Biosafety levels n n n A biosafety level

Biosafety levels

n n n A biosafety level is a set of biocontainment precautions required to isolate dangerous biological agents in an enclosed laboratory facility. The levels of containment range from the lowest biosafety level 1 (BSL-1) to the highest at level 4 (BSL-4). In the United States, the Centers for Disease Control and Prevention (CDC) have specified these levels.

Biosafety Levels n Labs divided into 4 biosafety levels; protective practices increase with each level n Biosafety Level 1 labs - work with least dangerous agents, require fewest precautions n Biosafety Level 4 labs - have strictest methods because dealing with agents that are most dangerous to human health


Barriers n Primary barriers: physical barriers or personal protective equipment between lab worker and pathogen n n Gloves, masks, special breathing apparatuses Secondary barriers: structural aspects of the laboratory that make working environment safer against infection n Sinks for handwashing, special containment areas, special air ventilation patterns

Biosafety Level 1 (BSL-1) n Is suitable for work with well characterized agents which do not cause disease in healthy adults. n Have minimal potential hazard to laboratory personnel and environment.

Agents n n n non-pathogenic E. coli species Bacillus subtilis Saccharomyces cerevisiae transmission electron micrograph of E. coli

Biosafety Level 1 (BSL-1) n Standard practices required: n n n frequent hand washing door that can be kept closed when working; limits on access to the lab space when working; no smoking, eating, drinking, storage of food in laboratory; care to minimize splashes decontamination of work surfaces after every use after any spills;

Standard practices (continued): n n n decontamination of laboratory wastes; use of mechanical pipettes only (no mouth pipetting); "sharps" precautions, including special containers for disposing of needles and other sharp objects; maintenance of insect/rodent control program; use of personal protective equipment (lab coats, latex gloves, eye protection or face shields) Open bench top sink for hand washing

Biosafety Level 2 (BSL-2) n Suitable for work involving agents of moderate hazard to personnel and environment n Generally required for any human-derived blood, body fluids, tissues in which infectious agent may be unknown

Agents Measles virus, Salmonella species, Pathogenic Toxoplasma, Hepatitis A, B, C virus Plasmodium falciparum

Biosafety Level 2 (BSL-2) n Primary hazards: n n accidental needle sticks exposure to eyes and nose (mucous membranes) ingestion of infectious materials Agents do not cause lethal infections, are not transmissible via airborne route n (do not cause infection if tiny droplets become airborne and are inhaled, which might occur if the material were splattered) n Agents are pathogens for which immunization or antibiotic treatment is available n Extreme care should be taken with contaminated needles and sharp lab instruments

Biosafety Level 2 (BSL-2) n Standard practices include BSL-1 plus: n n n policies to restrict access to lab; biohazard warning signs posted outside lab; surveillance of laboratory personnel with appropriate immunizations offered; biosafety manual with definitions of needed waste decontamination or medical surveillance policies; supervisory staff who have experience working with infectious agents and specific training for laboratory personnel in handling these agents

Biosafety Level 2 (BSL-2) n n n Primary barriers: biosafety cabinets or other approved containment devices Personal protective equipment: lab coats, gloves, face protection as needed Protective clothing removed when personnel leave laboratory area Cabinets thoroughly decontaminated daily and monitored for radiation for personal protection Secondary barriers: BSL-1 barriers plus autoclave for glassware

Biosafety Level 2 (BSL-2) n Example of biosafety sign posted outside lab working with infectious agents Lab’s biosafety level Infectious agents under study Contact information for responsible person and 2 emergency contacts

Biosafety Level 3 (BSL-3) n appropriate for work involving microbes which can cause serious and potentially lethal disease through the inhalation route n Agents with potential for respiratory transmission, may cause serious and potentially lethal infection

Agents Mycobacterium tuberculosis, n Chlamydia psittaci, n Francisella tularensis, n Coxiella burnetii n West Nile virus n Yellow fever virus n Venezuelan equine encephalitis virus n

