Bioterrorism Preparedness Smallpox Contingency Planning Dr Bonnie Henry

"Bioterrorism Preparedness: Smallpox Contingency Planning" Dr Bonnie Henry Associate Medical Officer of Health, Emergency Services Unit, Toronto Public Health

Public Health Role • Health effects of emergencies recently highlighted • MOH part of City EOC • Mandated lead role in events involving biologic agents

Public Health Role • Early Detection • Mass Patient Care • Mass Immunization/Prophylaxis • Epidemiologic investigation • Command Control

Public Health Role • Mass Fatality Management • Evacuations/sheltering • Environmental Surety • Community Recovery
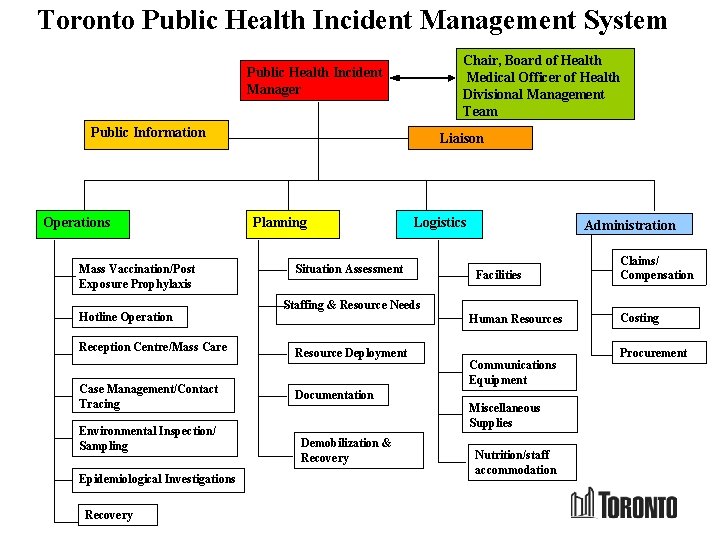
Toronto Public Health Incident Management System Chair, Board of Health Medical Officer of Health Divisional Management Team Public Health Incident Manager Public Information Liaison Operations Mass Vaccination/Post Exposure Prophylaxis Hotline Operation Reception Centre/Mass Care Case Management/Contact Tracing Environmental Inspection/ Sampling Epidemiological Investigations Recovery Planning Logistics Situation Assessment Administration Staffing & Resource Needs Human Resources Resource Deployment Documentation Demobilization & Recovery Claims/ Compensation Facilities Communications Equipment Miscellaneous Supplies Nutrition/staff accommodation Costing Procurement

Bioterrorism Preparedness

Bioterrorism is the intentional use of microorganisms (bacteria, viruses, and fungi) or toxins to produce death or disease in humans, animals or plants. Electron micrograph of anthrax bacteria Electron micrograph of ebola virus

Category A “Biologic Threat Agents” • Can be easily disseminated or transmitted person-to-person; • Cause high mortality, w/potential for major public health impact; • Might cause public panic and social disruption; and • Require special action for public health preparedness.

Biological Agents of Highest Concern Category A • Smallpox – variola major • Anthrax – Bacillus anthracis • Plague – Yersinia pestis • Botulism – Clostridium botulinum toxin • Tularemia – Francisella tularensis • Viral hemorrhagic fevers – arenaviruses, filoviruses (Ebola, Marburg, Lassa, Junin)

Category B: Second Highest Priority • Moderately easy to disseminate • Cause moderate morbidity and low mortality • Require specific enhancements of diagnostic capacity and enhanced disease surveillance • • • Coxiella burnetti (Q fever) Brucella Burkholderia mallei (glanders) Alphaviruses (Venezuelan encephalomyelitis and Eastern and Western equine) Rickettsia prowazekii Toxins (Ricin, Staph enterotoxin B) Chlamydia psittaci Food safety threats (e. g. Salmonella, Shigella. E. coli O 157: H 7) Water safety threats (Vibrio cholerae, Cryptosporidium parvum)

Category C: Third Highest Priority • Pathogens that could be engineered for mass destruction because of availability, ease of production and dissemination and potential for high morbidity and mortality and major health impact • Nipah virus • Hantavirus • Tickborne hemorrhagic fever viruses • Tickborne encephalitis viruses • Yellow fever • MDR TB

