Biosimilar medicines By Abbas Orabi What is a

Biosimilar medicines By Abbas Orabi
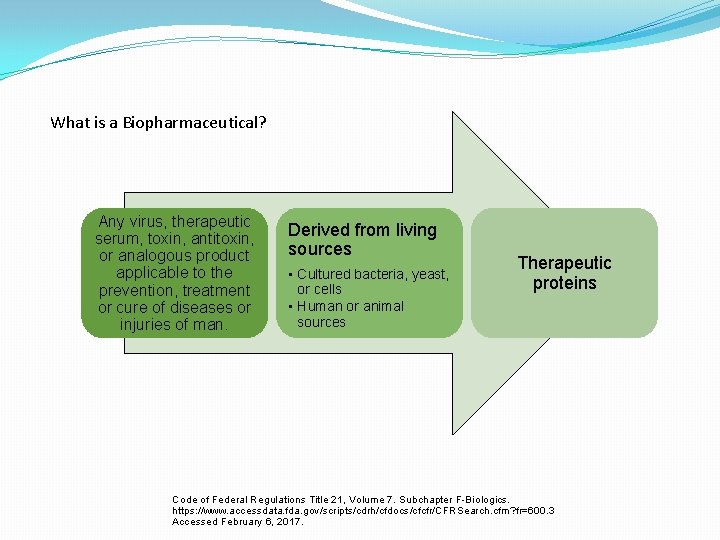
What is a Biopharmaceutical? Any virus, therapeutic serum, toxin, antitoxin, or analogous product applicable to the prevention, treatment or cure of diseases or injuries of man. Derived from living sources • Cultured bacteria, yeast, or cells • Human or animal sources Therapeutic proteins Code of Federal Regulations Title 21, Volume 7. Subchapter F-Biologics. https: //www. accessdata. fda. gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch. cfm? fr=600. 3 Accessed February 6, 2017.
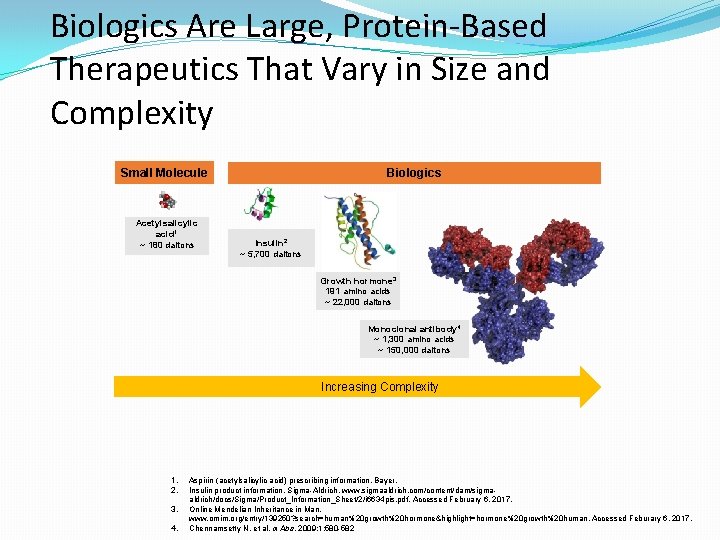
Biologics Are Large, Protein-Based Therapeutics That Vary in Size and Complexity Small Molecule Acetylsalicylic acid 1 ~ 180 daltons Biologics Insulin 2 ~ 5, 700 daltons Growth hormone 3 191 amino acids ~ 22, 000 daltons Monoclonal antibody 4 ~ 1, 300 amino acids ~ 150, 000 daltons Increasing Complexity 1. 2. 3. 4. Aspirin (acetylsalicylic acid) prescribing information, Bayer. Insulin product information, Sigma-Aldrich. www. sigmaaldrich. com/content/dam/sigmaaldrich/docs/Sigma/Product_Information_Sheet/2/i 6634 pis. pdf. Accessed February 6, 2017. Online Mendelian Inheritance in Man. www. omim. org/entry/139250? search=human%20 growth%20 hormone&highlight=hormone%20 growth%20 human. Accessed Feburary 6, 2017. Chennamsetty N, et al. m. Abs. 2009; 1: 580 -582

What Is a Biosimilar? A biosimilar is a “copy” of a commercially available biopharmaceutical (reference product) that no longer is protected by patent which has: • Undergone rigorous analytical and clinical assessment, in comparison to its reference product • AND • Been approved by a regulatory agency according to a specific pathway for biosimilar evaluation A biosimilar is “highly similar” to its reference product in physicochemical characteristics, efficacy, and safety. Dörner T, et al. Ann Rheum Dis. 2016; 75(6): 974 -982.

