Biosynthesis of Terpenes Chem465 Isopentenyl Pyrophosphate The Biological

Biosynthesis of Terpenes Chem-465

Isopentenyl Pyrophosphate: The Biological Isoprene Unit

References Dellas, N. , Thomas, S. T. , Manning, G. , and Noel, J. P. (2013) Discovery of a metabolic alternative to the classical mevalonate pathway. e. Life 2, e 00672 2. Vinokur JM, Korman TP, Cao Z, Bowie JU (2014) Evidence of a novel mevalonate pathway in archaea. Biochemistry 53: 4161– 4168.

Azami Y, Hattori A, Nishimura H, Kawaide H, Yoshimura. T, Hemmi H (2014) (R)-mevalonate-3 -phosphate is an intermediate of the mevalonate pathway in Thermoplasma acidophilum. J Biol Chem 289: 15957– 15967. 4. Goldstein, J. L. , and Brown, S. B. (1990) Regulation of the mevalonate pathway. Nature 343, 425− 430

Biosynthesis of terpenes may be studied in two parts: (A) Synthesis of Activated 5 -C units: (B) Condensation of activated 5 -C units IPP and DPP to form terpenes: DPP=Dimethyallyl pyrophosphate IPP=Isopentenyl pyrophosphate
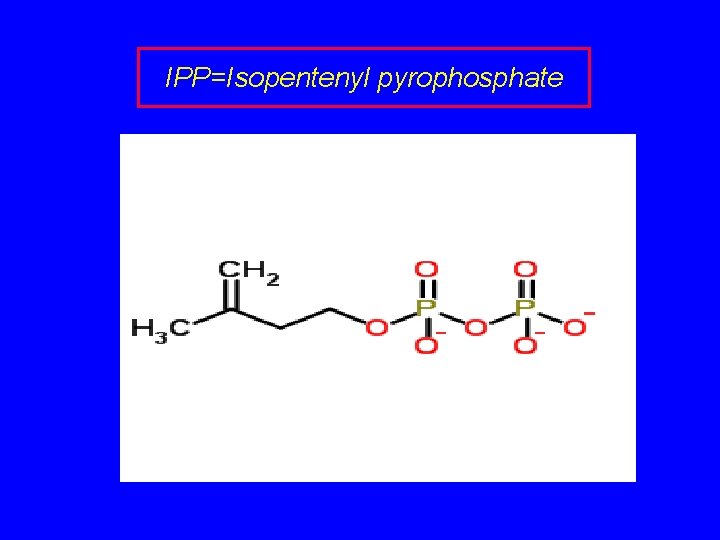
IPP=Isopentenyl pyrophosphate
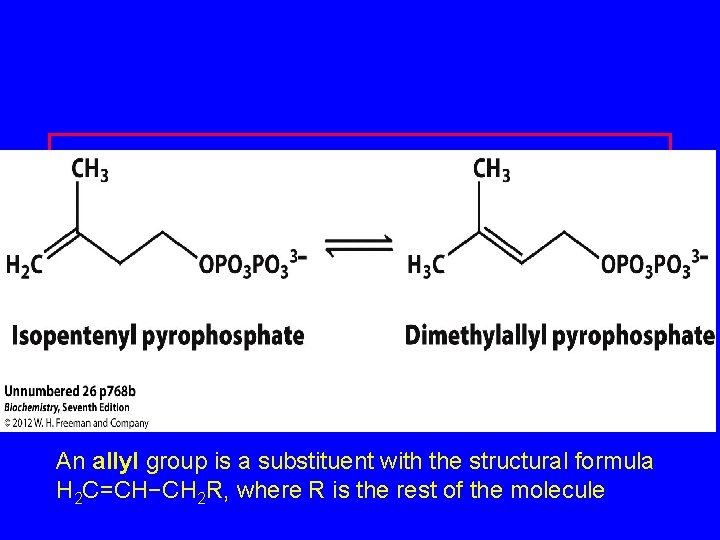
DPP=Dimethyallyl pyrophosphate An allyl group is a substituent with the structural formula H 2 C=CH−CH 2 R, where R is the rest of the molecule

The Biological Isoprene Unit The isoprene units in terpenes do not come from isoprene. They come from isopentenyl pyrophosphate. Isopentenyl pyrophosphate (5 carbons) comes from acetate (2 carbons) via Mevalonate (6 carbons).
