Biosensors A focus on peroxidasemodified electrodes and their

Biosensors A focus on peroxidase-modified electrodes and their practical applications by Ivo Frébort

Biosensor - an analytical device that exploits a biocatalytic reaction Consists of: biocatalyst (enzyme, cells, tissue) transducer (converts the biological or biochemical signal into a quantifiable electrical or optical signal)

First biosensor - Clark (1962): glucose sensor with glucose oxidase and oxygen electrode Glucose + O 2 Gluconic acid + H 2 O 2 Oxygen electrode (1956) working electrode: Pt cathode (-0. 6 V) reference electrode: Ag/Ag. Cl electrodes separated from measured solution with a gas permeable mebrane Leland C. Clark, Jr. with the first enzyme electrode
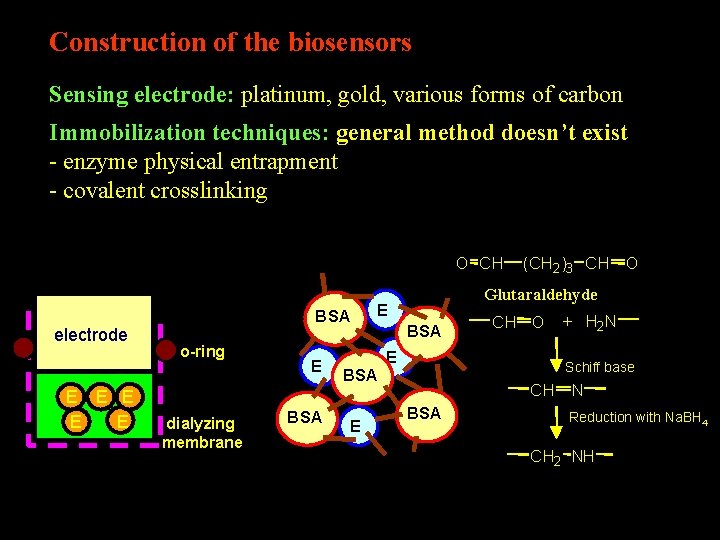
Construction of the biosensors Sensing electrode: platinum, gold, various forms of carbon Immobilization techniques: general method doesn’t exist - enzyme physical entrapment - covalent crosslinking O CH electrode E E E BSA o-ring dialyzing membrane E BSA E Glutaraldehyde E BSA (CH 2)3 CH O BSA E CH O + H 2 N Schiff base CH N BSA Reduction with Na. BH 4 CH 2 NH