Biosimilars: definition (EMA) Per EMA “A biosimilar medicinal product is a medicinal product which is similar to a biological medicinal product that has already been authorized (the ‘biological reference medicinal product’). The active substance of a biosimilar medicinal product is similar to the one of the biological reference medicinal product” Note: The name, appearance and packaging of a biosimilar medicinal product may differ from those of the biological reference medicinal product. The product may also contain different inactive ingredients http: //gabionline. net/Biosimilars/General/EMA-definitions-of-generics-and-biosimilars BOEHRINGER INGELHEIM-LILLY ALLIANCE CONFIDENTIAL. FOR TRAINING PURPOSES ONLY.
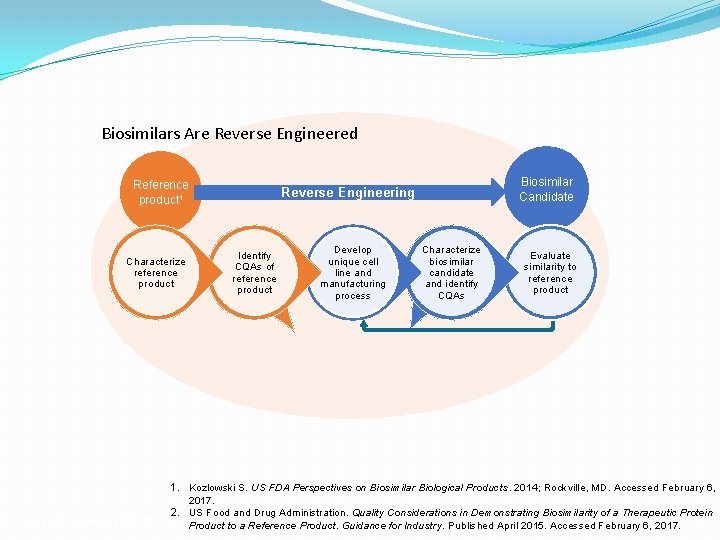
Biosimilars Are Reverse Engineered Reference product 1 Characterize reference product CQAs = critical quality attributes. Biosimilar Candidate Reverse Engineering Identify CQAs of reference product Develop unique cell line and manufacturing process Characterize biosimilar candidate and identify CQAs Evaluate similarity to reference product 1. Kozlowski S. US FDA Perspectives on Biosimilar Biological Products. 2014; Rockville, MD. Accessed February 6, 2017. 2. US Food and Drug Administration. Quality Considerations in Demonstrating Biosimilarity of a Therapeutic Protein Product to a Reference Product. Guidance for Industry. Published April 2015. Accessed February 6, 2017.

Why Is it Important to Understand the Biosimilar Development Process? Known DNA sequence 1 Unique manufacturing 1, 2 Unique biologic 2 Understanding the biosimilar development process allows prescribers to make informed decisions about the potential utility of biosimilars as a treatment option 1. Roger SD. Nephrology (Carlton). 2006; 11: 341 -346. 2. Mellstedt H, et al. Ann Oncol. 2008; 19: 411 -419.
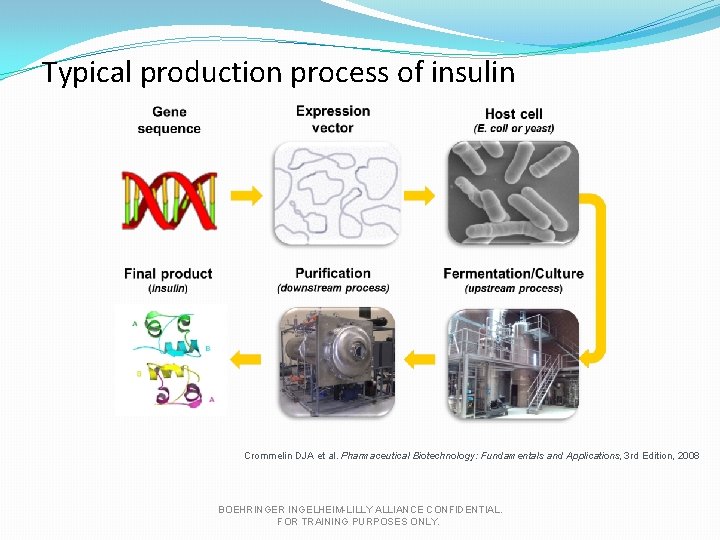
Typical production process of insulin Crommelin DJA et al. Pharmaceutical Biotechnology: Fundamentals and Applications, 3 rd Edition, 2008 BOEHRINGER INGELHEIM-LILLY ALLIANCE CONFIDENTIAL. FOR TRAINING PURPOSES ONLY.

Impact of manufacturing-process differences between biosimilars and their reference biologics 1– 3 Host cell: impacts on the amino acid sequence and post-translational modification such as glycosylation and de-amination Purification process: impacts on the level and type of impurities Differences in formulation, excipients and container closure system: impact on product -quality attributes Storage, shipping and cold chain: impact on product-quality attributes 1. Mellstedt H et al. Ann Oncol 2008; 19: 411– 9; 2. Owens DR et al. Diabetes Technol Ther 2012; 14: 989– 96; 3. Sharma B. Biotechnol Adv 2007; 25: 325– 31 BOEHRINGER INGELHEIM-LILLY ALLIANCE CONFIDENTIAL. FOR TRAINING PURPOSES ONLY.