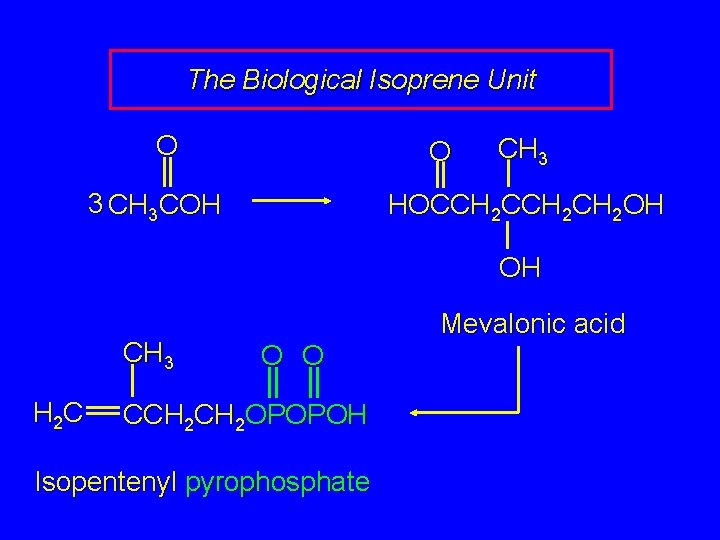
The Biological Isoprene Unit O O 3 CH 3 COH CH 3 HOCCH 2 CH 2 OH OH CH 3 H 2 C Mevalonic acid O O CCH 2 OPOPOH Isopentenyl pyrophosphate
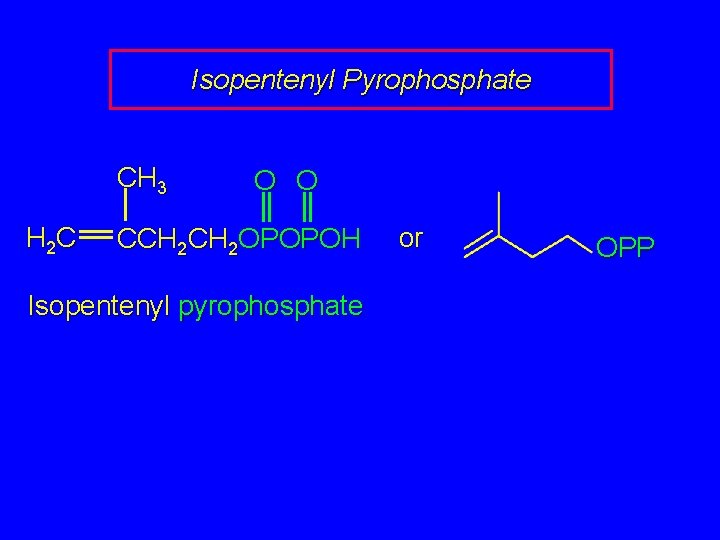
Isopentenyl Pyrophosphate CH 3 H 2 C O O CCH 2 OPOPOH Isopentenyl pyrophosphate or OPP
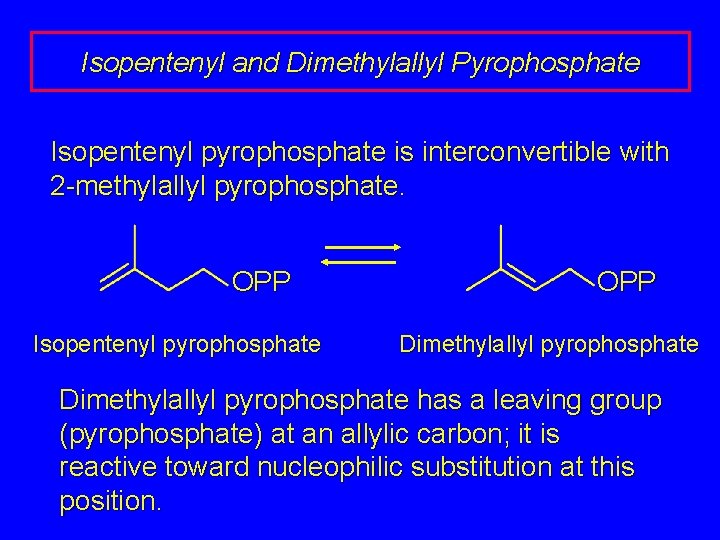
Isopentenyl and Dimethylallyl Pyrophosphate Isopentenyl pyrophosphate is interconvertible with 2 -methylallyl pyrophosphate. OPP Isopentenyl pyrophosphate OPP Dimethylallyl pyrophosphate has a leaving group (pyrophosphate) at an allylic carbon; it is reactive toward nucleophilic substitution at this position.

Carbon-Carbon Bond Formation in Terpene Biosynthesis
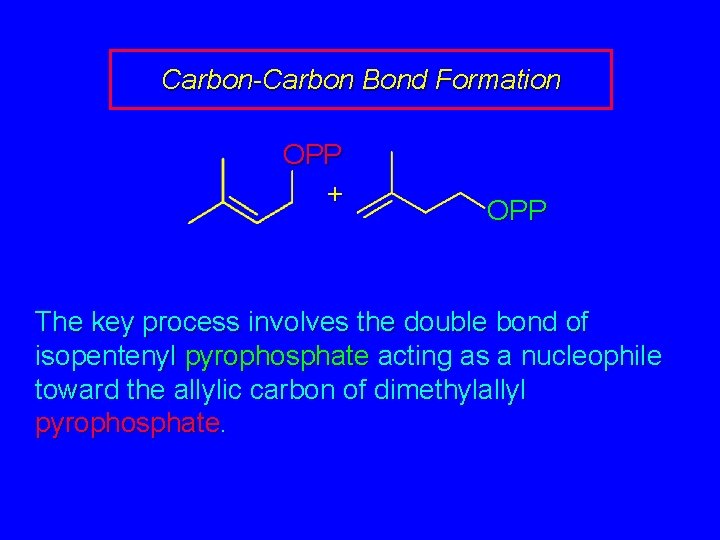
Carbon-Carbon Bond Formation OPP + OPP The key process involves the double bond of isopentenyl pyrophosphate acting as a nucleophile toward the allylic carbon of dimethylallyl pyrophosphate.