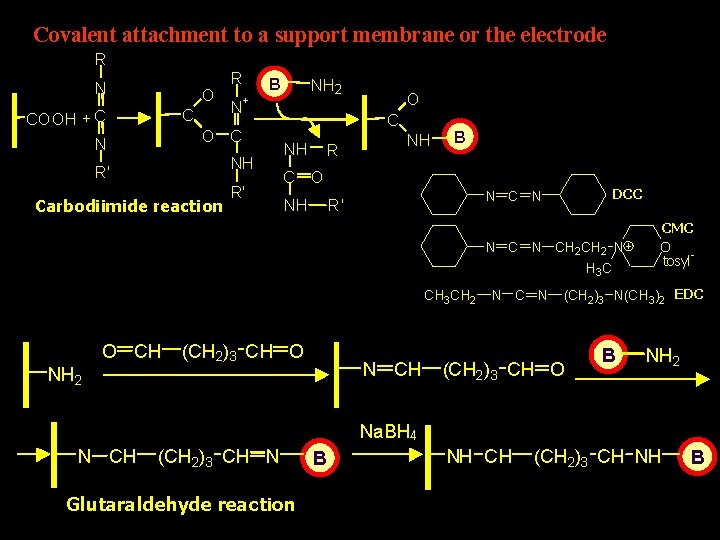
Covalent attachment to a support membrane or the electrode R N O C COOH + C N R N B + O C NH R' Carbodiimide reaction NH 2 R' O C NH R NH B C O DCC N R' NH N CH 2 CH 2 N H 3 C CH 3 CH 2 O CH NH 2 (CH 2)3 CH O N CH N CMC O tosyl- (CH 2)3 N(CH 3)2 EDC (CH 2 )3 CH O B NH 2 Na. BH 4 N CH (CH 2)3 CH N Glutaraldehyde reaction B NH CH (CH 2)3 CH NH B
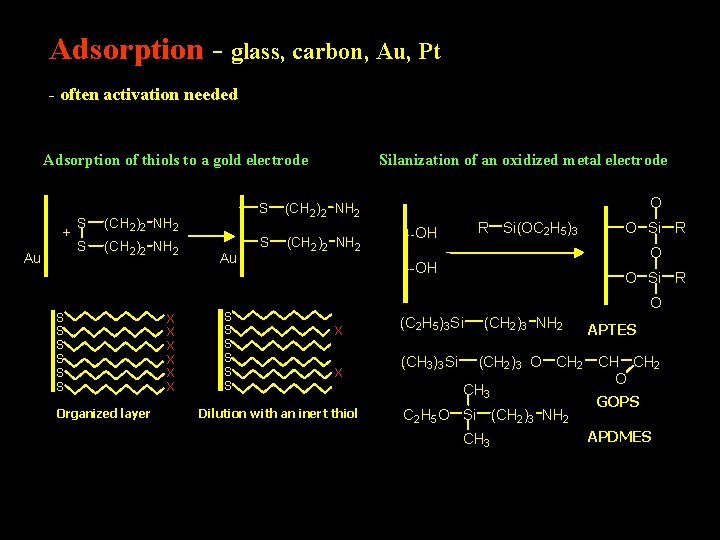
Adsorption - glass, carbon, Au, Pt - often activation needed Adsorption of thiols to a gold electrode + Au S S (CH 2)2 NH 2 S S S Organized layer X X X Au S S S Silanization of an oxidized metal electrode S (CH 2)2 NH 2 O R Si(OC 2 H 5)3 --OH O Si R O X X Dilution with an inert thiol (C 2 H 5)3 Si (CH 3)3 Si (CH 2)3 NH 2 (CH 2)3 O CH 2 CH 3 C 2 H 5 O Si (CH 2)3 NH 2 CH 3 APTES CH CH 2 O GOPS APDMES
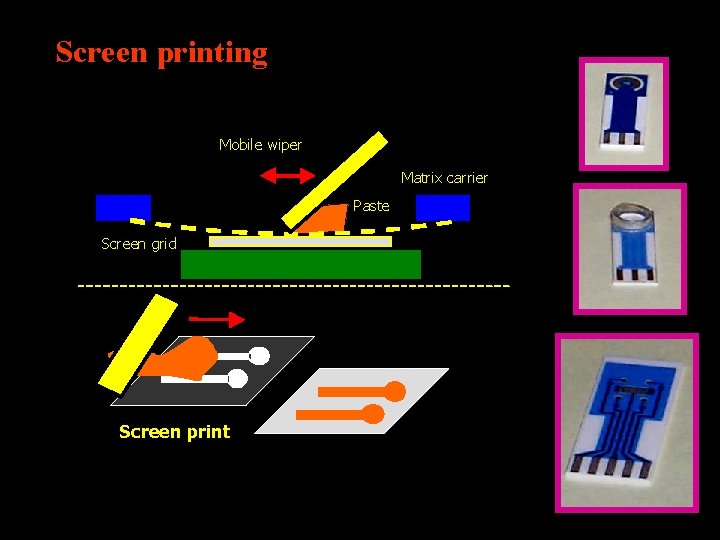
Screen printing Mobile wiper Matrix carrier Paste Screen grid Screen print