Generic versus biosimilar • Copies of small-molecule medicinal products derived from chemical manufacturing processes 1 • Identical chemical structures to those of already marketed products 1 • Similar versions of biological medicinal products derived from living cells / systems using biotechnological manufacturing processes 1 • Amino acid sequence should be identical to that of the reference product 2 • Differences in biotechnological manufacturing processes between companies mean that biosimilar products cannot be described as identical 1, 3 1. Sekhon BS, Saluja V. Biosimilars 2011; 1: 1– 11; 2. http: //www. ema. europa. eu/docs/en_GB/document_library/Scientific_guideline/2012/05/WC 500127960. pdf; 3. Owens DR et al. Diabetes Technol Ther 2012; 14: 989– 96 BOEHRINGER INGELHEIM-LILLY ALLIANCE CONFIDENTIAL. FOR TRAINING PURPOSES ONLY.
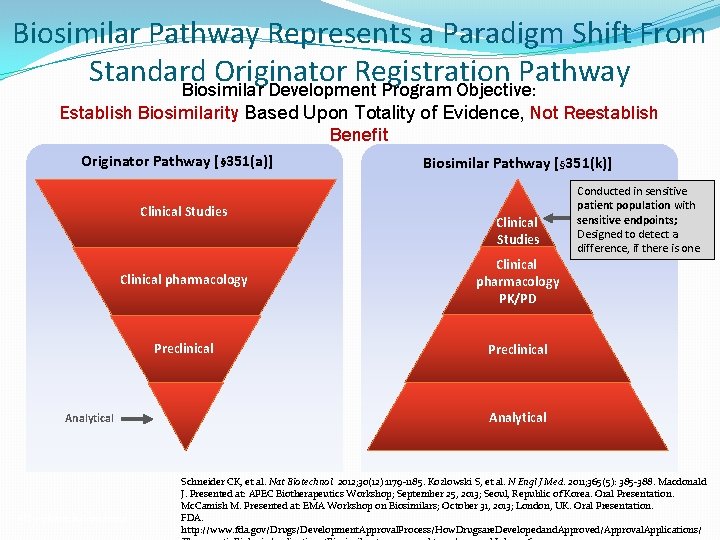
Biosimilar Pathway Represents a Paradigm Shift From Standard Originator Registration Pathway Biosimilar Development Program Objective: Establish Biosimilarity Based Upon Totality of Evidence, Not Reestablish Benefit Originator Pathway [§ 351(a)] Clinical Studies Biosimilar Pathway [§ 351(k)] Clinical Studies Clinical pharmacology PK/PD Preclinical Analytical PD = pharmacodynamics Conducted in sensitive patient population with sensitive endpoints; Designed to detect a difference, if there is one Analytical Schneider CK, et al. Nat Biotechnol. 2012; 30(12): 1179 -1185. Kozlowski S, et al. N Engl J Med. 2011; 365(5): 385 -388. Macdonald J. Presented at: APEC Biotherapeutics Workshop; September 25, 2013; Seoul, Republic of Korea. Oral Presentation. Mc. Camish M. Presented at: EMA Workshop on Biosimilars; October 31, 2013; London, UK. Oral Presentation. FDA. http: //www. fda. gov/Drugs/Development. Approval. Process/How. Drugsare. Developedand. Approved/Approval. Applications/

Biosimilars development programme 1, 2 Analytical studies: demonstrate that the biological product is highly similar to the reference product, notwithstanding minor differences in clinically inactive components Animal studies: for example, toxicology A clinical study or studies (including the assessment of immunogenicity and PK / PD): demonstrate safety, purity and potency in 1 or more appropriate conditions of use for which the reference product is licensed or intended to be used, for which licensure is sought for the biological product CT, clinical trial 1. http: //www. fda. gov/downloads/drugs/guidancecomplianceregulatoryinformation/guidances/ucm 291134. pdf 2. http: //www. ema. europa. eu/docs/en_GB/document_library/Scientific_guideline/2013/06/WC 500144124. pdf BOEHRINGER INGELHEIM-LILLY ALLIANCE CONFIDENTIAL. FOR TRAINING PURPOSES ONLY.

Demonstrating Biosimilarity: General Principles Clinical efficacy and safety of reference biopharmaceutical have already been demonstrated Biosimilar must demonstrate no significant difference from its reference product • Robust analytical, toxicologic, PK/PD, and immunogenicity studies in comparison to reference product • Smaller comparative effectiveness clinical trial(s) conducted in patients in a disease for which the reference product is licensed • No need to demonstrate efficacy in all indications No differences in safety or efficacy are expected between an approved biosimilar and its reference product PK = pharmacokinetics PD = pharmacodynamics http: //www. fda. gov/downloads/Drugs. Guidance. Compliance. Regulatory. Information/Guidances/UCM 291128. pdf. Accessed February 6, 2017.