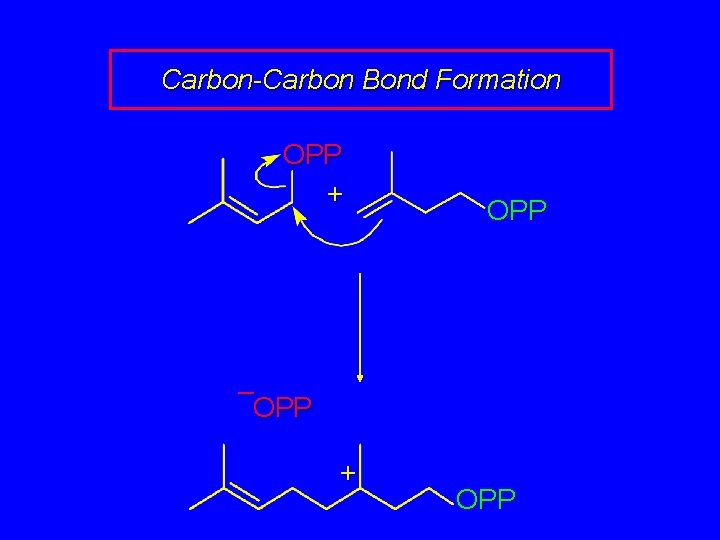
Carbon-Carbon Bond Formation OPP + OPP – OPP + OPP
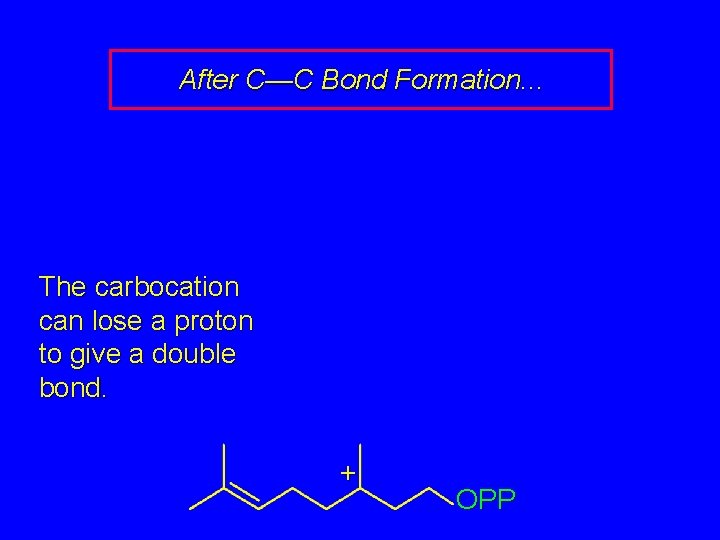
After C—C Bond Formation. . . The carbocation can lose a proton to give a double bond. + OPP
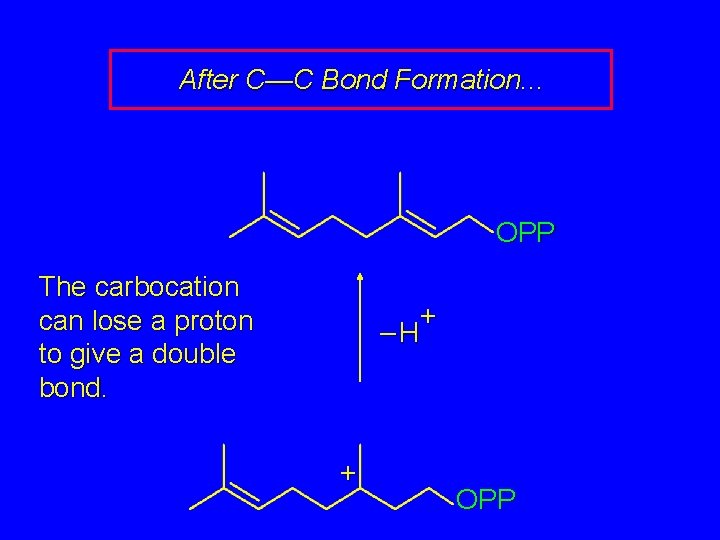
After C—C Bond Formation. . . OPP The carbocation can lose a proton to give a double bond. + –H + OPP
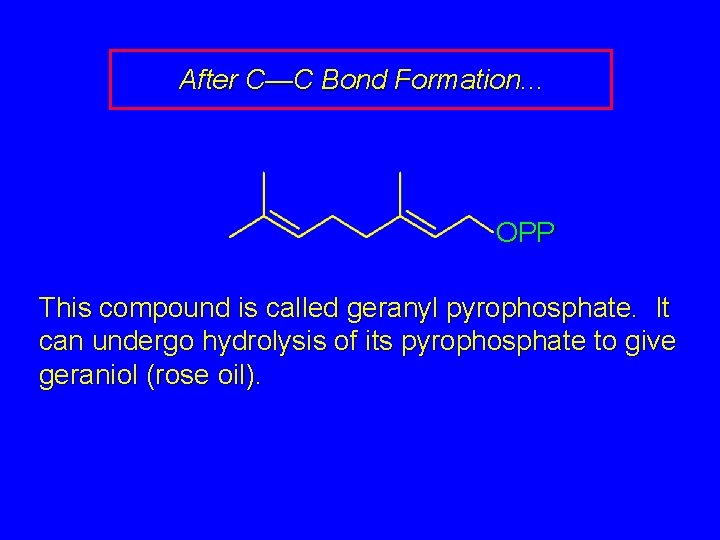
After C—C Bond Formation. . . OPP This compound is called geranyl pyrophosphate. It can undergo hydrolysis of its pyrophosphate to give geraniol (rose oil).
