Biotherapeutics Understanding Immunogenicity for Clinical Practice Moderator Hendrik

Biotherapeutics Understanding Immunogenicity for Clinical Practice Moderator Hendrik Schulze-Koops, MD, Ph. D Professor Ludwig-Maximilians-University Munich, Germany

Faculty Hector Chinoy, Ph. D, FRCP Program Director Salford Royal NHS Foundation Trust Salford, United Kingdom Mark Lazarus, Ph. D, MRCP Doctor Southend University Hospital Southend, Essex, United Kingdom Gertjan Wolbink, MD Rheumatologist Sanquin Research Amsterdam, The Netherlands

Introduction • Biotherapeutics have improved the management of chronic inflammatory diseases, such as RA, [a] psoriasis, [b] and IBD[c, d] • As biologics are proteins rather than chemical substances, they can be recognized by the immune system of the patient and elicit an immune response[e] • As a result, clinicians may lack confidence in moving patients from conventional DMARD therapy onto biologics or switching patients to biosimilar products in clinical practice a. Smolen JS, et al. Ann Rheum Dis. 2017; 76: 960 -977; b. Gossec L, et al. Ann Rheum Dis. 2015; 75: 499 -510; c. Harbord M, et al. J Crohns Colitis. 2017; 11: 769 -784; d. Gomollón F, et al. J Crohns Colitis. 2017; 11: 3 -25; e. Bloem K, et al. Ther Drug Monit. 2017; 39: 327 -332.

Program Outline • Immunogenicity • ADAs – Development – Clinical relevance • Use of biosimilar agents for the treatment of patients with chronic inflammatory diseases

Immunogenicity of Biologics in Chronic Inflammatory Diseases Biologics have demonstrated efficacy across a range of disease states, but a substantial proportion of patients fail to respond or have an inadequate response[a] • With initial treatment (primary failure); or • Lose response over time (secondary failure) In a systemic review of chronic inflammatory disease states*, ADAs were detected in as many as 50% of patients[a] • Importantly, the incidence of ADA can vary greatly between same -class products and different patient populations[b] *Controlled and observational studies of biologics/biosimilars administered for treatment of RA, Ps. A, JIA, AS, RA, nr -ax. Sp. A, psoriasis, CD, and UC a. Strand V, et al. Bio. Drugs. 2017; 31: 299 -316; b. Gunn GR 3 rd, et al. Clin Exp Immunol. 2016; 184: 137 -146.

Complex Nature of ADAs Polyclonal • May include different isotypes (ie, Ig. G, Ig. A, Ig. M, or Ig. E) • Bind to different regions ("domains") of the drug molecule • Vary in binding strength • Can differ between patients Gunn GR 3 rd, et al. Clin Exp Immunol. 2016; 184: 137 -146.

Neutralizing and Non-Neutralizing Antibodies • Bind to drug and inhibit pharmacologic function by preventing target binding • Can inhibit drug activity soon after the drug is administered • Can form immune complexes upon binding drug • Bind to sites on the drug molecule that do not affect target binding; do not impact drug's PD activity • Do not inhibit the PD activity of the drug, but can lower the drug's systemic exposure by increasing the rate of drug clearance • Can form immune complexes upon binding drug It is a fallacy that only neutralizing antibodies are clinically relevant; both neutralizing and non-neutralizing antibodies are likely to have similar effects on efficacy due to the relatively rapid clearance of antigen-antibody complexes. Gunn GR 3 rd, et al. Clin Exp Immunol. 2016; 184: 137 -146

Neutralizing and Non-Neutralizing Antibodies (Cont. ) • Both neutralizing and non-neutralizing antibodies can impact clinical responses to biologics/biosimilars by forming immune complexes that may influence their pharmacokinetics and lowering serum concentrations Strand V, et al. Bio. Drugs. 2017; 31: 299 -316.
Impact of ADAs PK Efficacy Safety and Tolerability Treatment Survival Strand V, et al. Bio. Drugs. 2017; 31: 299 -316.