Characteristics of Bioterrorist Agents • • • Mainly inhaled - may be ingested or absorbed Particles may remain suspended for hours May be released silently with no immediate effect Person-to-person spread happens for some agents Long incubation periods mean "first responders” may be primary health care providers • Agents may be lethal or incapacitating • Vaccines & antitoxins exist for some agents

Recent Examples of Bioterrorism 1984: Salad bars contaminated with Salmonella to influence local election in Oregon / 751 people affected (8 salad bars) 1995: Sarin nerve gas release by Aum Shinrikyo in Tokyo subway / At least 9 failed attempts to use biological weapons 1996: Pastries contaminated with Shigella by disgruntled lab worker in Dallas

Recent Examples of Bioterrorism Former Soviet Union’s extensive biological weapons program thought to have found their way to other nations Iraq acknowledged producing and weaponizing anthrax and botulinum toxin Currently, at least 17 nations believed to have biological weapons programs

Anthrax: Soviet incident An accident at a Soviet military compound in Sverdlovsk (microbiology facility) in 1979 resulted in an estimated 66 deaths downwind.

Smallpox • Variola virus • Declared eradicated by WHO in 1980 • Civilian vaccination stopped 1972, healthcare workers stopped in 1977 and CF stopped 1988 • Known stockpiles remain in CDC and Institute for Viral Preparations, Moscow • Virus spread by aerosol • Incubation period: average 12 days (7 -19 days)

Last Case, Variola major Rahmina, 1975 Rahmina Banu, 2001

Smallpox • Clinical symptoms: abrupt onset of malaise, fever, rigors, headache, emesis, backache, delirium (15%) • Onset of rash 2 -3 days later on face, hands, forearms, and legs, then spreading centrally – Lesions progress from macules to papules to pustular vesicles – Lesions typically in same stage of development • Patients highly infectious during initial respiratory phase and until all eschars are off • Mortality in unvaccinated about 30%
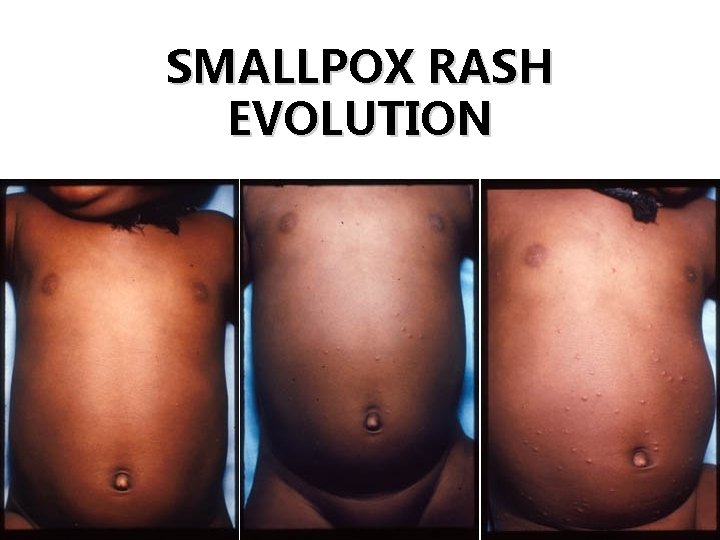
SMALLPOX RASH EVOLUTION Day 1 Day 2 Day 3
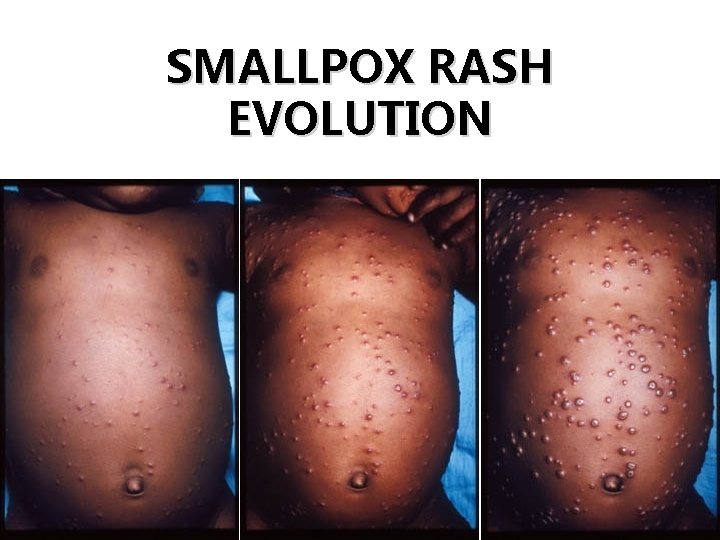
SMALLPOX RASH EVOLUTION Day 4 Day 5 Day 7
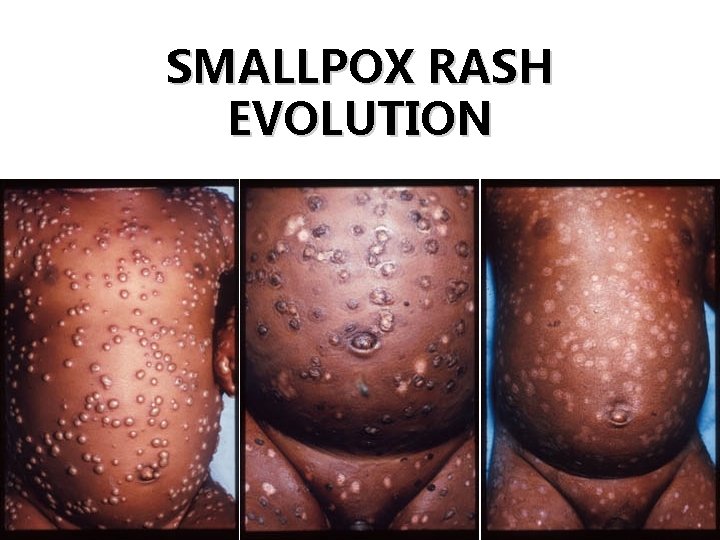
SMALLPOX RASH EVOLUTION Days 8 -9 20 Days 10 -14 Day

