BIOTON S A www bioton pl BIOTON S

BIOTON S. A www. bioton. pl

BIOTON S. A Company Mission � BIOTON aims to reinforce its position of a biotechnological company with a global reach. The Company is able to pursue its strategic objectives of protecting Patients’ health and improving the quality of their lives by participating in research and development projects, promoting the expansion of its product portfolio and making well-informed business decisions. BIOTON fulfils its stated mission by implementing strategies and development plans aimed at: � reinforcing BIOTON's status on the Polish insulin market � promoting BIOTON's insulin world-wide by maximising its share of the existing markets and entering new ones � developing innovative, clinically important medicinal products, including biotechnological drugs developed by members of the BIOTON Group � expanding BIOTON’s generic drug portfolio.

BIOTON S. A Jubo Liu – President of the Management Board Mr. Jubo Liu is the founder and has served as the CEO of Cogenes Biotechnology Incorporation (focused on in vitro diagnosis R&D, manufacturing and marketing) since August 2011 and is the founder and has served as the CEO of Novo. Tek Pharmaceuticals Limited (Hong Kong) and Beijing Novo. Tek Medicinal Technology Development Co. Ltd (Beijing) (international platform to globalise healthcare products both from within and outside of China) since August 2009. From April 2008 to July 2009 he was the China Chief Representative of Immtech Pharmaceuticals Inc. (US New York) (establishing Chinese activity infrastructure and investment plans). From November 2007 to April 2008, he acted as a Clinical Trial Liaison with US-based Pharmaceuticals Inc. (US New York) (carrying out global clinical trial operation in China). From October 2006 to November 2007 he was a scientist at Boston Vertex Pharmaceuticals Inc. (formulation and drug delivery). From July of 2005 to January of 2007 he was as a cofounder and served as the Director of Business Development of JCS Biopolytech Incorporation (modified biopolymer R&D and marketing used for drug delivery and nanotechnology). From August 1997 to February 2001, he served as the Director of Marketing and Regulatory Affairs for Huayuanlong Medicinal Technology Development Co. Ltd. of Beijing, China. Mr. Liu obtained his Bachelor’s Degree in Pharmacy from the Department of Pharmacy of the Pharmaceutical University of Shenyang (September 1992 to July 1997) and a Ph. D. from the Department of Pharmaceutical Sciences at the University of Toronto (from
BIOTON S. A Products: � Recombinant human insulin in pharmaceutical substance form and as preparations for injections; � Oral hypoglycemic drug - glimepiride � Oral hypoglycemic drug – metformin � „BIOTON FOR HEALTH” is a new OTC products line of the BIOTON company. � The products line focuses on diet supplements, products for particular nutritional uses and cosmetics dedicated to patients with certain health problems (metabolic syndrome, diabetes) helping them to keep their organisms in optimal condition. � The products from „BIOTON FOR HEALTH” line contain natural ingredients of the highest quality, carefully balanced in composition.

BIOTON S. A Members of the BIOTON Group: Sci. Gen Ltd � Biotechnology company Sci. Gen was established in Singapore in 1988 and has been listed on the Australian Stock Exchange (ASX code SIE) since 2002. Sci. Gen sells recombinant human insulin under the trade name Sci. Lin in Southeast Asia, Australia and Oceania (excluding Japan) under licence from Savient Pharmaceuticals Inc. (formerly known as Bio-Technology General Corp. ). The company manufactures and sells licensed biotechnological products, including a recombinant human growth hormone under the trade name Sci. Tropin and a hepatitis B vaccine under the trade name Sci-B-Vac. � Sci. Gen has offices in Australia, China, Hong Kong, India, Singapore, South Korea, Philippines, USA and Vietnam. � BIOTON S. A. holds 90. 54% of the share capital of Sci. Gen. Subsidiary companies of Sci. Gen Ltd: � Sci. Gen India Pvt Ltd – Sci. Gen acquired 100% shares of an Indian company in 2006. The new facility in Pune, India manufactures recombinant human insulin and other biotechnology products in accordance with European and American standards. � Sci. Gen Australia Pty Ltd – a company established by Sci. Gen in Sydney for the wholesale of biotechnological products. � Sci. Gen Korea Ltd – a company established by Sci. Gen to sell biotechnological products.