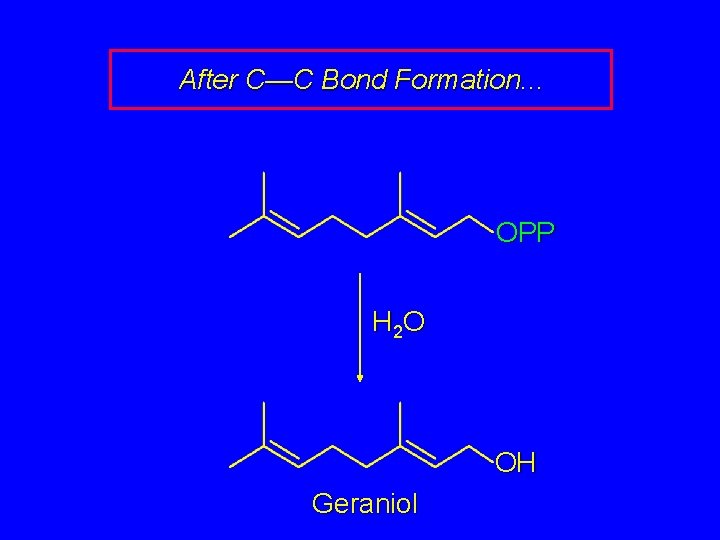
After C—C Bond Formation. . . OPP H 2 O OH Geraniol
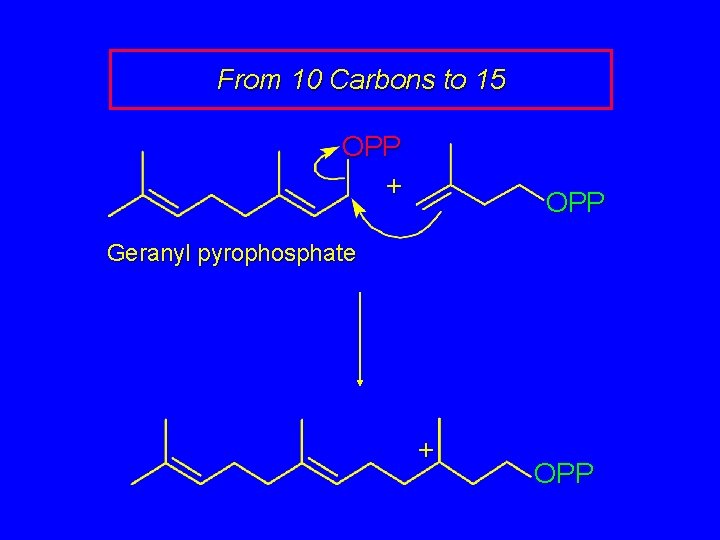
From 10 Carbons to 15 OPP + OPP Geranyl pyrophosphate + OPP
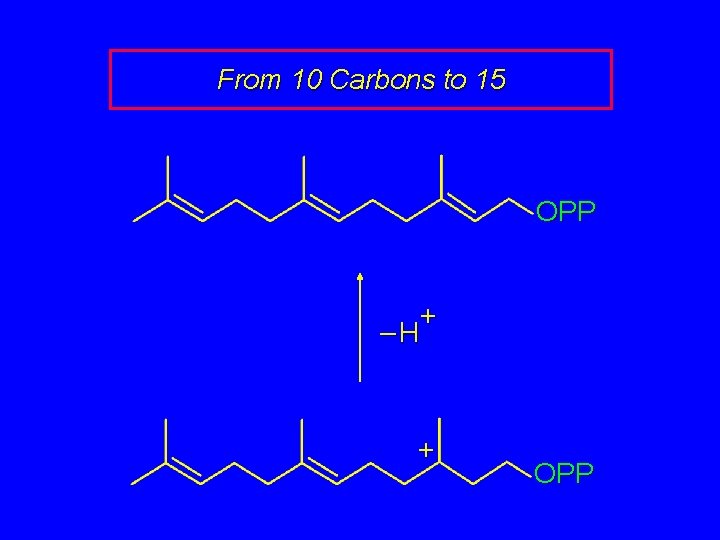
From 10 Carbons to 15 OPP + –H + OPP
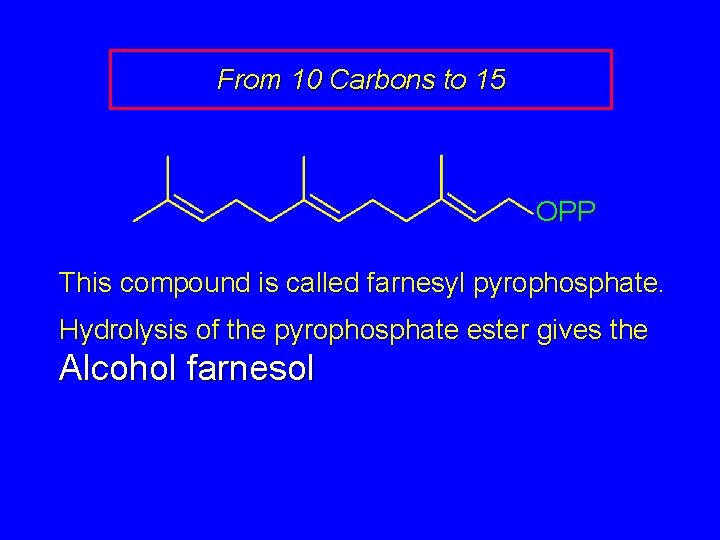
From 10 Carbons to 15 OPP This compound is called farnesyl pyrophosphate. Hydrolysis of the pyrophosphate ester gives the Alcohol farnesol
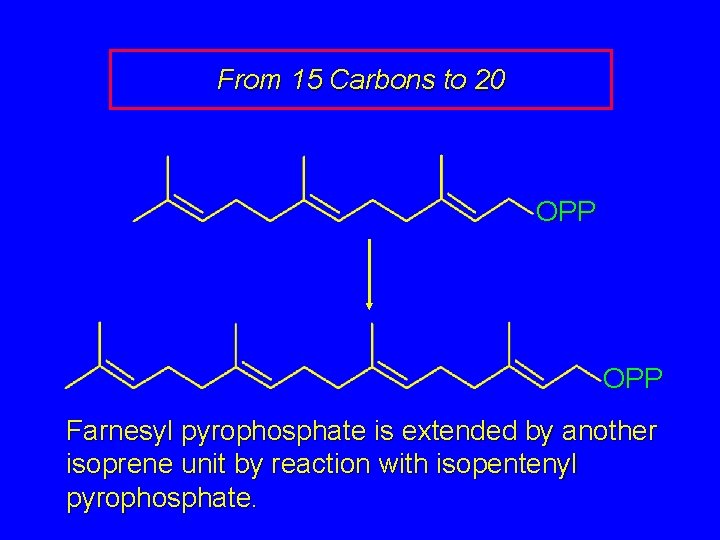
From 15 Carbons to 20 OPP Farnesyl pyrophosphate is extended by another isoprene unit by reaction with isopentenyl pyrophosphate.