Smallpox Characteristics differentiating the rashes of Smallpox and Varicella Variola Varicella Centrifugal Centripetal Lesions all at the same stage Lesions in various stages Slow evolution Rapid evolution Deep lesions: circular and regular Scarring: severe Superficial lesions: oval or irregular Scaring: Mild

Smallpox • Vaccination – Within 3 days will likely prevent disease – Within 5 days is life-saving (ameleorates) – Canada has about 320, 000 doses – ? long term immunity – Cell culture and oral vaccine in research – Research on antivirals also ongoing (particularly Cidofovir)

TYPES OF SMALLPOX Type Proportion of cases (%) Case fatality rate (%) Variola major 90 30 Variola minor 2* <1 Hemorrhagic <3 100 Flat/malignant 7 97 * 25% of vaccinated cases present as variola minor
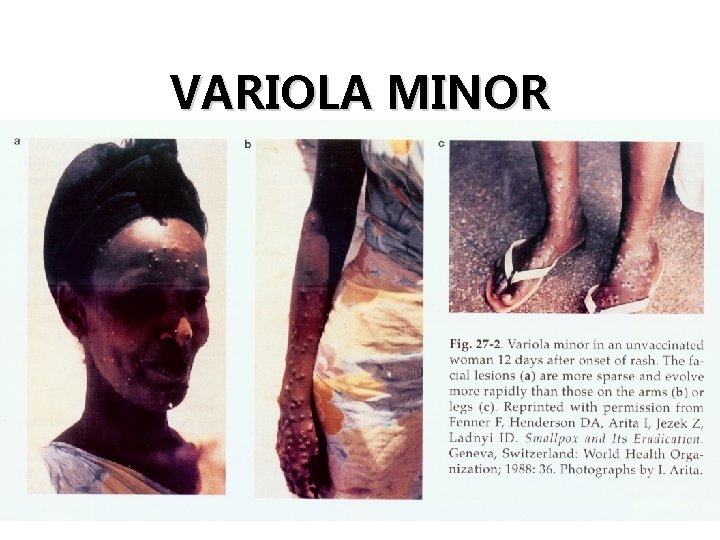
VARIOLA MINOR
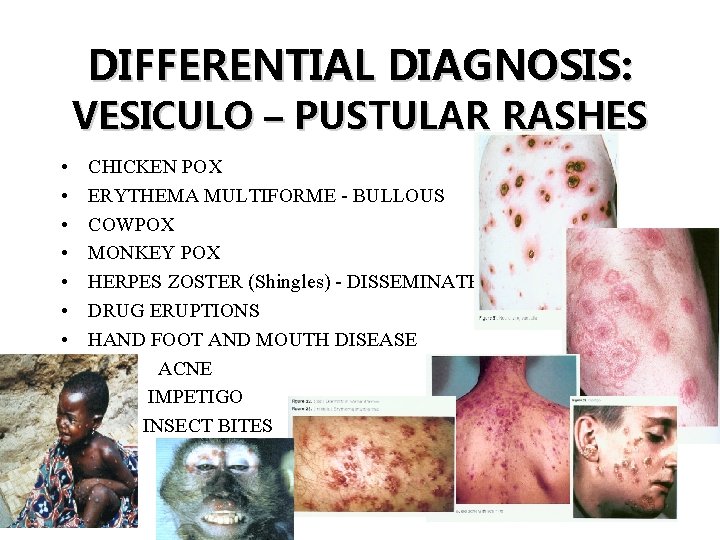
DIFFERENTIAL DIAGNOSIS: VESICULO – PUSTULAR RASHES • • • CHICKEN POX ERYTHEMA MULTIFORME - BULLOUS COWPOX MONKEY POX HERPES ZOSTER (Shingles) - DISSEMINATED DRUG ERUPTIONS HAND FOOT AND MOUTH DISEASE ACNE IMPETIGO INSECT BITES