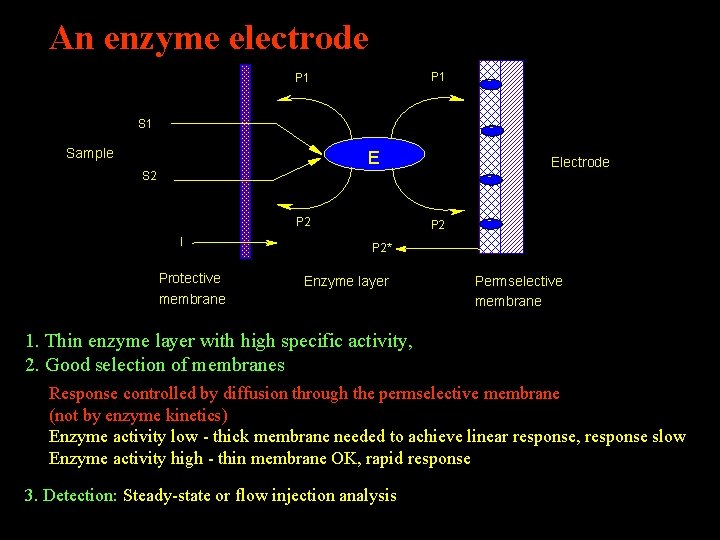
An enzyme electrode P 1 S 1 - - Sample E S 2 - P 2 I Protective membrane P 2 Electrode - P 2* Enzyme layer Permselective membrane 1. Thin enzyme layer with high specific activity, 2. Good selection of membranes Response controlled by diffusion through the permselective membrane (not by enzyme kinetics) Enzyme activity low - thick membrane needed to achieve linear response, response slow Enzyme activity high - thin membrane OK, rapid response 3. Detection: Steady-state or flow injection analysis
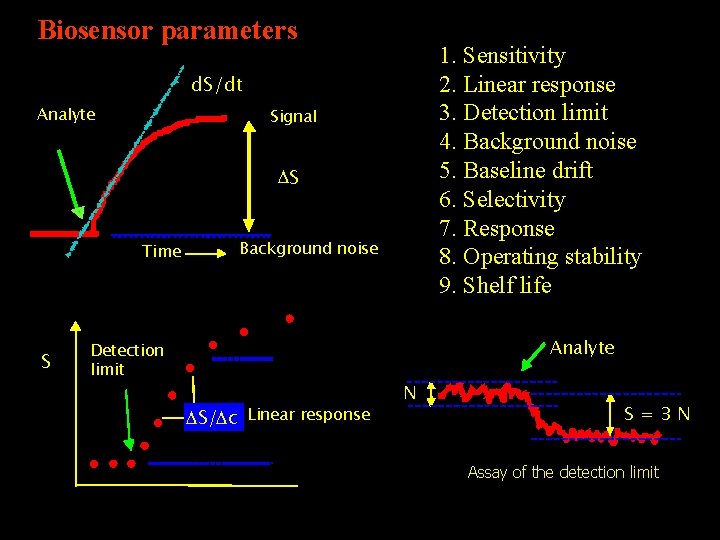
Biosensor parameters 1. Sensitivity 2. Linear response 3. Detection limit 4. Background noise 5. Baseline drift 6. Selectivity 7. Response 8. Operating stability 9. Shelf life d. S/dt Analyte Signal DS Background noise Time S Analyte Detection limit DS/Dc Linear response N S=3 N Assay of the detection limit c
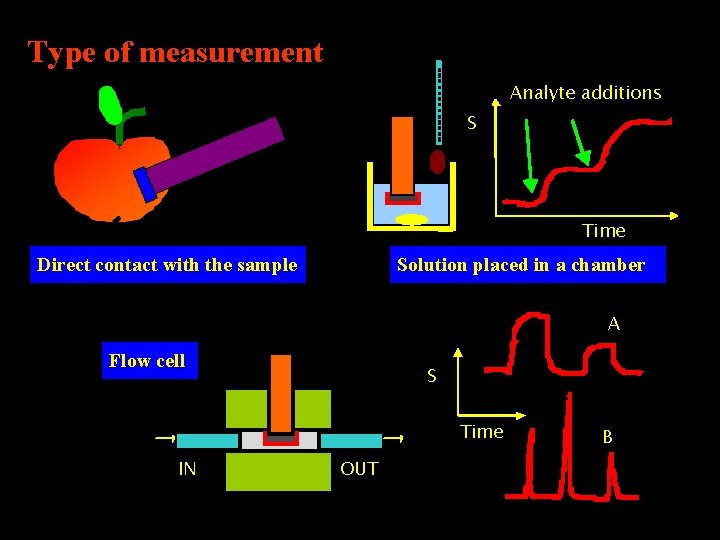
Type of measurement Analyte additions S Time Direct contact with the sample Solution placed in a chamber A Flow cell S Time IN OUT B