BIOTON S. A Bio. Partners Holdings AG A Swiss based company established in 2000 which mainly specialises in developing innovative biotechnological pharmaceuticals using recombinant DNA technologies. The company was the second to be granted marketing authorisation for a biosimilar drug by the EMEA for the human growth hormone Valtropin. The distribution rights for this product cover all of Europe, including the Commonwealth of Independent States, Japan, Australia and some Asian countries. Other products include: � A sustained release human growth hormone, this product is a breakthrough in growth hormone therapy because it allows weekly instead of daily injections. The distribution rights of Valtropin SR cover all of Europe, including the Commonwealth of Independent States, Japan, Australia and some Asian countries. � Interferon Beta, for the treatment of multiple sclerosis is sold under the trade name Biferonex with worldwide distribution rights. � Ribavirin, for the treatment of chronic hepatitis C in combination with interferon alpha is sold under the trade name Ravanex with worldwide distribution rights. � BIOTON S. A. purchased 100% of the shares in Bio. Partners in March 2007.

BIOTON S. A R&D � The BIOTON Group is oriented towards innovation and development and actively seeks out new opportunities by promoting research and development projects. BIOTON Group’s extensive experience, its highly qualified team of specialists and its long-term investment strategy, enable the company to move into new areas of activity which offer the greatest long-term growth potential and which best contribute to protecting Patients' health and improving their quality of life. � The Institute of Biotechnology and Antibiotics (IBA) has been BIOTON S. A. 's main research division ever since the Company commenced operations. IBA is Poland’s leading research institute and employs highly qualified and experienced specialists. IBA has been researching drug production by chemical synthesis and biosynthesis using genetically modified organisms for over 50 years. BIOTON and IBA conduct research in biotechnological products and new forms of pharmaceutical products and carry out research programs in the following areas: � Genetic engineering: these programs focus on the search for new plasmids in wild-type bacterial strains, plasmid isolation, creation of sequence regions for gene cloning and building new expression vectors based on the isolated plasmids. The aim is to develop a methodology for creating effective, stable, genetically modified microorganisms which can be used in the industrial production of therapeutic protein substances. � Biotechnology: this program investigates the isolation and modification of biological proteins so as to select the optimal methodology for processing recombinant proteins. � Pharmacology: research is conducted into the resistance mechanisms of bacterial strains to therapeutic antibiotics, analyses of synthesis methods and the properties of new derivatives of anthracycline antibiotics, and the pharmacokinetics and pharmacodynamics of medicinal substances for the purpose of developing new medicinal products and new research methods to assess them.

BIOTON S. A R&D � Bio. Partners – strategic partnerships, close collaboration with scientific experts and access � � to state-of-the-art manufacturing facilities make it possible to develop innovative products. Biopartners’ R&D partners include: LG Life Science - a South-Korean biotechnology company with a state-of-the-art R&D facility that has been engaged in biopharmaceuticals development since 1982 Rentschler Biotechnologie - a German company which specialises in the development and manufacture of biopharmaceuticals, including interferons, and contract manufacture of drugs in various pharmaceutical forms. Fisiopharma S. R. L. – Fisiopharma works with Italian universities and other pharmaceutical companies on research and development projects designed to stimulate innovation and improve the manufacturing technologies of active pharmaceutical ingredients and finished forms of generic pharmaceuticals. Medipolis GMP-Oulu – Medipolis GMP is a company specialising in Good Manufacturing Practice production for microbiogical fermentation and in producing and developing products for biotechnological companies. The company focuses on the small-scale manufacture of active pharmaceutical ingredients (API) for clinical and toxicological purposes (these include therapeutic proteins, enzymes, therapeutic injections and DNA plasmids). The company meets FDA and EMEA requirements for biopharmaceutical production and is the ideal production plant for manufacturing biotechnological products for clinical research required as part of EMEA and FDA registration processes.