Factors Associated With Immunogenicity Structure/Target Molecule Immune Complex Formation • Differences in molecular structures of different agents • Rates of ADA formation are higher with chimeric TNF inhibitor m. Abs vs some fully human TNF inhibitor m. Abs and fusion proteins • Formation of immune complexes between biologics/ biosimilars and the target protein Strand V, et al. Bio. Drugs. 2017; 31: 299 -316. Background Therapy • Background immunosuppressive/antiproliferative therapy reduces immunogenicity Biologic Dose/Regimen • High biologic doses and induction therapy can be associated with decreased incidence of ADA • Continuous vs intermittent therapy has been less likely to illicit ADA • IV therapy is associated with less immunogenicity than SC administration
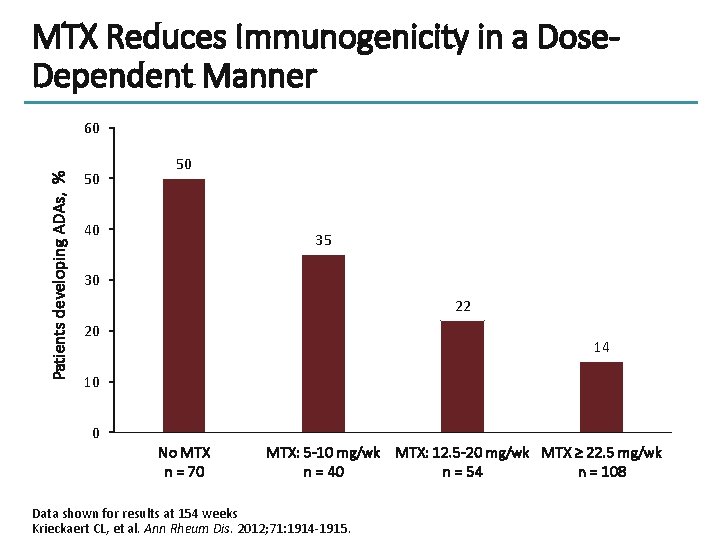
MTX Reduces Immunogenicity in a Dose. Dependent Manner Patients developing ADAs, % 60 50 50 40 35 30 22 20 14 10 0 No MTX n = 70 MTX: 5 -10 mg/wk MTX: 12. 5 -20 mg/wk MTX ≥ 22. 5 mg/wk n = 40 n = 54 n = 108 Data shown for results at 154 weeks Krieckaert CL, et al. Ann Rheum Dis. 2012; 71: 1914 -1915.

Biologic Agent Monitoring in Daily Practice The drug trough concentration may be the most useful test • There is a close relationship between biologic DMARD trough serum concentration and ADA development • There are difficulties associated with ADA detection/measurement Schaeverbeke T, et al. Rheumatology (Oxford). 2016; 55: 210 -220.

Model: ADA Development and Decreased Biologic Serum Concentration Figure No Longer Available Reproduced from Ann Rheum Dis. , van Schouwenburg PA, et al. , 72, 1680 -1686, 2013 with permission from BMJ Publishing Group Ltd.

Biologic Agent Monitoring in Daily Practice In the case of lack/loss of efficacy of a biologic agent, the dosage of the drug could give the most important information • If serum trough level is sufficient consider switching to another class of biologic agent • If serum trough level is low insufficient dose or immunization to the drug – Continue using same drug at increased dosage/more frequent administration or switch to another biologic Schaeverbeke T, et al. Rheumatology (Oxford). 2016; 55: 210 -220.