Today’s Perspective in Canada: Pros vs Cons • “Moderately” contagious • Virus not robust • No natural reservoir • Able to vaccinate • Able to control • Improved medical care • Better pop’n health • • 30% mortality Misdiagnosis Long incubation Low level of “Immunity” • Pop’n mobility • Immuno-compromised • Mass panic, hysteria

National Smallpox Contingency Plan (v. 4) • Canada’s ‘search and contain’ strategy highlights: – Early detection, immediate notification – Immediate isolation of cases – Immediate deployment of smallpox responders – Immediately vaccinate all those directly exposed, all known direct contacts, all local personnel… – Intensive contact tracing – Rapid set up of isolation facilities – Rapid set-up of local Smallpox assessment centres • Assumption: In the absence of smallpox anywhere in Canada A risk of disease and death from a vaccine, no matter how small, may be unacceptable Especially when preattack vaccination is considered

Political Divisions • Canada’s search and contain strategy consists primarily of public health measures, which fall under provincial/territorial jurisdiction • Federal role: – – Immediate mobilization of vaccine Deployment of ‘federalized’ smallpox response teams (SERF) Provision of supplies 24 -hour support line to the public, professional and other governments – International notification and consultation

Smallpox Isolation, Toronto (1909)

“WHO’s success with isolation” WHO’s experience in India : • 1960 – 1973 Smallpox transmission continued during this time under a mass vaccination strategy. • In 1973, a search and containment strategy was introduced, stressing isolation of cases. • Smallpox was then eliminated in just two years, in 1975. We will come back to this….
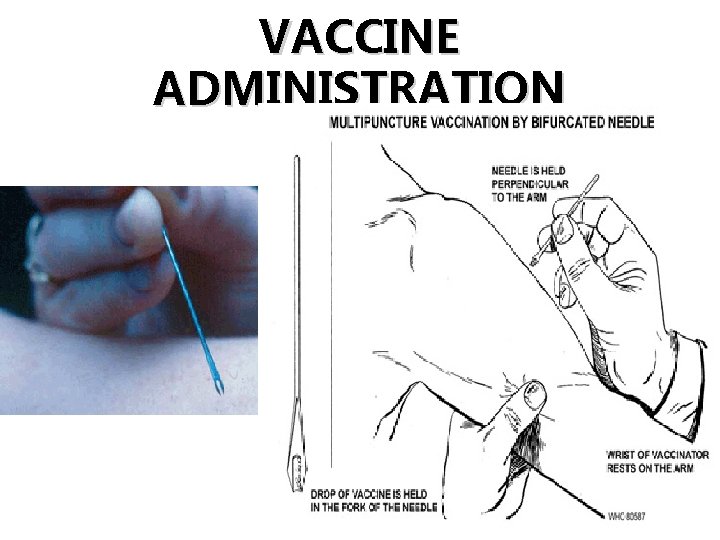
VACCINE ADMINISTRATION
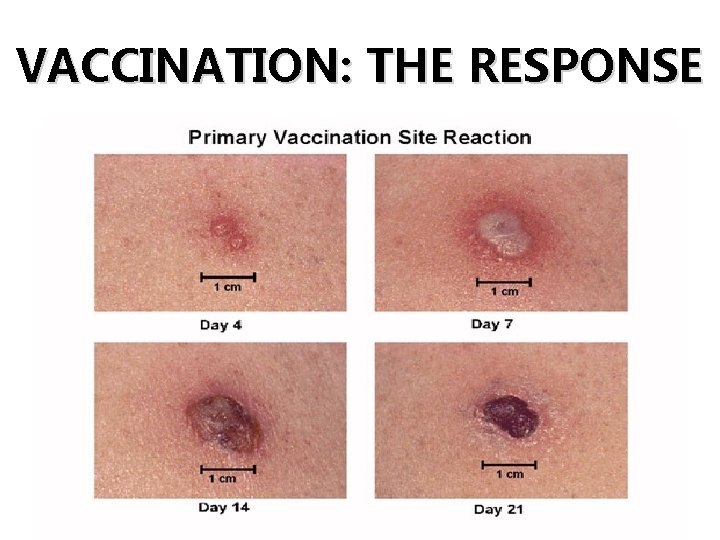
VACCINATION: THE RESPONSE

