Biowave PENS Percutaneous Electrical Nerve Stimulation System Presented

Biowave. PENS Percutaneous Electrical Nerve Stimulation System Presented by BIOMEDI, INC.

Introduction • The Biowave. PRO is an FDA, 510(k) cleared percutaneous electrical nerve stimulator (PENS) • PENS is a DIAGNOSTIC MODALITY used in the continuum of care process to determine next steps; confirm or rule out the potential therapeutic usefulness of a permanent electrical nerve stimulator for patients suffering from chronic pain • Biowave. PRO holds six US utility patents: Four on the signal technology and two on the electrodes • The surgical procedure requires physicians to utilize a surface implantable needle electrode to deliver electrical signals to deep tissue nerve fibers • Based on the recommended frequency of the procedures (2 x wk x 3 -4 wks), a physician should be able to rule out or confirm the appropriateness of an implantable pain management device within 30 days

Percutaneous Electrical Nerve Stimulation System • Biowave. PRO is the only FDA cleared PENS device on the market • Utilizes patented deep tissue signal technology • Signals are optimized for passing through skin into deep tissue • Utilizes patented percutaneous needle electrodes • Percutaneous needle electrode provides direct conductive pathway through skin into deep tissue • More comfortable than other forms of electrical stimulation • • • No programming Only control is intensity Active feedback control prevents patients from receiving a burn

Two Sets of Electrodes B-Set • Two round equal area percutaneous electrodes (2. 5 inch diameter) for treating two equal points of pain. Used for bilateral or unilateral pain in the lower back, buttocks, cervical spine, and shoulders, for radiculopathies, for pain centered directly over the spine, for pain throughout the knee, and for pain over large areas E-Set • One round percutaneous electrode (2. 5 inch diameter) for a primary site of pain and one rectangular noninvasive electrode (2 inches x 4 inches) for a secondary pain site, or to be placed over a bony prominence in the region being treated. Used for pain in the extremities including the knee, ankle, foot, neck, shoulder, elbow, wrist, hand finger

Implantable Percutaneous Needle Electrode • Two US utility patents on the Biowave electrode • The needle electrode bypasses the impedance and capacitance of skin • Allows for the delivery of electrical signals to deep tissue nerve fibers • Sterile, single-use only • 2. 5” diameter electrode contains 1014 microneedles, 0. 74 mm in length • Feels like velcro to the touch • Allows for 3”- 3. 5” diameter hemisphere treatment area

Patented Signal Technology • Four US utility patents on the signal technology • Biowave delivers two continuously summed high frequency alternating current electric signals (sine waves) • Contact with polarized tissue (C-Fiber, A Delta Fibers, and muscle tissue) causes the two high frequency sine waves to further multiply • This creates a new spectrum of electrical signals forming in approx. a 3”- 3. 5” diameter beneath and surrounding the percutaneous electrode • The multiplication of the two sine waves creates a LOW FREQUENCY SIGNAL in the form of an active electrical field

Mechanism of Action Neurogenic Pain • C-Fibers: The low frequency signal blocks the transmission of pain signals by preventing the sodium and potassium ion exchange across the membrane of the C-Fibers • A Delta Fibers: Numbness is induced in the area where the low frequency signal is present, which is approximately a 3”- 3. 5” diameter beneath and surrounding each electrode Myofascial Pain • Muscle Tissue: The low frequency signal causes the muscle tissue to be held in tension during the treatment, which creates a deep smooth pressure sensation • The low frequency signal also causes increased blood flow in the volume of tissue beneath and surrounding each percutaneous electrode