Measurement of Biologic Drug Serum Trough Concentration Several factors to consider • In which patients should the serum drug trough concentration be measured? • How often should the measurement be obtained? • When should the measurement(s) be obtained to receive the greatest value?
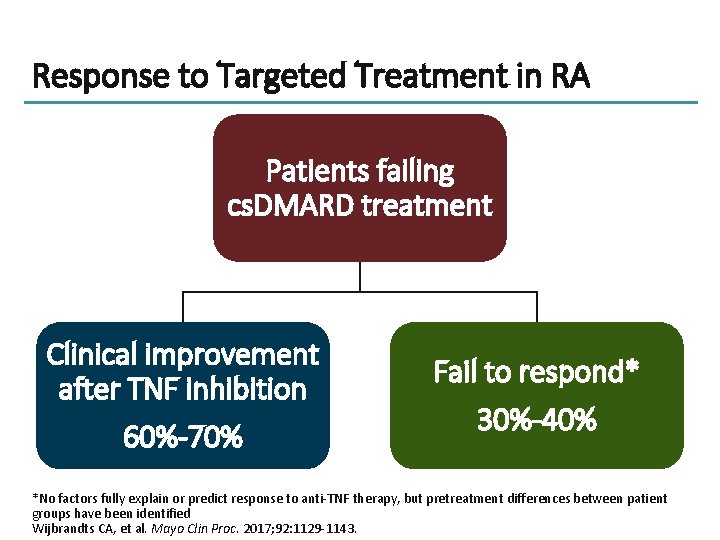
Response to Targeted Treatment in RA Patients failing cs. DMARD treatment Clinical improvement after TNF inhibition 60%-70% Fail to respond* 30%-40% *No factors fully explain or predict response to anti-TNF therapy, but pretreatment differences between patient groups have been identified Wijbrandts CA, et al. Mayo Clin Proc. 2017; 92: 1129 -1143.
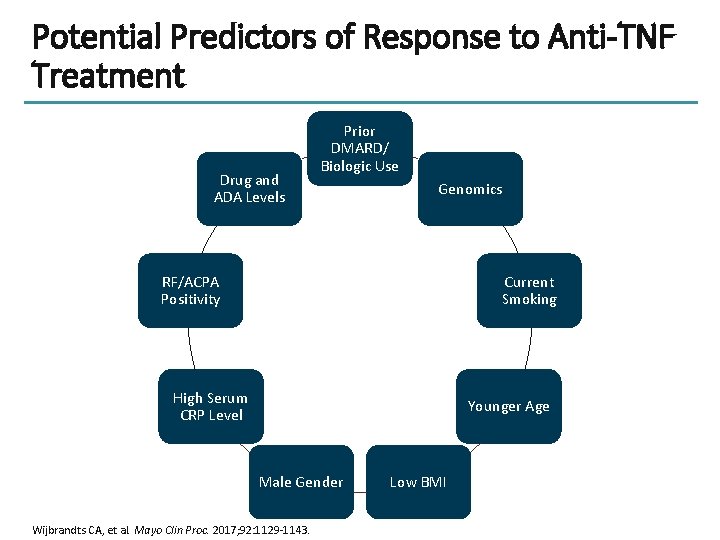
Potential Predictors of Response to Anti-TNF Treatment Drug and ADA Levels Prior DMARD/ Biologic Use Genomics RF/ACPA Positivity Current Smoking High Serum CRP Level Younger Age Male Gender Wijbrandts CA, et al. Mayo Clin Proc. 2017; 92: 1129 -1143. Low BMI