VACCINE CONTRAINDICATIONS • History or presence of eczema • Other acute , chronic or exfoliative skin condition • Immunosuppression ( HIV, AIDS, cancer, immunodeficiency disorders, chemotherapy, radiotherapy, organ transplant, high dose corticosteroids • Pregnancy • History of anaphylaxis to a vaccine component
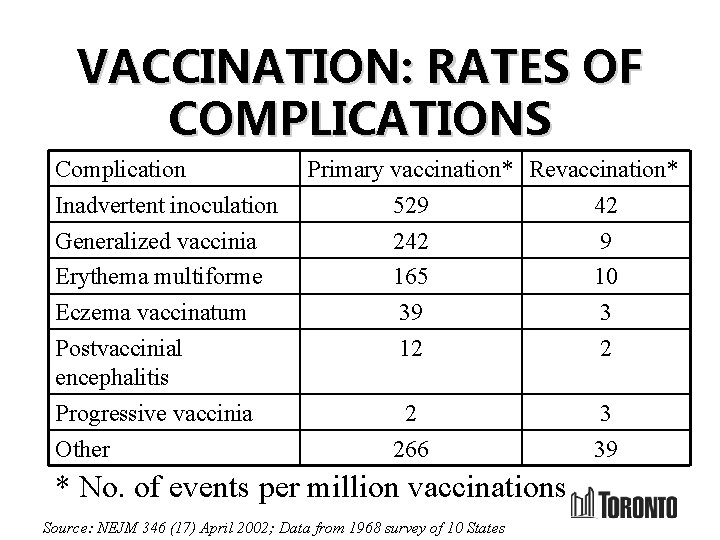
VACCINATION: RATES OF COMPLICATIONS Complication Inadvertent inoculation Generalized vaccinia Erythema multiforme Eczema vaccinatum Postvaccinial encephalitis Progressive vaccinia Other Primary vaccination* Revaccination* 529 42 242 9 165 10 39 3 12 2 2 266 * No. of events per million vaccinations Source: NEJM 346 (17) April 2002; Data from 1968 survey of 10 States 3 39

Consider Recent Smallpox Response Models • Kaplan et al. (Proc Natl Acad Sci USA) • Halloran et al. (Science) • [Mention: – Epstein et al. (Brookings Working Paper) – Bozzette et al. (N Eng J Med)]

Technical Discussions Highlight Different Modeling Approaches • Kaplan et al. – free mixing; explicit logistics • Halloran et al. – “structured stochastic simulation” – [Epstein et al. – agent-based – Bozzette et al. – simulation with assumed response efficacy from historical data]

Other Factors Matter More • Scale of model – Kaplan et al. consider population of 10 million – Halloran et al. look at “community “ of 2, 000 • [Epstein et al. consider “county” of 800 • Bozzette et al. – no role for population in model]