Extensive Analytical Characterization Is Used Attributes related to the amino acid sequence and all post-translational modifications, including glycans Integrity of the secondary, tertiary, and quaternary structure Primary structure Higher order structure Biological and functional activities, including receptor binding and immunochemical properties Biological function Product-related substances and impurities Quantitative levels of product variants and their identities General properties and excipients Properties of the finished drug product, including strength and formulation CQA = critical quality attribute Process-related impurities Impurities from host cells and downstream process Receptor binding and immunochemical properties Kinetics and thermodynamics of binding, related to functional activity Stability Degradation profiles denoting stability US Food and Drug Administration. Quality Considerations in Demonstrating Biosimilarity of a Therapeutic Protein Product to a Reference Product. Guidance for Industry. April 2015.
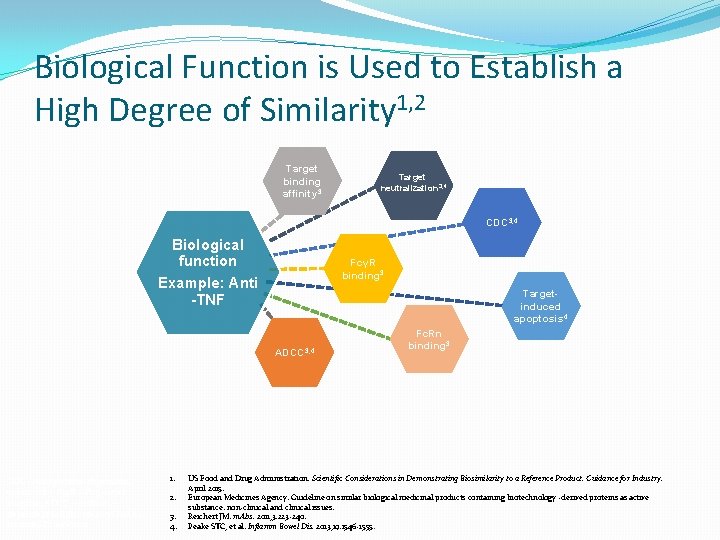
Biological Function is Used to Establish a High Degree of Similarity 1, 2 Target binding affinity 3 Target neutralization 3, 4 CDC 3, 4 Biological function Example: Anti -TNF FcγR binding 3 Targetinduced apoptosis 4 ADCC 3, 4 CDC = complement-dependent cytotoxicity; Fc R = Fc gamma receptor; ADCC = antibodydependent cellular toxicity; Fc. Rn = neonatal Fc receptor 1. 2. 3. 4. Fc. Rn binding 3 US Food and Drug Administration. Scientific Considerations in Demonstrating Biosimilarity to a Reference Product. Guidance for Industry. April 2015. European Medicines Agency. Guideline on similar biological medicinal products containing biotechnology -derived proteins as active substance: non-clinical and clinical issues. Reichert JM. m. Abs. 2011; 3: 223 -240. Peake STC, et al. Inflamm Bowel Dis. 2013; 19: 1546 -1555.
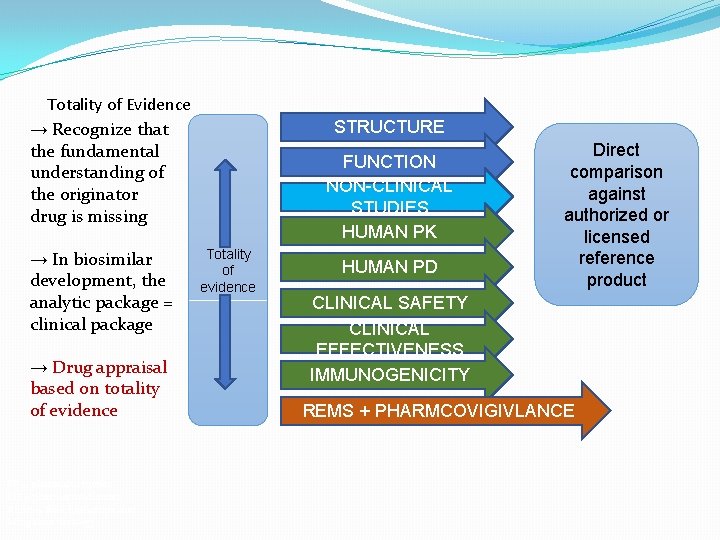
Totality of Evidence → Recognize that the fundamental understanding of the originator drug is missing → In biosimilar development, the analytic package = clinical package → Drug appraisal based on totality of evidence PK = pharmacokinetics PD = pharmacodynamics REMS = Risk Evaluation and Mitigation Strategy STRUCTURE FUNCTION NON-CLINICAL STUDIES HUMAN PK Totality of evidence HUMAN PD Direct comparison against authorized or licensed reference product CLINICAL SAFETY CLINICAL EFFECTIVENESS IMMUNOGENICITY REMS + PHARMCOVIGIVLANCE