Biowave Clinical Studies • Randomized Controlled Study on the treatment of post-operative pain from TKA surgery - Hospital for Special Surgery, New York City • Podium Presentation at Eastern Orthopedic Association Annual Meeting, Oct 2008; Poster Presentation at AAOS Annual Meeting Feb 2009; Study submitted to the Journal of Arthroplasty • Randomized Blinded Controlled Study Treating OA Knee Patients Rush Univ. Medical Center - Published in ORTHOPEDICS June 2007 • Weill Cornell Medical College Chronic Low Back Pain Study (Phase 1 + 2) Abstract from Anesthesia & Analgesia, Mar 2003 • NY Giants White Paper - IRB approved study on treatment of Acute Sports Injuries on NY Giants football players June 2003 -Dec 2005, 600 treatments on 55 players • Clinical Study on treating extremities to reduce pain and improve range of motion - August 2005
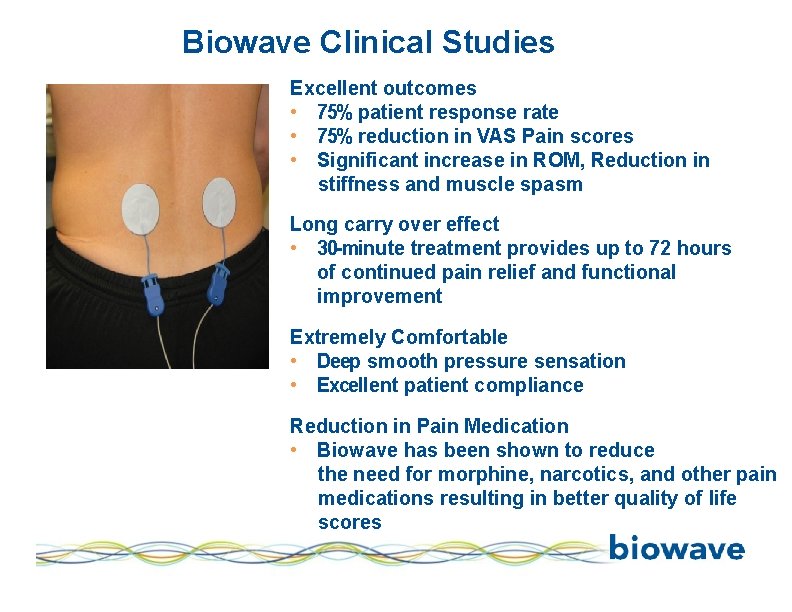
Biowave Clinical Studies Excellent outcomes • 75% patient response rate • 75% reduction in VAS Pain scores • Significant increase in ROM, Reduction in stiffness and muscle spasm Long carry over effect • 30 -minute treatment provides up to 72 hours of continued pain relief and functional improvement Extremely Comfortable • Deep smooth pressure sensation • Excellent patient compliance Reduction in Pain Medication • Biowave has been shown to reduce the need for morphine, narcotics, and other pain medications resulting in better quality of life scores
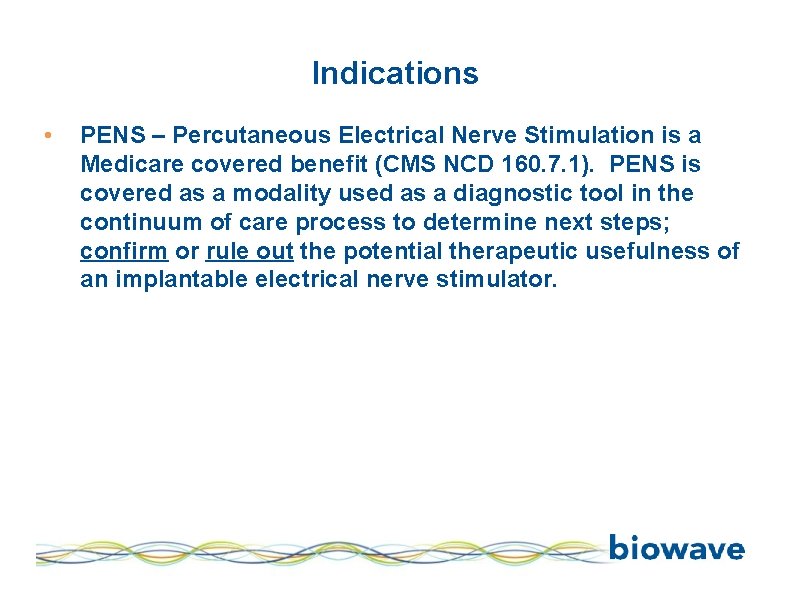
Indications • PENS – Percutaneous Electrical Nerve Stimulation is a Medicare covered benefit (CMS NCD 160. 7. 1). PENS is covered as a modality used as a diagnostic tool in the continuum of care process to determine next steps; confirm or rule out the potential therapeutic usefulness of an implantable electrical nerve stimulator.