Considerations for Patients With Loss of Response to Biologic Therapy No/low drug concentrations Verify drug compliance/perform therapeutic drug monitoring High antibodies Adequate drug No antibodies Personal communication, Gertjan Wolbink, MD. Consider switching to another anti-TNF Consider switching out of therapeutic class
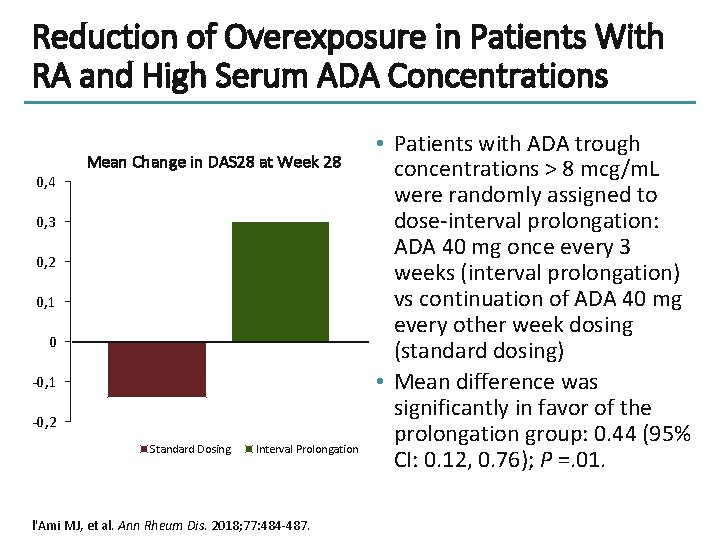
Reduction of Overexposure in Patients With RA and High Serum ADA Concentrations 0, 4 Mean Change in DAS 28 at Week 28 0, 3 0, 2 0, 1 0 -0, 1 -0, 2 Standard Dosing Interval Prolongation l'Ami MJ, et al. Ann Rheum Dis. 2018; 77: 484 -487. • Patients with ADA trough concentrations > 8 mcg/m. L were randomly assigned to dose-interval prolongation: ADA 40 mg once every 3 weeks (interval prolongation) vs continuation of ADA 40 mg every other week dosing (standard dosing) • Mean difference was significantly in favor of the prolongation group: 0. 44 (95% CI: 0. 12, 0. 76); P =. 01.

RA Treatment Based on Antibody Responses? Individualized Drug Dose Based on Trough Concentration/ADA Response One-Size-Fits-All Drug Dose Research Agenda

Serum Drug Levels of Biologic Agents in the Management of RA: A Systematic Review 12 studies (6 retrospective, 4 prospective, and 2 RCTs) including 1482 patients (1281 RA, 201 Sp. A) suggested: • Measurement of drug levels may be useful in patients in remission • Higher drug levels predict a longer relapse-free optimization • In patients with failure to a biologic agent, treatment may need individual adjustment according to the presence of drug levels or ADA • MTX influences the association between response and drug levels • Monitoring drug levels would allow optimal use of current biologic therapies, but more and better-quality studies are needed to draw definitive conclusions Martin-Lopez M, et al. Rheumatol Int. 2018; 38: 975 -983.
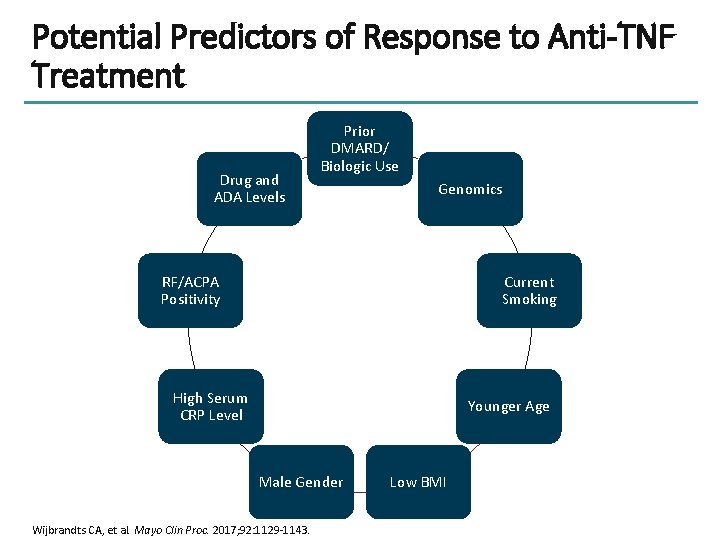
Potential Predictors of Response to Anti-TNF Treatment Drug and ADA Levels Prior DMARD/ Biologic Use Genomics RF/ACPA Positivity Current Smoking High Serum CRP Level Younger Age Male Gender Wijbrandts CA, et al. Mayo Clin Proc. 2017; 92: 1129 -1143. Low BMI

EULAR Recommendations for the Management of RA With cs. DMARDs and b. DMARDs Therapy with DMARDs should be started as soon as the diagnosis of RA is made MTX should be part of the first treatment strategy If the treatment target is not achieved with the first cs. DMARD strategy, when poor prognostic factors are present, addition of a b. DMARD should be considered Smolen JS, et al. Ann Rheum Dis. 2017; 76: 960 -977.