Why Biosimilars Biologic therapeutics in multiple fields have revolutionized the treatment for autoimmune, autoinflammatory, and cancer Biosimilars are expensive: drugs for rheumatoid arthritis cost $50, 000/year 1 Costs have increased progressively: cost of TNFi drugs have doubled since their introduction Biosimilar TNFi in Norway in 2014 led to nearly a 60% annual saving Estimates of cost saving: >$66 billion USD over the next decade (4% of total biologics)3 1. TNFi, tumor necrosis factoralpha inhibitor http: //www. fda. gov/downloads/advisorycommittees/committeesmeetingmaterials/drugsadvisorycommitteef orpharmaceuticalscienceandclinicalpharmacology/ucm 315764. pdf 2. Dörner T, et al. Ann Rheum Dis. 2016; 75(6): 974 -982. 3. Mulcahy AW, et al. The Cost Savings Potential of Biosimilar Drugs in the United States. RAND Corporation.
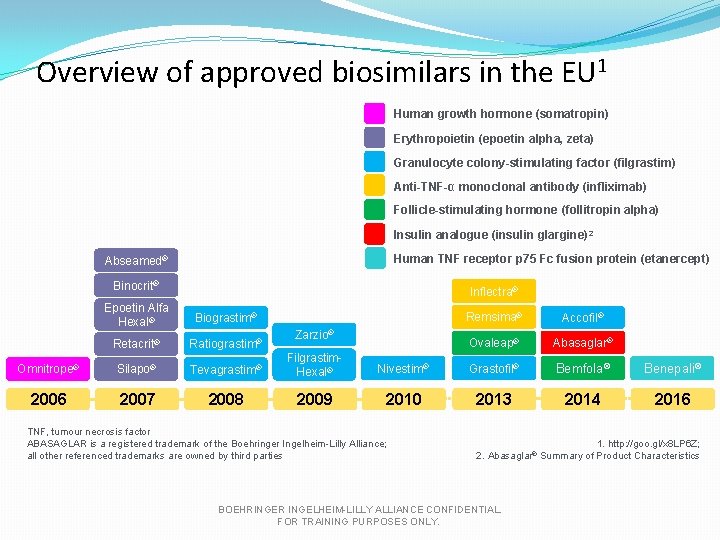
Overview of approved biosimilars in the EU 1 Human growth hormone (somatropin) Erythropoietin (epoetin alpha, zeta) Granulocyte colony-stimulating factor (filgrastim) Anti-TNF-α monoclonal antibody (infliximab) Follicle-stimulating hormone (follitropin alpha) Insulin analogue (insulin glargine) 2 Human TNF receptor p 75 Fc fusion protein (etanercept) Abseamed® Binocrit® Inflectra® Epoetin Alfa Hexal® Biograstim® Retacrit® Ratiograstim® Omnitrope® Silapo® Tevagrastim® Filgrastim. Hexal® 2006 2007 2008 2009 Remsima® Accofil® Ovaleap® Abasaglar® Nivestim® Grastofil® Bemfola® Benepali® 2010 2013 2014 2016 Zarzio® TNF, tumour necrosis factor ABASAGLAR is a registered trademark of the Boehringer Ingelheim-Lilly Alliance; all other referenced trademarks are owned by third parties 1. http: //goo. gl/x 8 LP 6 Z; 2. Abasaglar® Summary of Product Characteristics BOEHRINGER INGELHEIM-LILLY ALLIANCE CONFIDENTIAL. FOR TRAINING PURPOSES ONLY.
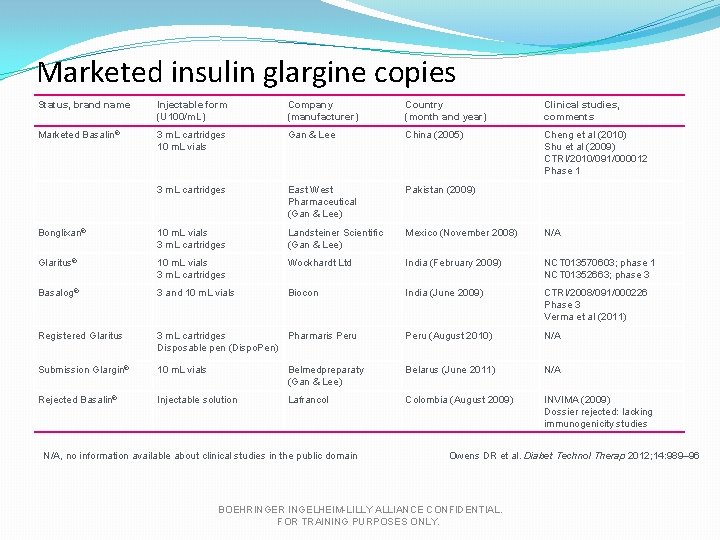
Marketed insulin glargine copies Status, brand name Injectable form (U 100/m. L) Company (manufacturer) Country (month and year) Clinical studies, comments Marketed Basalin® 3 m. L cartridges 10 m. L vials Gan & Lee China (2005) Cheng et al (2010) Shu et al (2009) CTRI/2010/091/000012 Phase 1 3 m. L cartridges East West Pharmaceutical (Gan & Lee) Pakistan (2009) Bonglixan® 10 m. L vials 3 m. L cartridges Landsteiner Scientific (Gan & Lee) Mexico (November 