First generation biosensors - response to the substrates in solution Glucose + O 2 Gluconic acid + H 2 O 2 1. Reduction of oxygen with a Clark type electrode at -0. 6 V (vs SCE) 2. Oxidation of hydrogen peroxide at a Pt electrode at +0. 7 V 3. Measuring of p. H change Examples of hydrogen peroxide measurning biosensor Analyte Enzyme Reaction Alcohol oxidase Ethanol + O 2 Acetaldehyde + H 2 O 2 D-Glucose oxidase β-D-Glucose + O 2 Gluconic acid + H 2 O 2 Lactose Galactose oxidase Lactose + O 2 Galactose dialdehyde der. + H 2 O 2 L-Lactate oxidase L-Lactate + O 2 Pyruvate + H 2 O 2 Starch Amyloglucosidase Glucose oxidase Starch + H 2 O β-D-Glucose + O 2 Gluconic acid + H 2 O 2 Sucrose Invertase Mutarotase Glucose oxidase Sucrose + H 2 O α-D-Glucose + β-D-Fructose α-D-Glucose β-D-Glucose + O 2 Gluconic acid + H 2 O 2

Types of transducers used in biosensors Type Amperometric Detectable species O 2, H 2 O 2, I 2, NADH Ion-selective electrode H+, Na+, Cl- Field effect transistors H+, Na+, Cl- Gas sensing electrode CO 2, NH 3 Photomultiplier Light emission ATP/Luciferase/Luciferin, H 2 O 2/Peroxidase/Luminol, etc. Thermistor Heat of reaction

Second generation biosensors - mediated electron transfer between enzyme and electrode - can be easily miniaturized blood glucose measuring system in situ Third generation biosensors - direct electron transfer between enzyme and electrode Cell-based biosensors - cheaper than purified enzymes, Nocardia erythropolis cells immobilised in polyacrylamide or agar (cholesterol oxidase) Cholesterol + O 2 Cholest-4 -en-3 -one + H 2 O 2 Enzyme immunosensors - many types, based on ELISA techniques - often use chemiluminiscence or bioluminiscence human chorionic gonadotropin - pregnancy
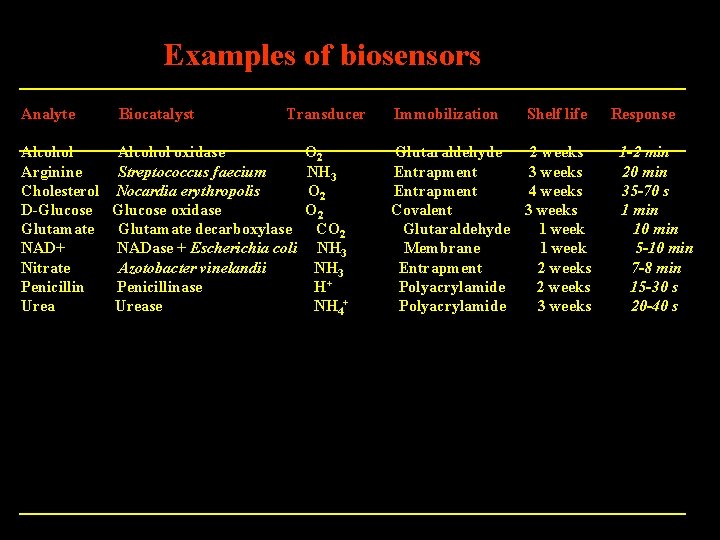
Examples of biosensors Analyte Biocatalyst Transducer Alcohol oxidase Arginine Streptococcus faecium Cholesterol Nocardia erythropolis D-Glucose oxidase Glutamate decarboxylase NAD+ NADase + Escherichia coli Nitrate Azotobacter vinelandii Penicillinase Urease O 2 NH 3 O 2 CO 2 NH 3 H+ NH 4+ Immobilization Shelf life Glutaraldehyde 2 weeks Entrapment 3 weeks Entrapment 4 weeks Covalent 3 weeks Glutaraldehyde 1 week Membrane 1 week Entrapment 2 weeks Polyacrylamide 3 weeks Response 1 -2 min 20 min 35 -70 s 1 min 10 min 5 -10 min 7 -8 min 15 -30 s 20 -40 s
Personal glucose meter for diabetics (Medisense Britain, Ltd. )