MTX Use in Patients With Ps. A or ax. Sp. A • If immunogenicity is a concern, should MTX be used in combination with b. DMARDs for the treatment of patients with Ps. A or ax. Sp. A, as it is for patients with RA? • Considering adding a b. DMARD to MTX vs discontinuing MTX and then adding a b. DMARD in patients with Ps. A or ax. Sp. A

Biosimilars and Their Reference Agents Are Interchangeable • Many clinical studies have compared the safety and efficacy of biosimilars to originator agents[a, b] • A systemic review of 90 studies, which included 7 molecular entities that treated 14 disease indications, and enrolled a total of 14, 225 individuals, reported[a] • The great majority of studies did not report differences in safety, efficacy, or immunogenicity after a single switch event vs patients who were not switched a. Cohen HP, et al. Drugs. 2018; 78: 463 -478; b. Schulze-Koops H, et al. Rheumatology (Oxford). 2017; 56: iv 30 -iv 48

The Data Requirements for Biosimilar Approval Are Comprehensive Figure No Longer Available Reprinted by permission from Springer Nature. Rheumatol Ther. Treatment Outcomes with Biosimilars: Be Aware of the Nocebo Effect, Rezk MF, et al. , Springer Nature 2017.

NOR-SWITCH: Switching From Originator Infliximab to Biosimilar CT-P 13 Risk Difference (of Disease Worsening) According to Disease* Figure No Longer Available Per protocol data set *CT-P 13 is not inferior to infliximab originator. Reprinted from Lancet. , 389, Jorgensen KK, et al. , Switching from originator infliximab to biosimilar CT-P 13 compared with maintained treatment with originator infliximab (NOR-SWITCH): a 52 -week, randomised, doubleblind, non-inferiority trial, 2304 -2316, Copyright 2017, with permission from Elsevier.
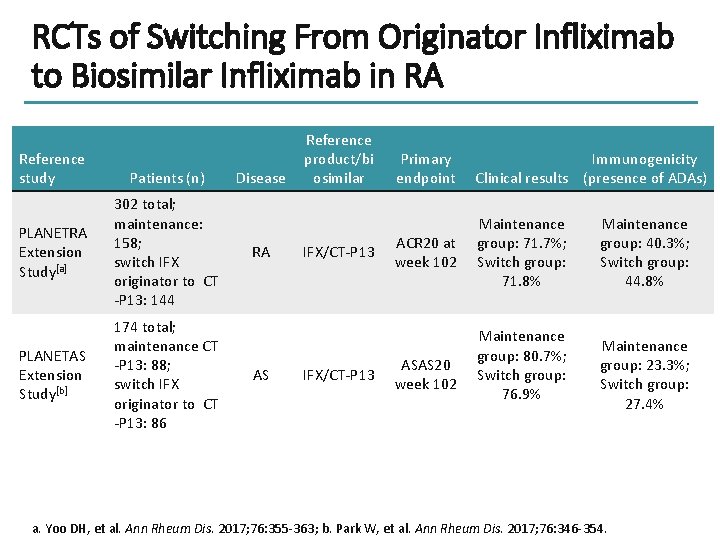
RCTs of Switching From Originator Infliximab to Biosimilar Infliximab in RA Reference study Patients (n) PLANETRA Extension Study[a] 302 total; maintenance: 158; switch IFX originator to CT -P 13: 144 PLANETAS Extension Study[b] 174 total; maintenance CT -P 13: 88; switch IFX originator to CT -P 13: 86 Disease RA AS Reference product/bi osimilar IFX/CT-P 13 Primary endpoint Immunogenicity Clinical results (presence of ADAs) ACR 20 at week 102 Maintenance group: 71. 7%; Switch group: 71. 8% ASAS 20 week 102 Maintenance group: 80. 7%; Switch group: 76. 9% Maintenance group: 40. 3%; Switch group: 44. 8% Maintenance group: 23. 3%; Switch group: 27. 4% a. Yoo DH, et al. Ann Rheum Dis. 2017; 76: 355 -363; b. Park W, et al. Ann Rheum Dis. 2017; 76: 346 -354.