Other Factors Matter More • Rate of vaccination and logistics – Traced (ring, targeted) vaccination proceeds with the pace of the epidemic – need to see symptomatic cases to trigger vaccination – Mass vaccination proceeds at a pace limited only by available resources • number of vaccinators • time required to vaccinate

Important To See If Models Have Different Policy Implications • To do so, need to control for inputs as much as possible to see if different assumptions on model structure lead to different results

Kaplan et al. (PNAS) • • Focus on a large city (10, 000) Construct “traced vaccination” (TV) model Contrast with “mass vaccination” (MV) Consider TV/MV switch if TV fails to control outbreak after 2 generations of cases • Consider pre-attack vaccination

Kaplan et al. (PNAS) • Disease transmission/progression: 4 disease stages (includes infected but vaccine sensitive), free mixing in population (“worst case”), imperfect vaccination and (low) vaccine-related mortality • Response logistics: consistent tracing with disease transmission/progression linked to index case (“race to trace”), TV queues (finite TV capacity), MV rate higher than TV rate, quarantine capacity requirements • State transitions governed by both disease transmission/progression and response logistics; epidemic and response are on the same time scale!

TV or MV: 50% Tracing Accuracy • MV is optimal (fewer deaths) for any R 0 > 1. 3

TV or MV: 100% Tracing Accuracy • Still favor MV for any R 0 > 2 • If initial attack > 20, favor MV for R 0 > 1. 3 (same as 50% tracing accuracy)

TV or MV: Asymmetries • Consequences of choosing the wrong policy are not symmetric! • If TV is optimal, choosing MV would lead to few incremental deaths • If MV is optimal, choosing TV could lead to a disaster with many incremental deaths • Would therefore suggest choosing TV only if extremely confident (i. e. highly certain) that initial attack size and R 0 fall on the TV-favorable side of the tradeoff curve
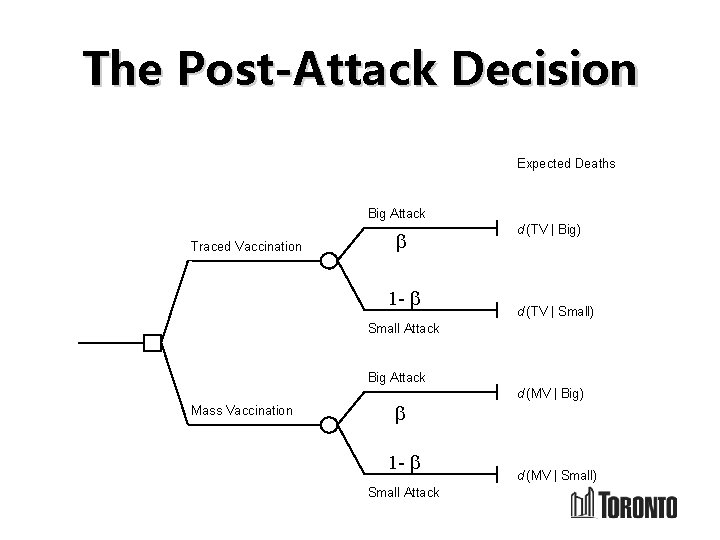
The Post-Attack Decision Expected Deaths Big Attack Traced Vaccination b 1 - b d (TV | Big) d (TV | Small) Small Attack Big Attack Mass Vaccination b 1 - b Small Attack d (MV | Big) d (MV | Small)

The Post-Attack Decision: Example • Suppose attack/response yields deaths as: • Choose MV if b > 7. 4 x 10 -5

Switching Helps, But Delay is Costly • In base case, switching from TV to MV after two generations of cases (28 days) results in 15, 570 cases and 4, 680 deaths • Cost of delay is high – 4, 120 incremental deaths compared to MV • Given option to switch, still would only start with TV if extremely confident that both R 0 and initial attack size are small

Pre-Attack Vaccination • Reduces degree of susceptibility in the population • Effect is to reduce R 0 and initial attack size • Pre-attack vaccination makes post-attack TV more attractive as a result

TV with Pre-Attack Vaccination

Pre-Attack Vaccination? • Suppose 100% successful pre-attack vaccination – expect 10 vaccine-related deaths • Let a = Pr{Smallpox Attack}, d(p) = deaths post attack from response policy p – Note: think of attack risk over 5 -10 year time frame • Solve 10 = a d(p) for a; consider pre-attack vaccination if perceived attack risk exceeds a • Base case results: – for p = TV, a = 9 in 100, 000 – for p = MV, a = 1. 8% (!!) – for p = TV/MV (CDC policy), a = 2 in 1, 000