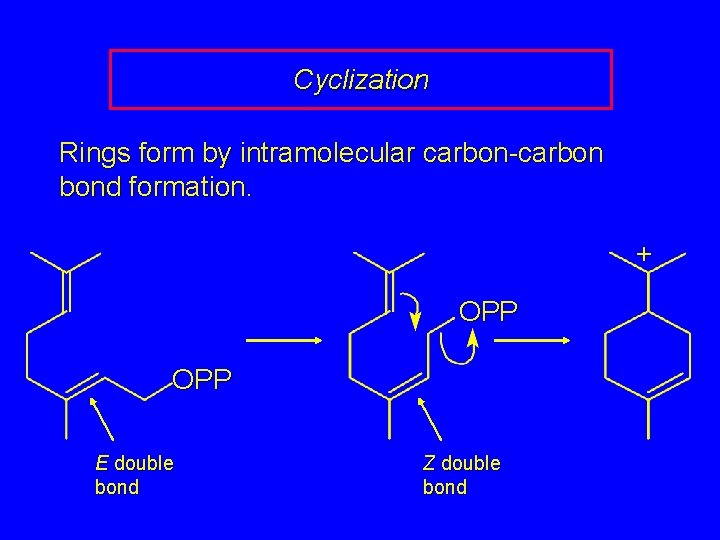
Cyclization Rings form by intramolecular carbon-carbon bond formation. + OPP E double bond Z double bond
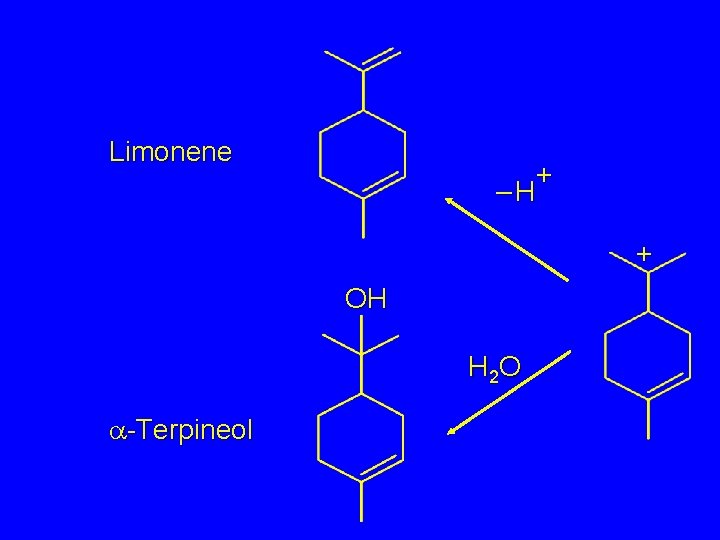
Limonene + –H + OH H 2 O a-Terpineol
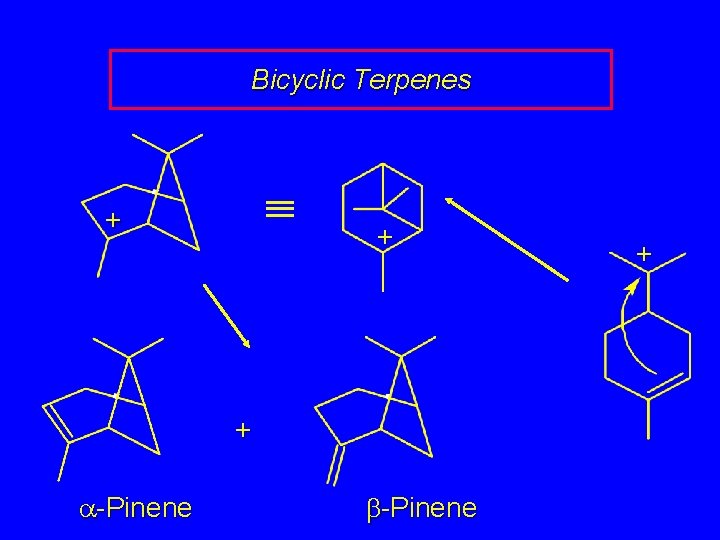
Bicyclic Terpenes + + + a-Pinene b-Pinene +

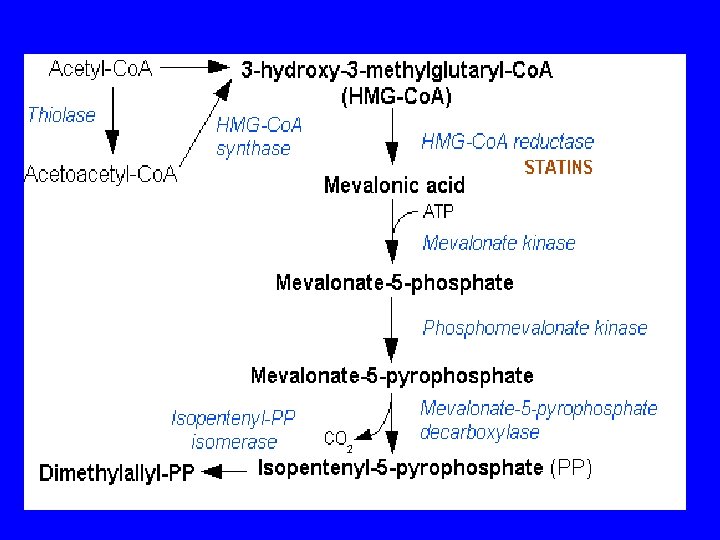
The Pathway from Acetate to Isopentenyl Pyrophosphate

Thiolases, also known as acetyl-coenzyme A Acetyltransferases (ACAT), are enzymes which convert two units of acetyl-Co. A to acetoacetyl Co. A in the mevalonate pathway.