Automated affinity systems Biacore 2000 (Biacore) www. bioacore. com IAsys (Affinity Sensors) www. affinity-sensors. com KI 1 (Bio. Tu. L) www. biotul. com IBIS II (Xan. Tec) www. xantec. com
Peroxidase-based electrodes PEROXIDASE (EC 1. 1. 7) Protein of 35 -45 k. Da, prosthetic group - heme, Mn 2+ Convenient sources: horse radish root, soybean, tobacco leaves, various fungi Ruiz-Duenas, F. J. , Martinez, M. J. , Martinez, A. T. : Peroxidase from the ligninolytic fungus Pleurotus eryngii. Mol Microbiol 31 pp. 223 (1999)

The catalytic cycle of peroxidase Native peroxidase + H 2 O 2 (Fe 3+) Compound-I + AH 2 (Fe 4+=O, Por+) Compound-II + AH 2 (Fe 4+=O) Compound-I + H 2 O (Fe 4+=O, Por+) Compound-II + AH* (Fe 4+=O) Native peroxidase+ AH* + H 2 O (Fe 3+)

Applications of peroxidase-based electrodes 1. Detection of hydrogen peroxide in aqueous solutions industry, environmental protection, clinical control photochemical smog, pathological processes in lungs, etc. 2. Detection of organic peroxides in water and organic solutions free radical injury, oxidative stress, autooxidation of unsaturated lipids 3. Detection of compounds based on peroxidase inhibition CN-, F-, hydroxylamine 4. Detection of aromatic amines and phenolic compounds environmental control: chlorophenols in water 5. Immunosensors based on peroxidase electrodes peroxidase conjugates with antibody, H 2 O 2 -producing enzyme conjugates 6. Detection of analytes based on peroxidase/oxidase-coupled reactions glucose, ethanol, lactate, choline, xanthine, cholesterol, bilirubin, glutamate

Electrode designs A. Surface modified electrodes Electrode material: graphite, glassy carbon, gold, Sn. O 2 Coupling: carbodiimide, glutaraldehyde, adsorption B. Polymer-based electrodes Crosslinking with Os(bpy)23+/2+ redox polymer, electropolymerized polypyrrole, o-phenylethylamine C. Bulk modified composite electrodes Graphite-silicone oil paste, paraffin oil paste, epoxy composite D. Tissue-modified carbon paste electrodes Asparagus tissue, tobacco callus tissue, horseradish root, kohlrabi skin
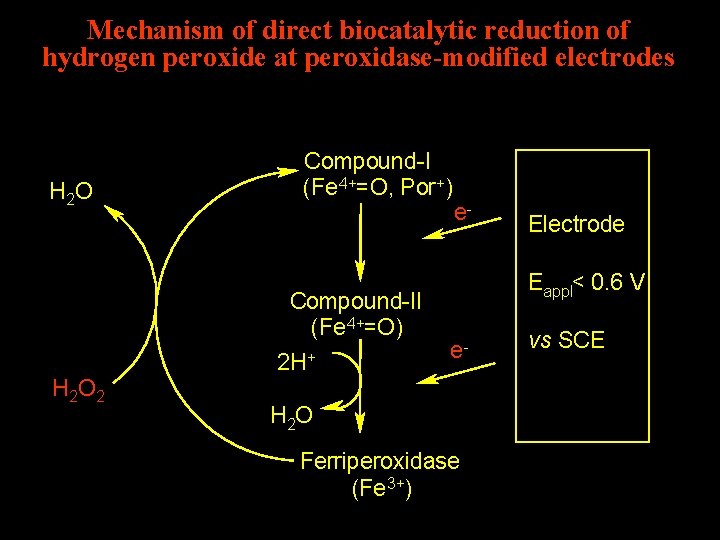
Mechanism of direct biocatalytic reduction of hydrogen peroxide at peroxidase-modified electrodes H 2 O Compound-I (Fe 4+=O, Por+) Compound-II (Fe 4+=O) 2 H+ H 2 O 2 e- Electrode Eappl< 0. 6 V e- H 2 O Ferriperoxidase (Fe 3+) vs SCE
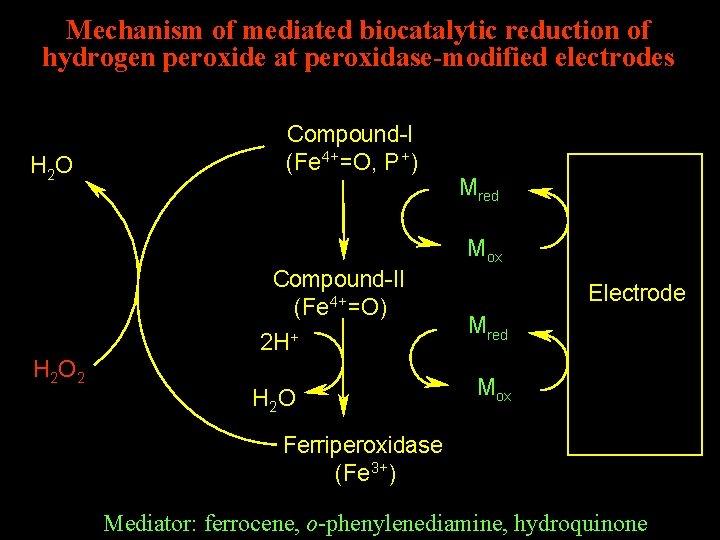
Mechanism of mediated biocatalytic reduction of hydrogen peroxide at peroxidase-modified electrodes H 2 O Compound-I (Fe 4+=O, P +) Mred Mox Compound-II (Fe 4+=O) 2 H + H 2 O 2 H 2 O Electrode Mred Mox Ferriperoxidase (Fe 3+) Mediator: ferrocene, o-phenylenediamine, hydroquinone
The mediators Ferrocene o-Phenylenediamine Hydroquinone