Patient Selection / Medical Necessity • The patients whom you select for the procedure should present with conditions / diagnoses that are compatible from a continuum of care standpoint; the goal is to assess the patient’s suitability for therapeutic usefulness of ongoing treatment via a permanent stimulator • It is important that the physician’s documentation entails all prior conventional & conservative interventions. • TENS, P. T. , and medications (e. g. , NSAIDS, Narcotics, and Opioids) are all examples of interventions that would be expected to have been tried and failed prior to proceeding with the use of PENS for the treatment of chronic pain.

Sample Patient Type • Patient is a 55 y/o female who presents with lumbar post -laminectomy syndrome, and lumbar radiculitis, which has not responded favorably to conventional treatments. • Patient is currently taking the following medications: Percocet, Lyrica, Elavil, Voltaren gel, Lidoderm patches. • Previous interventions have included prednisone, physical therapy, TENS and nerve blocks, all of which have failed to control the patient’s pain resulting in an increase in functional declines and a decreased quality of life.

Recommended Treatment Protocol • 6 -8 treatments within 30 days (2 x wk x 3 -4 wks) • 30 minutes per treatment • After completing 6 -8 treatments the patient’s VAS score should be documented • In most cases a positive response is considered to be a reduction in the VAS score by 30% or more • If the patient’s VAS score represents a positive response, then the patient may be considered for a trial for a permanent electrical nerve stimulator • If the patient’s VAS score represents a negative response, then the patient should be reevaluated at appropriate intervals for further treatment options • Biowave training video for providers on Youtube

Contraindications Biowave is contraindicated if patients have the following: • • • Implanted Electrical Device Cardiac Pacemaker Defibrillator Metal Allergy Epilepsy Biowave electrodes may not be placed over the following areas: • • Open wound Heart Front or Side of Neck Top of the Head

“Incident to” Procedure • In your office, qualifying “incident to” services must be provided by a caregiver whom you directly supervise, and who represents a direct financial expense to you (such as a “W-2” or leased employee, or an independent contractor). • You do not have to be physically present in the treatment room while the service is being provided, but you must be present in the immediate office suite to render assistance if needed. • If you are a solo practitioner, you must directly supervise the care. • If you are in a group, any physician member of the group may be present in the office to supervise.