2008) N/A Glaritus® 10 m. L vials 3 m. L cartridges Wockhardt Ltd India (February 2009) NCT 013570603; phase 1 NCT 01352663; phase 3 Basalog® 3 and 10 m. L vials Biocon India (June 2009) CTRI/2008/091/000226 Phase 3 Verma et al (2011) Registered Glaritus 3 m. L cartridges Pharmaris Peru Disposable pen (Dispo. Pen) Peru (August 2010) N/A Submission Glargin® 10 m. L vials Belmedpreparaty (Gan & Lee) Belarus (June 2011) N/A Rejected Basalin® Injectable solution Lafrancol Colombia (August 2009) INVIMA (2009) Dossier rejected: lacking immunogenicity studies N/A, no information available about clinical studies in the public domain Owens DR et al. Diabet Technol Therap 2012; 14: 989– 96 BOEHRINGER INGELHEIM-LILLY ALLIANCE CONFIDENTIAL. FOR TRAINING PURPOSES ONLY.
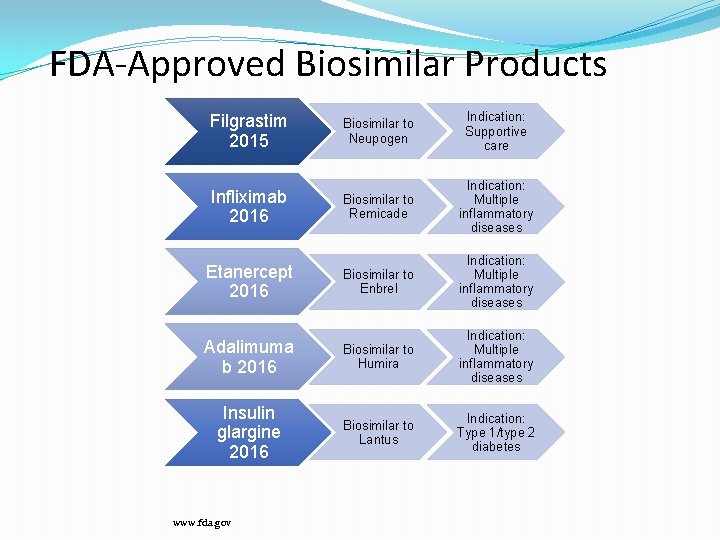
FDA-Approved Biosimilar Products Filgrastim 2015 Biosimilar to Neupogen Indication: Supportive care Infliximab 2016 Biosimilar to Remicade Indication: Multiple inflammatory diseases Etanercept 2016 Biosimilar to Enbrel Indication: Multiple inflammatory diseases Adalimuma b 2016 Biosimilar to Humira Indication: Multiple inflammatory diseases Insulin glargine 2016 Biosimilar to Lantus Indication: Type 1/type 2 diabetes www. fda. gov
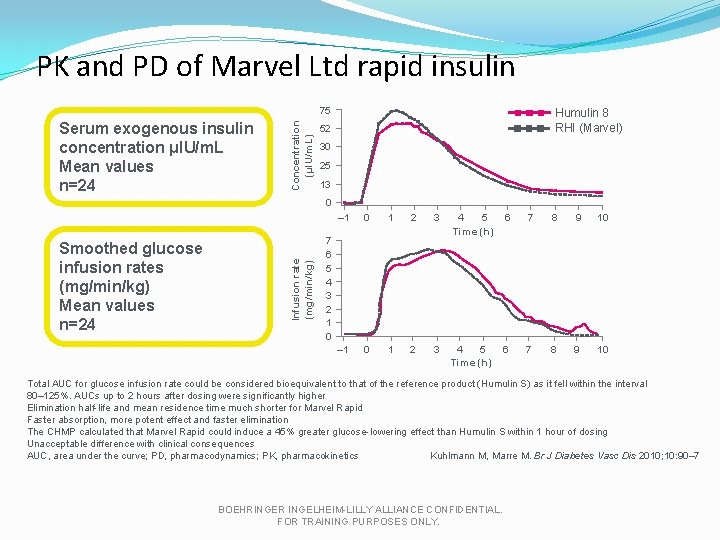
PK and PD of Marvel Ltd rapid insulin Serum exogenous insulin concentration μIU/m. L Mean values n=24 Concentration (µl. U/m. L) 75 Humulin 8 RHI (Marvel) 52 30 25 13 Smoothed glucose infusion rates (mg/min/kg) Mean values n=24 Infusion rate (mg/min/kg) 0 – 1 0 1 2 3 4 5 Time (h) 6 7 8 9 10 7 6 5 4 3 2 1 0 Total AUC for glucose infusion rate could be considered bioequivalent to that of the reference product (Humulin S) as it fell within the interval 80– 125%. AUCs up to 2 hours after dosing were significantly higher Elimination half-life and mean residence time much shorter for Marvel Rapid Faster absorption, more potent effect and faster elimination The CHMP calculated that Marvel Rapid could induce a 45% greater glucose-lowering effect than Humulin S within 1 hour of dosing Unacceptable difference with clinical consequences AUC, area under the curve; PD, pharmacodynamics; PK, pharmacokinetics Kuhlmann M, Marre M. Br J Diabetes Vasc Dis 2010; 10: 90– 7 BOEHRINGER INGELHEIM-LILLY ALLIANCE CONFIDENTIAL. FOR TRAINING PURPOSES ONLY.