BIOTON S. A According to the information received by BIOTON S. A. (“Company”) from the shareholders, the ownership structure of the share capital of the Company, as at 02 December 2016: 1. Uni. Apek S. A. 16 989 289 19, 79% 2. Perfect Trend Ventures Ltd. 8 586 419 9, 99% 3. Troqueera Enterprises Ltd 8 480 570 9, 88% 4. Basolma Holding Ltd 6 151 852 7, 16% 5. AIS Investment 2 Sp. z o. o. 5 151 852 6, 00% Others: 40 504 218 47, 18% Total 85 864 200 100, 00%

BIOTON S. A In the result of the redefining its business strategy, in 2016, the Company made a number of decisions concerning its further engagement in some development projects, and capital engagement in subsidiaries. Their main premise was the limitation of financial risk, reduction of operating costs of the Company, focusing the existing resources on key competence and acceleration of the financial effect of commercialization of the already owned assets on selected markets. In 2016, the Company continued operations implemented in the previous years and related to the increase of Company and Group products sales, and maintaining operating costs on sustainable level. These operations focused on the following key areas: � consolidation of products portfolio � commercialization of the key product of the Company - recombinant human insulin - and focusing the research and development activity on the products with the highest market potential, mainly including long and short acting human insulin analogues. � reduction of operating costs, and adjustment of the existing infrastructure to the new requirements of execution of the strategy on selected markets, � focusing the strategic operations of the Group on the specified key competences, i. e. : � manufacturing high quality biotechnological products, � research and development of new biotechnological products, � actions within the area of registration of biotechnological products in the key global markets for the purpose of their further commercialization in cooperation with the leading pharmaceutical concerns operating in the global market,
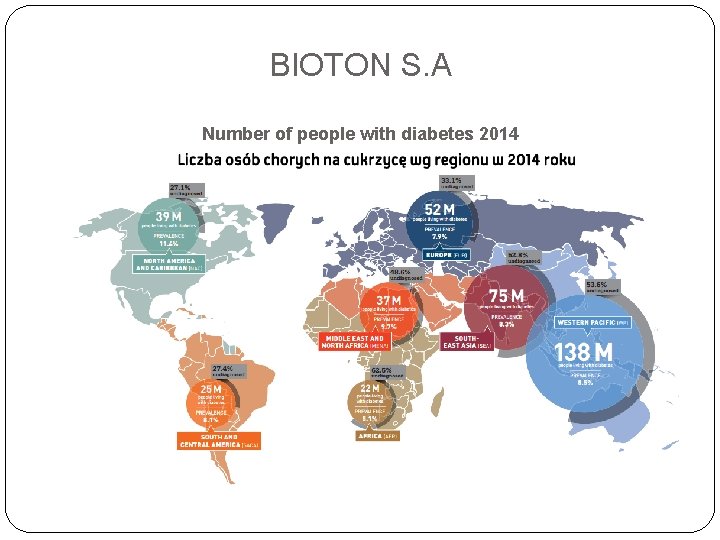
BIOTON S. A Number of people with diabetes 2014

BIOTON S. A According to Novo Nordisk, the global market value of antidiabetic drugs currently amounts to 38 billion Euro . Its average annual growth rate in the last decade was about 12 percent, and the market value is also expected to grow rapidly over the next few years - including a larger population of people with diabetes, improved awareness, and the need for more effective methods. Treat and market more advanced products. The largest share of the global diabetes market - 55 percent. - it is insulin. Oral drugs account for 38 percent, and incremental GLP-1 drugs represent 7 percent market value. P&S Market Research estimates that the global insulin market, estimated at $ 23. 9 billion Usd by the company, is set to reach $ 48. 5 billion by 2020 (average annual growth of 12. 5 percent). In Europe, the average annual growth rate for 2015 -2020 is 12. 6%, North America 11. 8%, and Asia. Pacific 13. 9%. Major players in this market are Sanofi, Novo Nordisk, Eli Lilly and Company, Glaxo. Smith. Kline, Pfizer, Merck & Co. and Julphar. According to the International Diabetes Federation (IDF), about 387 million people in the world suffer from diabetes (8. 3% of the population aged 20 to 79) in 2014 and by 2035 this number to increase to 592 million people. In nearly half of the patients (46. 3%), diabetes was not diagnosed. This is largely due to its low detection in the poorest countries, but even in the richer parts of the globe, such as in Europe or North America, at 33. 1%, respectively. And 27. 1% Diabetes is not diagnosed. In Poland, the number of people with diabetes is estimated at over 2 million people (7. 1 percent of the population aged 20 to 79), of which 34 percent. People with the disease remain undetected. In China, where Bioton seeks to increase sales of its products, more than 96 million people suffer from diabetes (9. 3 percent of the population aged 20 to 79), which is the largest in the world and 54 percent. People in this country have not been diagnosed at all. As estimated by the IDF, global diabetes costs in 2014 amounted to $ 612 billion (about 12 percent of all health costs).