Pre-Attack Vaccination? • Take home message: decision to vaccinate preattack should depend not only on the risk of vaccine and attack, but also on the response policy • If one does not have confidence in the response policy, one is much more likely to favor pre-attack vaccination (i. e. a is very small) • If one is confident that the response policy could contain an attack, desire for pre-attack vaccination lessens (i. e. a is larger)

Build the Button Now? Think like a terrorist: a¢ > a (An attack is less likely if you prepare)

Policy Conclusions • Optimal response policy depends critically on beliefs regarding initial attack size and R 0 • MV allows many fewer deaths and is much faster over a wide range of scenarios • TV or TV/MV switch are best if highly certain that R 0 and initial attack size are very small, or if pre-attack vaccination greatly reduces R 0 • Vaccine complications not an issue in choosing postattack response policy – any successful policy will vaccinate large percentage of population in big attack • Death-minimizing decision to vaccinate pre-attack should depend upon the risk of vaccine and attack, and the post-attack response policy employed

Halloran et al. (Science) • Uses “structured stochastic simulator” • Looks at 2, 000 person “community” of four neighborhoods, one high school, one middle school, two elementary schools, play groups and day care centers • Introduces 1 -5 infected terrorists who mingle in population

Main Finding • Absent residual immunity from vaccinations among adults 20+ years ago, deaths under TV only a factor of 2 higher than deaths under MV • With residual immunity, TV does better • Attributes difference from Kaplan “factor of 200” TV/MV death ratio to difference between structured and free mixing

A Different Interpretation. . . • If we place the Science inputs (population of 2, 000, single initial infection, R 0 = 3. 2, 80% vaccination coverage, response delays to match the detection of smallpox after the 1 st, 15 th, and 25 th case) look what happens: Deaths per 1000 Halloran et al (1) Kaplan et al (2) 80% MV after: 1 case 0. 9 0. 4 15 th case 9. 4 6. 4 25 th case 13. 7 17. 8 80% TV after: 1 case 10. 9 8. 8 15 th case 19. 6 12. 0 25 th case 28. 2 33. 9

What Is Going On? • Newly identified cases required to trigger contact tracing – TV proceeds with the pace of epidemic – Number of deaths scales with population size; independent of initial infections • MV operates on its own timetable – 10 days in the examples above – Number of deaths depends on initial infections; independent of the population size • Ratio of deaths from TV/MV grows with population size

Canadian situation • 12. 5 million Canadians • Local populations are with no vaccination to connected by migration of smallpox individuals • Over 64% of Canada’s • By air alone: population live in the – Toronto-Chicago (1, 000/year) nation’s 27 census – Toronto-Vancouver metropolitan areas (822, 000/year) • 79. 4% of Canadians live in – Toronto-Montreal an urban centre of >10, 000 (1, 257, 000/year)

Need to consider Population Density • Population: • Population density determines how fast the infection may spread – (R 0 is proportional to population density) • Population density determines the amount of effort for control and eradication • Canada: 30, 007, 094 Toronto: 4, 682, 897 Montreal: 3, 426, 350 Vancouver: 1, 986, 965 • Population density: • Canada: 3. 3/km 2 Toronto 793/km 2 Montreal 847/km 2 Vancouver 690/km 2 Kitchener 501/km 2 Hamilton 483/km 2 Oshawa 328/km 2 Windsor 301/km 2