Marvel Ltd submission in 2007 Application for marketing authorisation for 3 different formulations of recombinant human insulin: A soluble rapid-acting human insulin (rapid) A long-acting isophane insulin product (long) A 30: 70 mixture of these 2 products (mix) The development programme consisted of studies carried out in 24 healthy volunteers and 1 phase 3 study in 526 patients with T 1 DM or T 2 DM; patients received either the Marvel insulins or the Humulin insulin comparators for up to 12 months The CHMP raised a number of concerns including the quality of the products, sparse information on development, manufacture and control as well as design of the clamp studies, and adequacy of clinical data and risk management plans None of the 3 products sufficiently demonstrated biosimilarity to a properly chosen reference product In January 2008, the EMA announced that Marvel Life. Sciences had withdrawn its applications for all 3 of its insulin formulations CHMP, Committee for Medicinal Products for Human Use; EMA, European Medicines Agency; T 1 DM, type 1 diabetes mellitus; T 2 DM, type 2 diabetes mellitus 1. Kuhlmann M, Marre M. Br J Diabetes Vasc Dis 2010; 10: 90– 7; 2. http: //goo. gl/8 PVM 1 q BOEHRINGER INGELHEIM-LILLY ALLIANCE CONFIDENTIAL. FOR TRAINING PURPOSES ONLY.
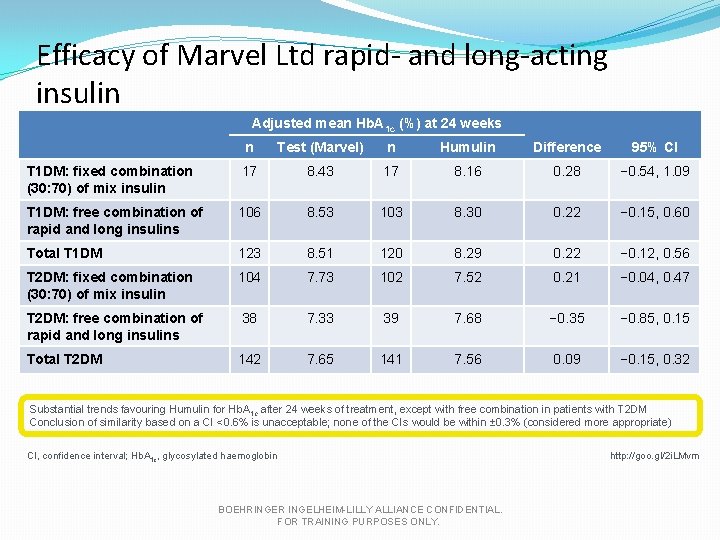
Efficacy of Marvel Ltd rapid- and long-acting insulin Adjusted mean Hb. A 1 c (%) at 24 weeks n Test (Marvel) n Humulin Difference 95% CI T 1 DM: fixed combination (30: 70) of mix insulin 17 8. 43 17 8. 16 0. 28 − 0. 54, 1. 09 T 1 DM: free combination of rapid and long insulins 106 8. 53 103 8. 30 0. 22 − 0. 15, 0. 60 Total T 1 DM 123 8. 51 120 8. 29 0. 22 − 0. 12, 0. 56 T 2 DM: fixed combination (30: 70) of mix insulin 104 7. 73 102 7. 52 0. 21 − 0. 04, 0. 47 T 2 DM: free combination of rapid and long insulins 38 7. 33 39 7. 68 − 0. 35 − 0. 85, 0. 15 Total T 2 DM 142 7. 65 141 7. 56 0. 09 − 0. 15, 0. 32 Substantial trends favouring Humulin for Hb. A 1 c after 24 weeks of treatment, except with free combination in patients with T 2 DM Conclusion of similarity based on a CI <0. 6% is unacceptable; none of the CIs would be within ± 0. 3% (considered more appropriate) CI, confidence interval; Hb. A 1 c, glycosylated haemoglobin BOEHRINGER INGELHEIM-LILLY ALLIANCE CONFIDENTIAL. FOR TRAINING PURPOSES ONLY. http: //goo. gl/2 i. LMvm