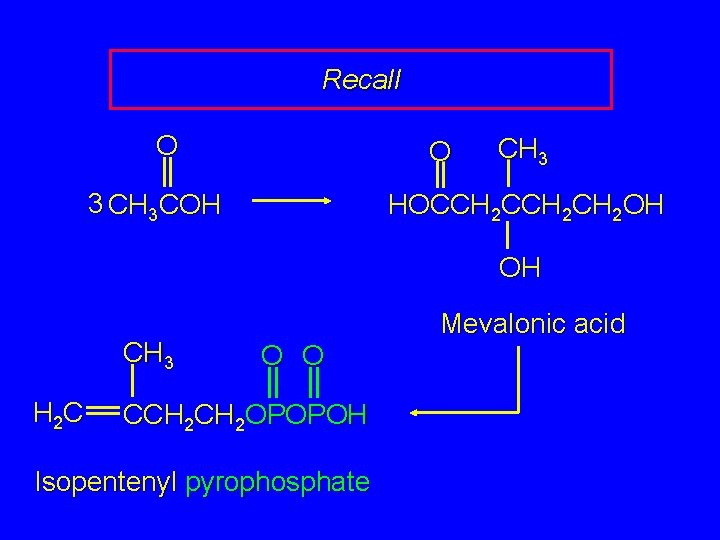
Recall O O 3 CH 3 COH CH 3 HOCCH 2 CH 2 OH OH CH 3 H 2 C Mevalonic acid O O CCH 2 OPOPOH Isopentenyl pyrophosphate
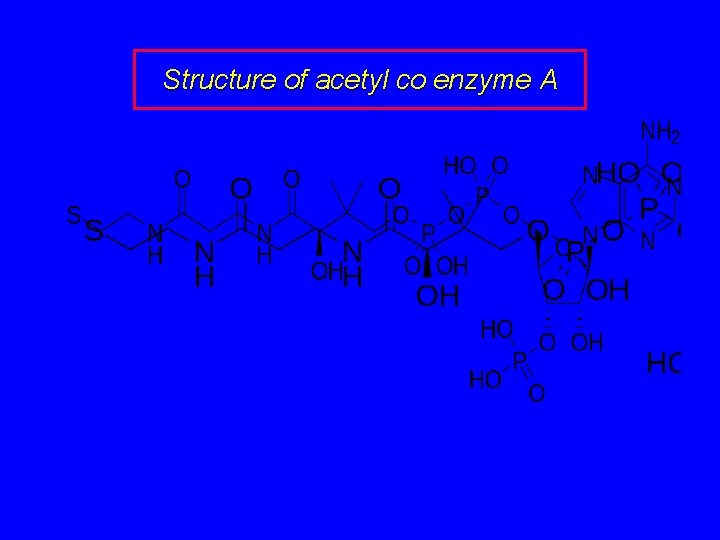
Structure of acetyl co enzyme A
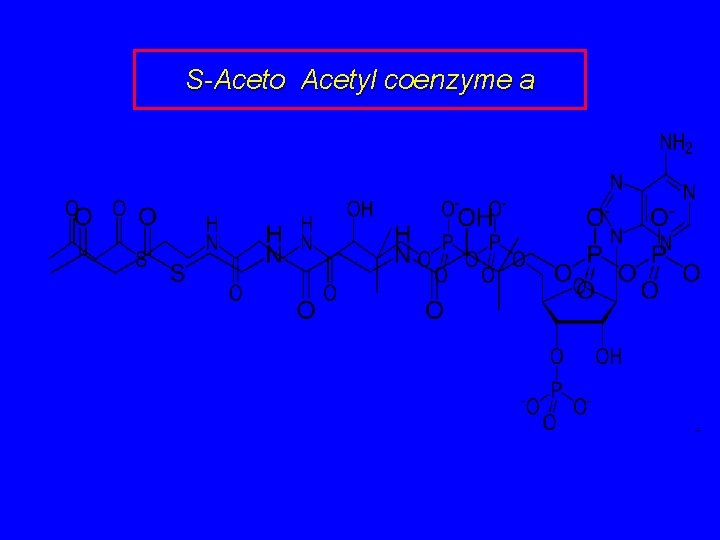
S-Aceto Acetyl coenzyme a

Biosynthesis of Mevalonic Acid In a sequence analogous to the early steps of fatty acid biosynthesis, acetyl coenzyme A is converted to S-acetoacetyl coenzyme A. O O CH 3 CCH 2 CSCo. A S-Acetoacetyl coenzyme A

Biosynthesis of Mevalonic Acid O O O CH 3 CCH 2 CSCo. A + CH 3 CSCo. A In the next step, S-acetoacetyl coenzyme A reacts with acetyl coenzyme A. Nucleophilic addition of acetyl coenzyme A (probably via its enol) to the ketone carbonyl of S-acetoacetyl coenzyme A occurs.
