Biotin Structure consists of two rings and a

Biotin • Structure consists of two rings and a valeric acid side chain • Sources – liver, soybeans, egg yolk, cereals, legumes, nuts – often found bound to protein • biocytin o or biotinyllysine – avidin binds biotin and inhibits absorption • glycoprotein found in egg whites • heat labile so cooking denatures

Biotin • Digestion and Absorption – Protein bound biotin requires digestion by proteolytic enzymes • yields free biotin or biocytin • Biotinidase hydrolyzes biocytin to free biotin and lysine • Undigested biocytin may be absorbed and digested in plasma by biotinidase • Unmetabolized biocytin is excreted in urine

Biotin • Biotinidase – Deficiency due to inborn error has been documented in infants and children – Clinical features include seizures, ataxia, skin rash, alopecia, acidosis • Bioavailability – varies from 100% in corn to 0% in wheat • Absorption – most takes place in proximal SI by active transport.

Biotin • Transport – Free or protein bound • Uptake – related to needs of cells • Storage – most found in muscle, liver and brain • Bacterial Synthesis of biotin – may be absorbed into body or excreted in feces

Biotin Functions • Biotin must be activated to biotinyl 5’adenylate – biotin reacts with a ATP (Mg required) – carboxylase joins the biotinyl moiety to form holoenzyme carboxylase with release of AMP • Biotin dependent enzymes – – acetyl Co. A carboxylase pyruvate carboxylase propionyl Co. A carboxylase Beta-methylcrotonyl Co. A carboxylase

Biotin • Carboxylases – biotin is attached by an amide linkage – carboxy terminus of biotin is linked to epsilon amino group of a specified lysine residue of apoenzyme – chain connecting biotin and apoenzyme is long and flexible • allows biotin to move from one active site to another • see figure 9. 23 and 9. 24 a

Pyruvate Carboxylase • adds a carboxyl group to pyruvate forming oxaloacetate – requires presence of acetyl Co. A as well as ATP and Mg • acetyl Co. A serves as allosteric activator – Fate of OAA • if surplus of ATP, gluconeogenic pathway • if deficiency of ATP, TCA cycle – see Figure 9. 24 b

Acetyl Co. A Carboxylase • Initiation of fatty acid synthesis • Transfers a carboxyl group to acetyl Co. A forming malonyl Co. A • Activated by citrate and isocitrate • Inhibited by palmitoyl Co. A

Propionyl Co. A Carboxylase • Important for catabolism of isoleucine, threonine and methionine and odd chain fatty acids • Catalyzes the conversion of propionyl Co. A to methylmalonyl Co. A • Methylmalonyl Co. A converted to succinyl Co. A via vitamin B 12 dependent enzyme

Beta-methylcrotonyl Co. A Carboxylase • During leucine catabolism – beta-methylcrotonyl Co. A is formed – carboxyl group added to betamethylcrotonyl Co. A to form betamethylglutaconyl Co. A • defect in BMCC results in accumulation of methyl crotonylglycine and hydroxyisovaleric acid – further catabolized to acetoacetate and acetyl Co. A
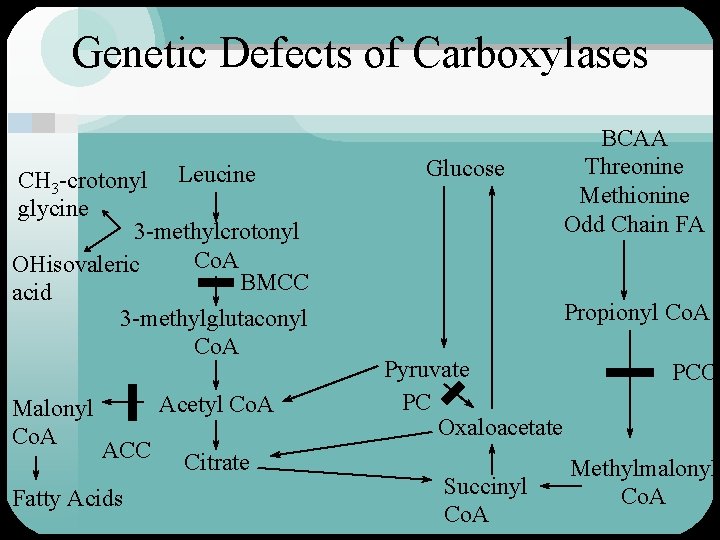
Genetic Defects of Carboxylases CH 3 -crotonyl Leucine glycine 3 -methylcrotonyl Co. A OHisovaleric BMCC acid 3 -methylglutaconyl Co. A Malonyl Co. A Acetyl Co. A ACC Fatty Acids Citrate Glucose BCAA Threonine Methionine Odd Chain FA Propionyl Co. A Pyruvate PC Oxaloacetate Succinyl Co. A PCC Methylmalonyl Co. A

Metabolism and Excretion • Catabolism of holocarboxylases by proteases yields biocytin • Biocytin degraded by biotinidase to yield free biotin • Some of free biotin is reused; some is further degraded to bisnorbiotin • See free biotin, bisnorbiotin and biocytin in urine primarily

Biotin • AI – 30 g/d (adult males and females) • Deficiency – depression, hallucinations, muscle pain, nausea, scaly dermatitis • excessive ingestion of raw egg white • GI disorders – IBD – achlorydia • Excessive alcohol ingestion • Certain medications

Biotin • Toxicity – none observed • Assessment – Evaluation of biotin in • blood, plasma or serum • urine
- Slides: 14