Marvel Ltd submission in 2011 • Second application for marketing authorisation for 3 different formulations of recombinant human insulin – A soluble rapid-acting human insulin (rapid; Solumarv) – A long-acting isophane insulin product (long; Isomarv) – A 30: 70 mixture of these 2 products (mix; Combimarv) • The CHMP again raised a number of concerns related to manufacturing processes, demonstration of sufficient similarity with the reference products, reliability of study data, and critical and major Good Clinical Practice compliance findings during study site inspections • None of the 3 products sufficiently demonstrated biosimilarity to the reference products: Humulin S, Humulin I or Humulin M 3 • In November 2012, the EMA announced that Marvel Life. Sciences had withdrawn its applications for all 3 of its insulin formulations 1. http: //goo. gl/A 6 g 27 I; 2. http: //goo. gl/r 6 MUj. X; 3. http: //goo. gl/dmw 6 v 9 BOEHRINGER INGELHEIM-LILLY ALLIANCE CONFIDENTIAL. FOR TRAINING PURPOSES ONLY.

Marvel Ltd submission in 2015 • Third application for marketing authorisation for the soluble rapid-acting recombinant human insulin (rapid; Solumarv) • Marvel Ltd presented results from: – Studies in healthy individuals, designed to show that the biological activity of Solumarv is similar to that of the reference product, Humulin S – Two additional studies in patients with T 1 DM and T 2 DM, which compared the efficacy and safety of Solumarv and Humulin S • The CHMPs main concerns were: – That the manufacturing process was not sufficiently defined to be confident that the Solumarv used in the clinical studies was representative of batches intended for the market – That the quality was not comparable with that of the reference product, Humulin S http: //goo. gl/Lo. UJk 5 BOEHRINGER INGELHEIM-LILLY ALLIANCE CONFIDENTIAL. FOR TRAINING PURPOSES ONLY.

Marketed ‘biosimilar’ recombinant human insulins in India and Poland Brand name Company (manufacturer) Clinical studies (phase 1, 2 and 3) Gensulin® Bioton, Poland N/A Biosulin® MJ Biopharm Pvt. Ltd. , India (Marvel Life. Sciences) N/A Wosulin® Wockhardt Ltd, India NCT 00596063 (1, completed) NCT 00752180 (1, completed) NCT 00719108 (1, completed) NCT 01308437 (3, recruiting) Insugen® R Biocon, India N/A, no information available about clinical studies in the public domain CTRI/2010/091/000627 (3, not yet recruiting) Owens DR et al. Diabet Technol Therap 2012; 14: 989– 96 BOEHRINGER INGELHEIM-LILLY ALLIANCE CONFIDENTIAL. FOR TRAINING PURPOSES ONLY.

Clinical Impact of Biosimilars Lower cost and increase access to medications due to prescriber behavior change Change our paradigm of considering study designs and relevant endpoints (extrapolation) Cause dispensers and prescribers to consider substituting interchangeable products Require providers to be aware of pharmacovigilance issues, such as naming and immunogenicity concerns Dörner T, et al. Ann Rheum Dis. 2016; 75(6): 974 -982

Biosimilars: conclusions (1 of 2) • Biosimilars – Are therapeutic protein molecules that should have an identical amino acid sequence, unless shown to be meaningless, to that of a previously marketed product, with no clinically meaningful difference in safety or efficacy 1, 2 – Are not generics; they are similar to but not the same as reference products 3 – Provide valuable options that create choice for prescribers and patients 4 • Biosimilar manufacturing quality matters – Manufacturing processes that may influence the quality and/or immunogenicity of biological products include protein production, purification, formulation, and storage and handling 5 1. http: //www. fda. gov/downloads/Drugs/Guidance. Compliance. Regulatory. Information/Guidances/UCM 291128. pdf; 2. http: //www. ema. europa. eu/docs/en_GB/document_library/Scientific_guideline/2014/06/WC 500167838. pdf; 3. Declerck PJ. Ga. BI J 2012; 1: 13– 6; 4. Wilkins AR et al. J Diabetes Sci Technol 2014; 8: 23– 5; 5. Sharma B. Biotechnol Adv 2007; 25: 325– 31 BOEHRINGER INGELHEIM-LILLY ALLIANCE CONFIDENTIAL. FOR TRAINING PURPOSES ONLY.

Biosimilars: conclusions (2 of 2) • To comply with regulatory guidelines, in comparison with the reference product, a biosimilar medicine must demonstrate: 1, 2 – In vitro and in vivo non-clinical characteristics similar to those of the reference product – Similar PK and PD within pre-defined regulatory acceptance limits – No clinically meaningful difference in efficacy (eg, based on non-inferiority studies) – No clinically meaningful differences in drug-related adverse events and immunogenicity • Currently, a number of biosimilar products are available in the EU 3, 4; in the USA, the first biosimilar, filgrastim-sndz, was approved in March 20155 1. http: //www. fda. gov/downloads/drugs/guidancecomplianceregulatoryinformation/guidances/ucm 291134. pdf; 2. http: //www. ema. europa. eu/docs/en_GB/document_library/Scientific_guideline/2014/04/WC 500165988. pdf; 3. http: //goo. gl/x 8 LP 6 Z; 4. Abasaglar® Summary of Product Characteristics; 5. http: //www. biosimilarnews. com/fda-approves-first-biosimilar-zarxio-filgrastim-sndz-from-sandoz BOEHRINGER INGELHEIM-LILLY ALLIANCE CONFIDENTIAL. FOR TRAINING PURPOSES ONLY.

Thank you
- Slides: 30