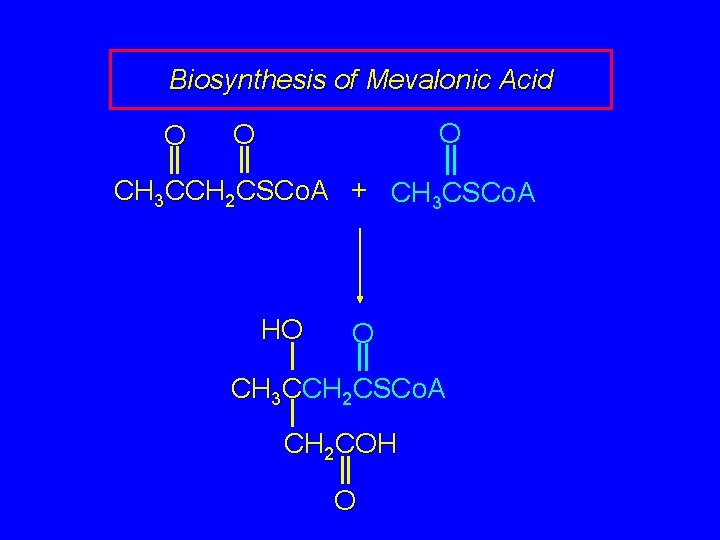
Biosynthesis of Mevalonic Acid O O O CH 3 CCH 2 CSCo. A + CH 3 CSCo. A HO O CH 3 CCH 2 CSCo. A CH 2 COH O
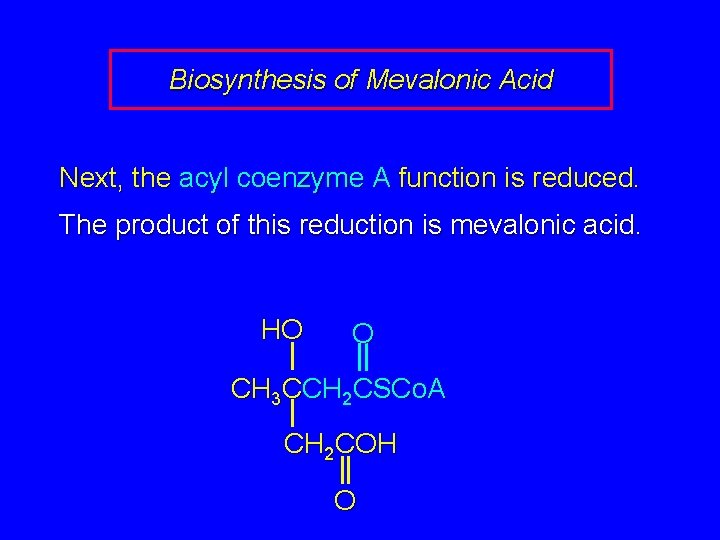
Biosynthesis of Mevalonic Acid Next, the acyl coenzyme A function is reduced. The product of this reduction is mevalonic acid. HO O CH 3 CCH 2 CSCo. A CH 2 COH O
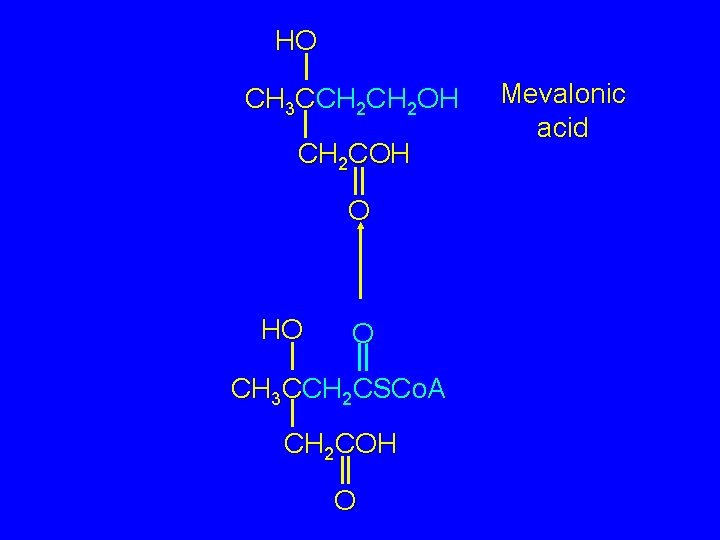
HO CH 3 CCH 2 OH CH 2 COH O HO O CH 3 CCH 2 CSCo. A CH 2 COH O Mevalonic acid
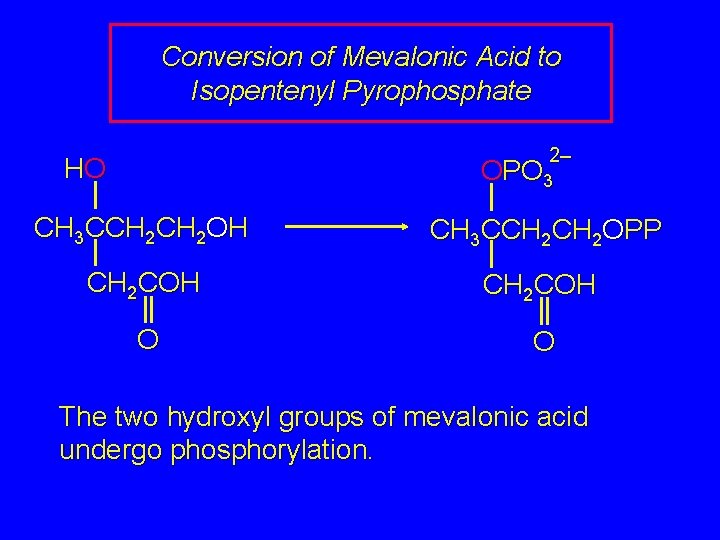
Conversion of Mevalonic Acid to Isopentenyl Pyrophosphate 2– HO OPO 3 CH 3 CCH 2 OH CH 3 CCH 2 OPP CH 2 COH O O The two hydroxyl groups of mevalonic acid undergo phosphorylation.