Detailed look at a practical example. . .

Copper amine oxidase-based electrodes for the assay of biogenic amines u. Monitoring the biomarkers of food freshness: histamine, putrescine, cadaverine u. Currently used methods: chromatographic techniques - they often require sample pre-treatment steps and skilled operators; the relatively long analysis time and high costs make these methods not suitable for routine use u. Aim of the work: design and construction of the amperometric biosensors for monitoring of biomarkers u. Two biosensor designs: monoenzymatic and bienzymatic, using both the direct and mediated electron transfer pathways u. Biological recognition element: copper amine oxidase (EC 1. 4. 3. 6) u. Mediator: poly(1 -vinylimidazole) complexed with [Os(4, 4'dimethylbipyridine)2 Cl]+/2+ (PVI 13 -dme. Os) u. Assay system: The biosensors were used in a flow-injection
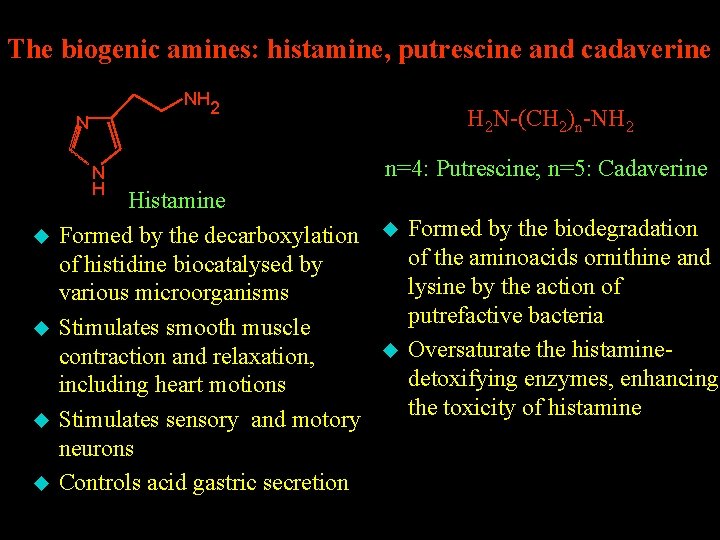
The biogenic amines: histamine, putrescine and cadaverine NH 2 N N H u u Histamine Formed by the decarboxylation of histidine biocatalysed by various microorganisms Stimulates smooth muscle contraction and relaxation, including heart motions Stimulates sensory and motory neurons Controls acid gastric secretion H 2 N-(CH 2)n-NH 2 n=4: Putrescine; n=5: Cadaverine u u Formed by the biodegradation of the aminoacids ornithine and lysine by the action of putrefactive bacteria Oversaturate the histaminedetoxifying enzymes, enhancing the toxicity of histamine

Copper amine oxidase (AO) Redox active polymer (PVI 13 -dme. Os) u u u Biological sources: bacteria, fungi, plants, animals Biological functions: involved in cell growth, proliferation and differentiation Cofactors: Topa quinone (TPQ) Copper Catalyzed reaction: R-CH 2 -NH 2 + H 2 O + O 2 R-CHO + NH 3 + H 2 O 2