BIOTON S. A At the end of June 2015, the fixed assets of the Bioton Capital Group were worth PLN 1. 67 billion and accounted for 83% total amount of assets. Among them, the most significant item was „other intangible assets”, amounting to PLN 1. 13 billion, mainly consisting of concessions, patents, licenses and computer software, development works and other intangible assets (the latter largely Resulting from the acquisition of BIOLEK sp. z oo). It must be said that there is a rather high risk that the Issuer will not achieve its intended development results, particularly in the field of biotechnology products. As a consequence, significant development costs that have been activated by the Bioton Group (shown as intangible assets in the balance sheet and amortized and not as an expense in the profit and loss account at the time they are incurred) would not be recovered (through sales of the developed products) as could have a significant negative impact on the business, financial situation and results of the Issuer. In 2015 the value of intangible assets was revised to the actual value, with a write-off of 739 million PLN.

BIOTON S. A Chinese market the world's largest number of people suffering from diabetes (although the average cost per patient is relatively low compared to North America, for example). Secondly, the government and consumers are more likely to enjoy less expensive alternatives to the more expensive medicines that the Bioton Group may benefit from. Thirdly, in recent months, the involvement of the Company's shareholders has been enhanced by Chinese investors who can assist Bioton with their contacts, distribution networks and experience in the home market and other Asian countries. So far the Issuer's sales on that market remains low - in the first half of 2015 it amounted to ca. PLN 7. 9 million (against PLN 5. 5 million a year earlier).

BIOTON S. A Russian market The Biotec Group, an integrated pharmaceutical holding company with annual turnover of $ 1. 2 billion, is responsible for the distribution and promotion of Company products (insulinready forms and insulin delivery pen). More than 300 pharmacies, a distribution company with nearly 30 local distribution centers and 4 manufacturing companies producing over 400 pharmaceuticals. While the Issuer's Management Board is counting on improving sales growth in Russia, sales to the Russian market are still relatively low - in the first six months of this year revenues amounted to PLN 7. 7 m in 2015 (PLN 13. 9 m in 2014).

BIOTON S. A The acquisition of Biolek continuously since 2011 is described by the Management Board of the Issuer as "a means to realize the strategic goal of the Company's creation of a second business line, which in the short term will positively impact the Company's financial results and provide additional cash to accelerate further development. Generations of insulin, including analogue insulin. " However, as the years go by, virtually no positive effects have been seen on the acquisition of veterinary shares of Biolek. It is no wonder that the shareholders of Bioton have long since ceased to believe that this could change in the future. In the first six months of this year, revenues of Biolek sp. z o. o. Amounted to PLN 14 thousand (compared with PLN 307 thousand of revenues a year earlier). For comparison: as part of settlements related to the acquisition of Biolek, Bioton issued shares for a total nominal value exceeding PLN 0. 6 billion in the years 2011 -2013 to Troqueera Enterprises Limited.

BIOTON S. A Little is known about slow release growth hormone, which was notified by the European Commission to the European Union, Norway and Iceland in August 2013. One of the few pieces of information that can be found in Bioton's latest reports is that the Company is negotiating a long-standing growth hormone sales agreement with a global pharmaceutical company. The Bio. Partners Group, which is responsible for the development of this product and which is owned by Bioton, did not achieve any sales revenue in the first half of 2015 (as in the previous year).

BIOTON S. A The main source of financing the business of the Bioton Capital Group is its equity, which at the end of the first half of the year was worth PLN 1. 5 billion, which accounted for 73%. Total amount of liabilities. At the same time, the Issuer's share capital amounts to PLN 1. 7 billion, of which approximately PLN 0. 6 billion comes from the 2011 -2013 series of share issues for Troqueera Enterprises Limited. However, it is worth noting, though as a result of the aforementioned share issue the Issuer's own capital increased significantly, the Bioton Group did not obtain any cash from them - they were connected with the settlement of the Biolek acquisition.
- Slides: 18