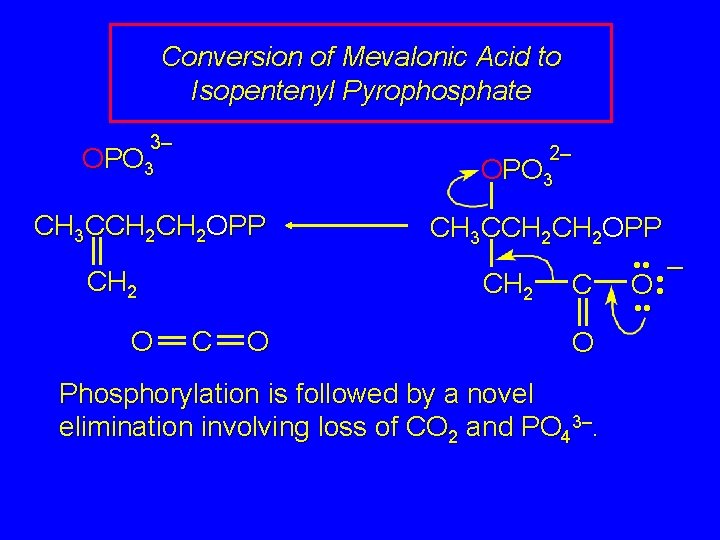
Conversion of Mevalonic Acid to Isopentenyl Pyrophosphate 3– 2– OPO 3 CH 3 CCH 2 OPP CH 2 O CH 3 CCH 2 OPP CH 2 C O Phosphorylation is followed by a novel elimination involving loss of CO 2 and PO 43–. • • – O • •

Conversion of Mevalonic Acid to Isopentenyl Pyrophosphate CH 3 CCH 2 OPP CH 2 The product of this elimination is isopentenyl pyrophosphate.
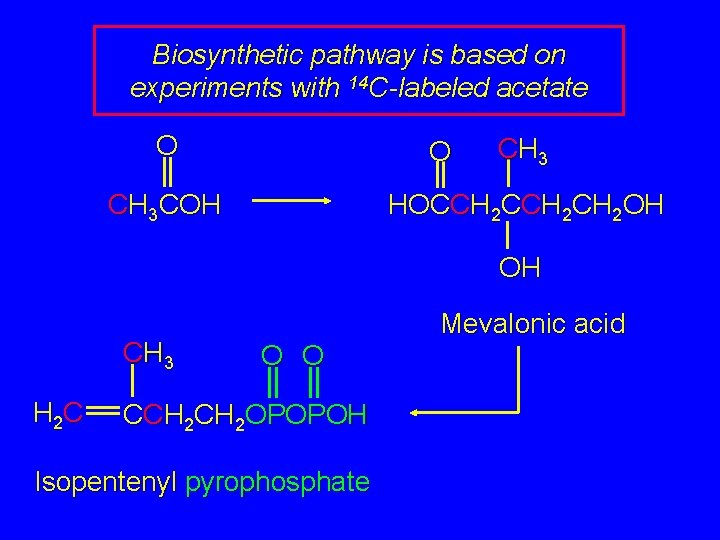
Biosynthetic pathway is based on experiments with 14 C-labeled acetate O O CH 3 COH CH 3 HOCCH 2 CH 2 OH OH CH 3 H 2 C Mevalonic acid O O CCH 2 OPOPOH Isopentenyl pyrophosphate
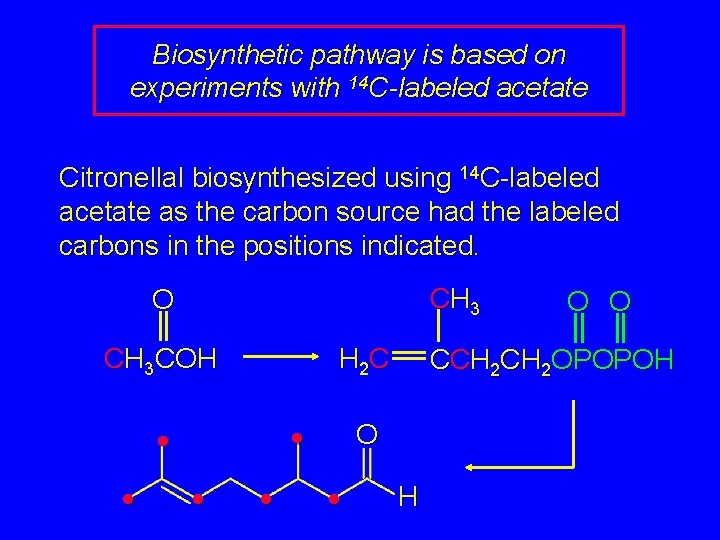
Biosynthetic pathway is based on experiments with 14 C-labeled acetate Citronellal biosynthesized using 14 C-labeled acetate as the carbon source had the labeled carbons in the positions indicated. CH 3 O CH 3 COH H 2 C • • • CCH 2 OPOPOH O • O O H
- Slides: 41