical Guidelines for Acquisition, Interpretation, and Reporting of Whole-body Magnetic Resonance Imaging-based Evaluations of Multiorgan Involvement in Advanced Prostate Cancer. Eur Urol 2017; 71(1): 81– 92. 16. National Institutes of Health: Office of Portfolio Analysis. i. Cite. https: //icite. od. nih. gov/analysis. Accessed June 4, 2019. 17. Radiology. ACR TI-RADS Webinar Part I: This Is How We Do It. You. Tube. https: //www. youtube. com/watch? v=s 0 f 1 c. U 7 rx. XA&t=113 s. Published March 8, 2018. Accessed June 4, 2019.

References 18. Radiology. ACR TI-RADS Webinar Part II: Case Based Review and Frequently Asked Questions. You. Tube. https: //www. youtube. com/watch? v=Y 9 JU 2 i 4 IF-M&t=111 s. Published April 9, 2018. Accessed June 4, 2019. Radiology. ACR. TI-RADS Thyroid Imaging Reporting and Data System Webinar Part III. You. Tube. https: //www. youtube. com/watch? v=fs. WAle 6 u-u. E&t=333 s. Published May 30, 2018. Accessed June 4, 2019. 20. Hoang J. ACR TI-RADS Overview and Approach. You. Tube. https: //www. youtube. com/watch? v=l. Aeyb-Qr. Xm 4&t=7 s. Published September 2, 2017. Accessed June 4, 2019. 21. Hoang J. ACR TI-RADS Structured reporting template. You. Tube. https: //www. youtube. com/watch? v=P 7 GHt. G_9 Gv. Q. Published September 3, 2017. Accessed June 4, 2019. 22. Hoang J. ACR TI-RADS DRAH Jan 9 2018. You. Tube. https: //www. youtube. com/watch? v=w 4 o. ZYkw 8 wfw&t=2 s. Published January 10, 2018. Accessed June 4, 2019. 23. UCSD Liver Imaging Group. Claude-athon 2018 (pt 7): Introduction to LI-RADS. You. Tube. https: //www. youtube. com/watch? v=Fml 7 w. H 5 ps 90&t=1 s. Published April 17, 2018. Accessed June 4, 2019. 24. Radio. Graphics. 2017 Version of LI-RADS for CT and MR Imaging: An Update. You. Tube. https: //www. youtube. com/watch? v=q-4 BEMvhr. Ng. Published November 3, 2018. Accessed June 4, 2019. 25. Valentin, L. Breast Cancer Review Series: BIRADS 5 th Edition. You. Tube. https: //www. youtube. com/watch? v=AYZcnh. Rwdxo&t=. Published October 23, 2015. Accessed June 4, 2019. 26. Rosenkrantz, A. Prostate MRI Using PI-RADS. You. Tube. https: //www. youtube. com/watch? v=GJPQ 1 a 4 xj. As&t=25 s. Published September 11, 2016. Accessed June 4, 2019.

References 27. Rastinehad, A. 8 Prostate MRI: Common Imaging Pitfalls. You. Tube. https: //www. youtube. com/watch? v=7 L 0 VC 9 JY 2 ak. Published March 1, 2016. Accessed June 5, 2019. 28. American College of Radiology. Lung Cancer Screening Education. https: //www. acr. org/Lifelong-Learning-and. CME/Learning-Activities/Lung-Cancer-Screening-Education. Accessed June 4, 2019. 29. American College of Radiology. Mammography Case Review. https: //www. acr. org/Lifelong-Learning-and-CME/Learning. Activities/Mammo-Case-Review. Accessed June 4, 2019. 30. American College of Radiology. LI-RADS teaching and case-based module. https: //cortex. acr. org/Presenters/Case. Script/Case. View? CDId=b. Sa. Qzu 9 Qe. R 0%3 d. Accessed June 4, 2019. 31. Rosenkrantz, A. Learn prostate MRI. Learnprostatemri. com http: //learnprostatemri. com/interactive-cases/. Accessed June 4, 2019.
- Slides: 54