Recommendations Regarding the Use of Biosimilars in Patients With RA EULAR evidence-based recommendations for the management of RA with cs. DMARDs and b. DMARDs[a] • Originator or respective EMA-approved biosimilar TNFinhibitors and biologics are recommended Consensus-based recommendations for the use of biosimilars to treat rheumatologic diseases[b] • Approved biosimilars can be used to treat appropriate patients in the same way as originator compounds • Current evidence indicates that a single switch from originator to biosimilar is safe and effective a. Smolen JS, et al. Ann Rheum Dis. 2017; 76: 960 -977; b. Kay J, et al. Ann Rheum Dis. 2018; 77: 165 -174.
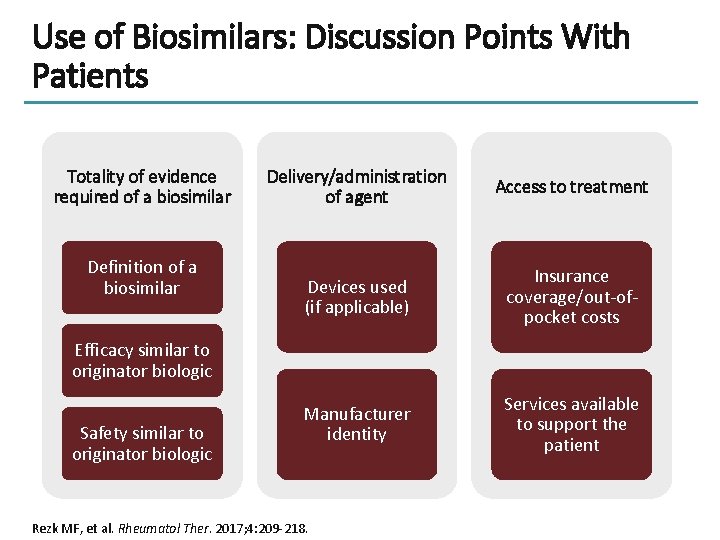
Use of Biosimilars: Discussion Points With Patients Totality of evidence required of a biosimilar Definition of a biosimilar Delivery/administration of agent Access to treatment Devices used (if applicable) Insurance coverage/out-ofpocket costs Manufacturer identity Services available to support the patient Efficacy similar to originator biologic Safety similar to originator biologic Rezk MF, et al. Rheumatol Ther. 2017; 4: 209 -218.

Biosimilar Switching: Evidence From Arthritis Studies Multiple switching studies have been conducted in various arthritis diseases[a] • RCTs (eg, NOR-SWITCH, [b] REFLECTIONS B 537 -02[c]) and registry studies (eg, DANBIO[d]) Efficacy consistent with historical controls of the reference/originator medicine[a] • Observed dropout rates with biosimilars are comparable to the rate observed with historical controls of the reference/originator medicine a. Cohen HP, et al. Drugs. 2018; 78: 463 -478; b. Jørgensen KK, et al. Lancet. 2017; 389: 2304 -2316; c. Cohen SB, et al. Arthritis Rheumatol. 2017; 69(suppl 10): 2798; d. Glintborg B, et al. Ann Rheum Dis. 2017; 76: 1426 -1431.

Thank you for participating in this activity. Please click Next below to see how your knowledge improved. The CME/CE posttest will follow. Please also take a moment to complete the program evaluation.
- Slides: 32