Flow-injection system used

Working mechanism for monoenzymatic electrodes

Working mechanism for bienzymatic electrodes

Biosensors characteristics Native enzyme Km - putrescine 0. 2 m. M, histamine 0. 35 m. M
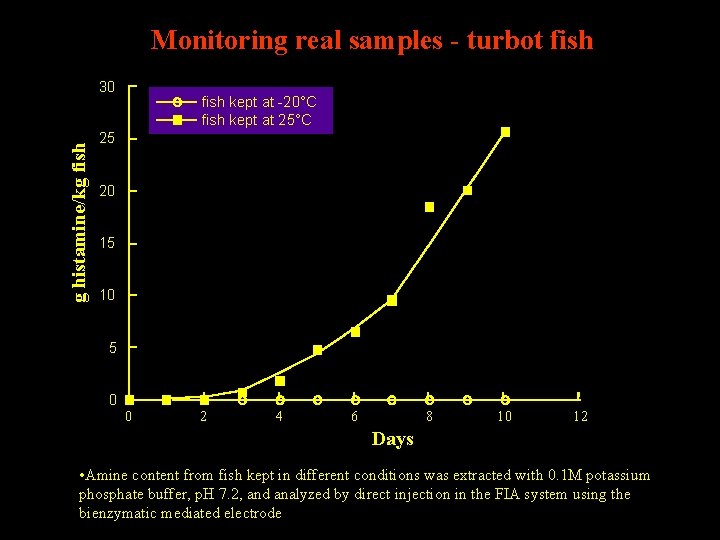
Monitoring real samples - turbot fish g histamine/kg fish 30 fish kept at -20°C fish kept at 25°C 25 20 15 10 5 0 0 2 4 6 8 10 12 Days • Amine content from fish kept in different conditions was extracted with 0. 1 M potassium phosphate buffer, p. H 7. 2, and analyzed by direct injection in the FIA system using the bienzymatic mediated electrode
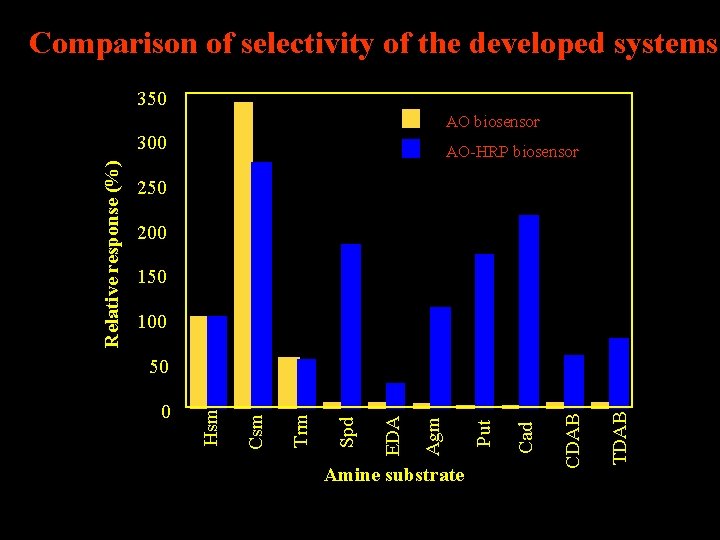
Comparison of selectivity of the developed systems 350 AO biosensor AO-HRP biosensor 250 200 150 100 TDAB Cad CDAB Amine substrate Put Agm EDA Spd Trm 0 Csm 50 Hsm Relative response (%) 300

Further oxidation of the histamine reaction product

Putrescine and cadaverine form cyclic products - cannot be further oxidized !!!

Conclusions 1. Combination of the monoenyzmatic (AO) and bienzymatic (AOHRP) electrode can be used for selective detection of histamine and diamines (putrescine and cadaverine). 2. The biosensors were tested for detection of fish product poisoning by putrefactive amines. 3. The monoenzymatic electrode (AO) is the first example of DET with copper amine oxidase, which can proceed anaerobically. 4. With histamine as an analyte, both DET and further oxidation of the product aldehyde contribute to the biosensor response.
- Slides: 36