Important Caveat • All of the models are “closed” – that is, no immigration or births – what about importing cases from one area to another? – historically, case importation allowed for “continued transmission” following widespread vaccination • Suppose you are the MOH of Toronto, and smallpox is detected in Vancouver – what is your new assessment of attack probability in Toronto? – do you worry about importing a case from Vancouver? – what do your citizens want?
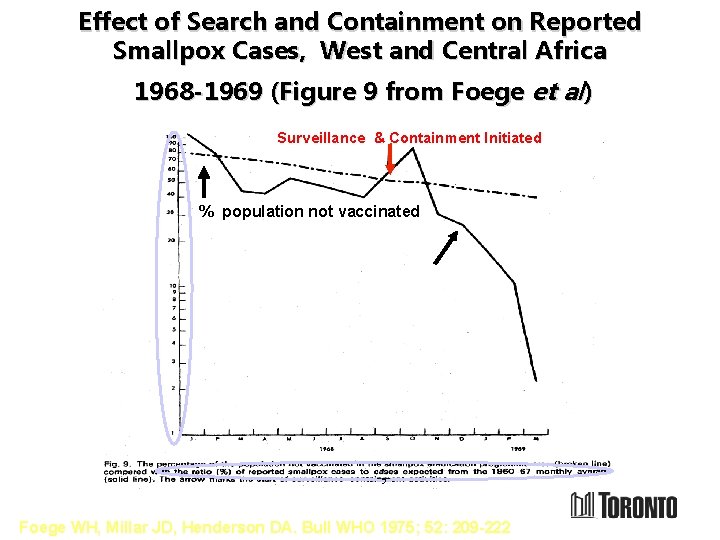
Effect of Search and Containment on Reported Smallpox Cases, West and Central Africa 1968 -1969 (Figure 9 from Foege et al) Surveillance & Containment Initiated % population not vaccinated Smallpox cases reported/expected ratio ; Foege WH, Millar JD, Henderson DA. Bull WHO 1975; 52: 209 -222

Decline in Reported Smallpox Cases Matches Decline in Susceptibility Over Time
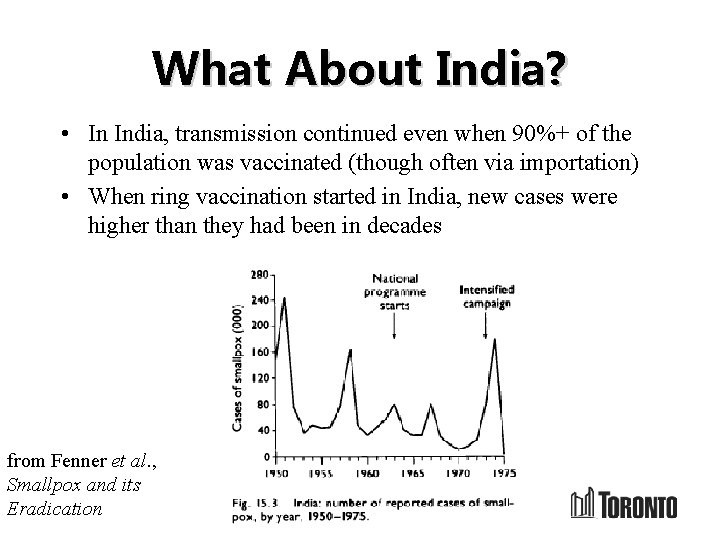
What About India? • In India, transmission continued even when 90%+ of the population was vaccinated (though often via importation) • When ring vaccination started in India, new cases were higher than they had been in decades from Fenner et al. , Smallpox and its Eradication

But Accounting For Population. . .

Policy Lessons • In all of the models (and in West and Central Africa, and in India), smallpox deaths decline as vaccination coverage increases • Absent pre-existing immunity (or pre-attack vaccination), both PNAS and Science explicitly show fewer deaths from postattack mass vaccination

Questions for us to Consider • Current Federal policy starts with surveillancecontainment – Should the policy begin with local MV instead (with priority to known close contacts)? • How many persons should be vaccinated now to “build Canada’s button? ” – 500? 5, 000? 500, 000? – answer depends on response policy and scale – In US: 500, 000 now; 10 million later this year; voluntary for public next year

Questions for us to Consider • “Vaccination within 2 -3 days after initial exposure to smallpox almost always prevents disease” – how confident are we in this claim? – if claim is wrong, would we do the same anyway? • Contact tracing – plan calls identifying both close contacts, and also contacts in: restaurant; grocery store; gas station; hair stylist; sporting event; movie theatres. . . – is it efficient to spend time searching for distant contacts at expense of more rapid clinic vaccination?

Questions for us to Consider • Is there a case for urban versus rural policies? – Ring vaccination is much more likely to work in a rural environment where people don’t travel as much, whereas in the urban setting (where 70% of Canadians live), tracing will be much tougher.

“The only thing more difficult than planning for an emergency is having to explain why you didn’t”
- Slides: 70