Commonly Used ICD 9 DESCRIPTION 722. 10 -722. 11 Displacement of thoracic or lumbar intervertebral disc without myelopathy 722. 51 – 722. 52 Degeneration of thoracic or lumbar intervertebral disc 722. 72 Intervertebral thoracic disorder with myelopathy, thoracic region 722. 73 Intervertebral disc disorder with myelopathy, lumbar region 722. 83 Post-laminectomy syndrome, lumbar region 723. 4 Brachial neuritis or radiculitis NOS 724. 2 Lumbago 724. 3 Sciatica 724. 4 Thoracic or lumbosacral neuritis or radiculitis, unspecified 729. 2 Neuralagia, neuritis, and radiculitis, unspecified neuropathic pain
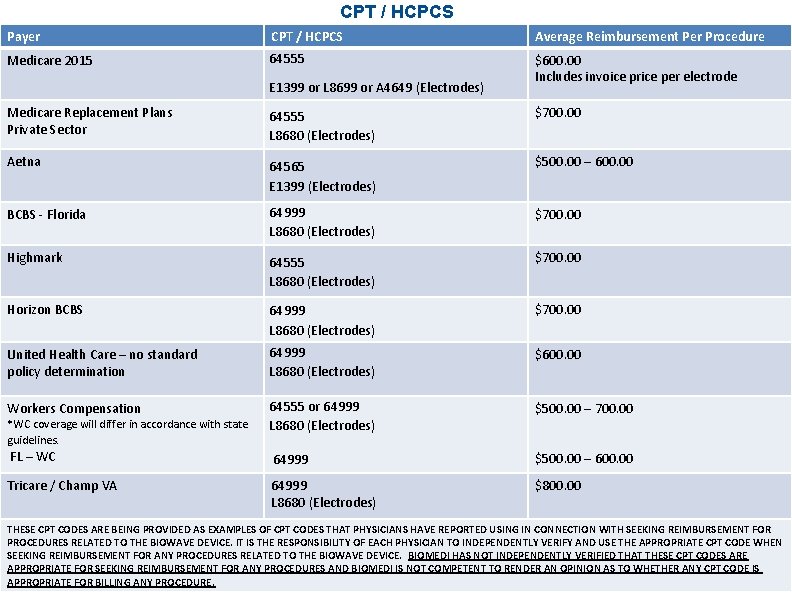
CPT / HCPCS Payer CPT / HCPCS Average Reimbursement Per Procedure Medicare 2015 64555 $600. 00 Includes invoice price per electrode E 1399 or L 8699 or A 4649 (Electrodes) Medicare Replacement Plans Private Sector 64555 L 8680 (Electrodes) $700. 00 Aetna 64565 E 1399 (Electrodes) $500. 00 – 600. 00 BCBS - Florida 64999 L 8680 (Electrodes) $700. 00 Highmark 64555 L 8680 (Electrodes) $700. 00 Horizon BCBS 64999 L 8680 (Electrodes) $700. 00 United Health Care – no standard policy determination 64999 L 8680 (Electrodes) $600. 00 Workers Compensation 64555 or 64999 L 8680 (Electrodes) $500. 00 – 700. 00 FL – WC 64999 $500. 00 – 600. 00 Tricare / Champ VA 64999 L 8680 (Electrodes) $800. 00 *WC coverage will differ in accordance with state guidelines. THESE CPT CODES ARE BEING PROVIDED AS EXAMPLES OF CPT CODES THAT PHYSICIANS HAVE REPORTED USING IN CONNECTION WITH SEEKING REIMBURSEMENT FOR PROCEDURES RELATED TO THE BIOWAVE DEVICE. IT IS THE RESPONSIBILITY OF EACH PHYSICIAN TO INDEPENDENTLY VERIFY AND USE THE APPROPRIATE CPT CODE WHEN SEEKING REIMBURSEMENT FOR ANY PROCEDURES RELATED TO THE BIOWAVE DEVICE. BIOMEDI HAS NOT INDEPENDENTLY VERIFIED THAT THESE CPT CODES ARE APPROPRIATE FOR SEEKING REIMBURSEMENT FOR ANY PROCEDURES AND BIOMEDI IS NOT COMPETENT TO RENDER AN OPINION AS TO WHETHER ANY CPT CODE IS APPROPRIATE FOR BILLING ANY PROCEDURE.

Direct Cost to Provider • Biowave. PRO System: $3, 500. 00 • One year manufacturer warranty on the Biowave. PRO (excludes attachment cables) • Electrodes: 6 sets per pack @ $1, 200 per pack • Capital investment options available through a third party • Priority Capital Memo provided

Summary • Biowave. PRO is an FDA cleared, patented PENS device • Proven efficacy through clinical trials • Excellent diagnostic procedure to support your existing pain management treatment regimen • Reimbursed by Medicare, most Medicare Replacement Products, and Commercial Carriers • Expert billing support services available • Capital investment options available

Biowave Set-up • Device needs to be charged before use • Charger is shipped with each device • Full charge last 8 -9 hours, office should charge overnight for best results • Electrode wire is pushed in and turned to lock in tightly • Unit timer is automatically set to 30 minutes • Staff can adjust timer by pressing timer button to the left of +/- buttons

Sales Call What To Bring • • • This Powerpoint presentation Provider Inserts, i. e. , (CMS - NCD, Medicare Advantage PENS Policy, Procedure Note, LMN) Rep Docs, i. e. , (Order Form) Sales Call Flow • • What it is How it works Stress Compliance (Patient Selection, Medical Necessity, Documentation) ASK for sale and complete Order Form Best Practices • • • Biowave is a DIAGNOSTIC modality used in the treatment of neurogenic pain Drs can use a credit card to purchase Biowave and equipment and get first reimbursements in 30 -60 days RIGHT patient, RIGHT documentation, RIGHT codes = COMPLIANCE
- Slides: 21