Biosimilars Snapshot of Humira Biosimilars in Pipeline UPDATED
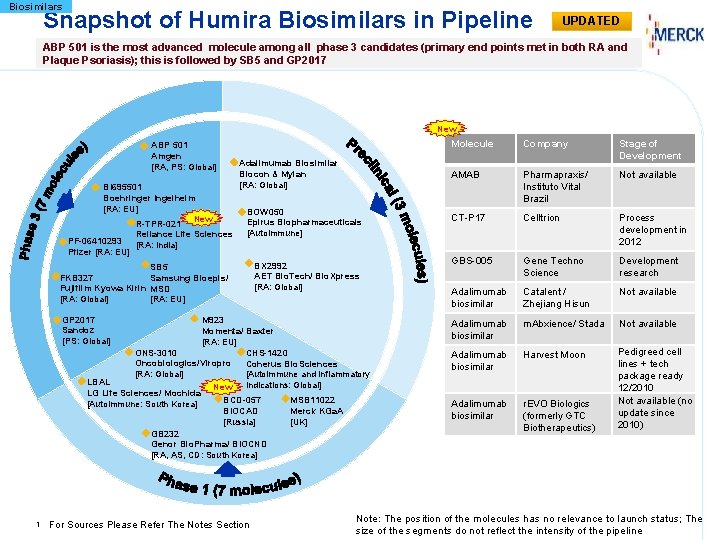
Biosimilars Snapshot of Humira Biosimilars in Pipeline UPDATED ABP 501 is the most advanced molecule among all phase 3 candidates (primary end points met in both RA and Plaque Psoriasis); this is followed by SB 5 and GP 2017 New ABP 501 Amgen [RA, PS: Global] BI 695501 Boehringer Ingelheim [RA: EU] New R-TPR-021 Reliance Life Sciences PF-06410293 [RA: India] Pfizer [RA: EU] Adalimumab Biosimilar Biocon & Mylan [RA: Global] BOW 050 Epirus Biopharmaceuticals [Autoimmune] SB 5 FKB 327 Samsung Bioepis/ Fujifilm Kyowa Kirin MSD [RA: Global] [RA: EU] GP 2017 Sandoz [PS: Global] BX 2992 AET Bio. Tech/ Bio. Xpress [RA: Global] M 923 Momenta/ Baxter [RA: EU] ONS-3010 CHS-1420 Oncobiologics/Viropro Coherus Bio. Sciences [RA: Global] [Autoimmune and inflammatory LBAL indications: Global] New LG Life Sciences/ Mochida BCD-057 MSB 11022 [Autoimmune: South Korea] BIOCAD Merck KGa. A [Russia] [UK] GB 232 Genor Bio. Pharma/ BIOCND [RA, AS, CD: South Korea] 1 For Sources Please Refer The Notes Section Molecule Company Stage of Development AMAB Pharmapraxis/ Instituto Vital Brazil Not available CT-P 17 Celltrion Process development in 2012 GBS-005 Gene Techno Science Development research Adalimumab biosimilar Catalent / Zhejiang Hisun Not available Adalimumab biosimilar m. Abxience/ Stada Not available Adalimumab biosimilar Harvest Moon Adalimumab biosimilar r. EVO Biologics (formerly GTC Biotherapeutics) Pedigreed cell lines + tech package ready 12/2010 Not available (no update since 2010) Note: The position of the molecules has no relevance to launch status; The size of the segments do not reflect the intensity of the pipeline
- Slides: 1