Biosafety Level 3 (BSL-3) n Standard practices include BSL-2 plus: n strictly controlled access to the lab; n specific training for lab personnel in handling potentially lethal agents n decontaminating all waste n changing contaminated protective lab clothing, decontaminating lab clothing before laundering; n institutional policies regarding specimen collection and storage from workers to establish exposure

Standard practices n All laboratory personnel are provided medical surveillance and offered relevant immunizations to reduce the risk of infection n Laboratory personnel must use solid-front protective clothing (i. e. gowns). This cannot be used outside of the laboratory and must be discarded or decontaminated after each use n All procedures involving infectious material must be done within a biological safety cabinet------HEPA filters

Biosafety Level 3 (BSL-3) n Primary barriers: n n n Similar to BSL-2 personal protective equipment Respiratory equipment if risk of infection through inhalation Secondary barriers: n n n All BSL-2 barriers Corridors separated from direct access to lab Access through self-closing double doors

Biosafety Level 3 n Researcher at US Centers for Disease Control, Atlanta, working with influenza virus under biosafety level 3 conditions, with respirator inside a biosafety cabinet.

Biosafety Level 4 (BSL-4) n n Highest level of biosafety precautions Is appropriate for work with agents that could be easily aerosol transmitted within lab Dangerous and exotic agents with high risk of life -threatening disease No available vaccines

Agents n n Marburg virus, Ebola virus, viruses that cause Congo-Crimean hemorrhagic fever, n Lassa fever n n ( transmission electron micrograph of Ebola virus

Biosafety Level 4 (BSL-4) n BSL-4 laboratories are generally set up to be either cabinet laboratories or protective suit laboratories. n In cabinet laboratories, all work must be done within a class III biosafety cabinet all materials within cabinet must be free of sharp edges in order to reduce the risk of damage to the gloves cabinets themselves are required to have smooth edges to allow for easy cleaning n n

n n In a protective suit laboratory, all work must be done in a class II biosafety cabinet by personnel wearing a positive pressure suit. In order to exit the BSL-4 laboratory, personnel must pass through a chemical shower for decontamination, then a room for removing the positive pressure suit, followed by a personal shower.
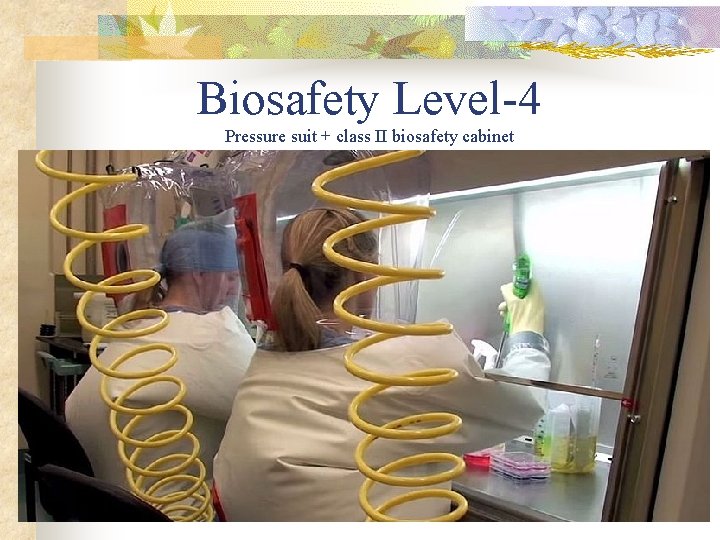
Biosafety Level-4 Pressure suit + class II biosafety cabinet

Biosafety Level-4 (BSL-4)

Class III biosafety cabinet

Standard practices include BSL-3 plus Entry into the BSL-4 laboratory is restricted to trained and authorized individuals n All persons entering and exiting the laboratory must be recorded. n BSL-4 laboratories must be separated from areas that receive unrestricted traffic n Changing clothing before entering and exiting lab (showering upon exiting recommended) n decontaminating of all waste material of lab n

Biosafety Level 4 (BSL-4) n Primary barriers: n n n Biosafety cabinets used at other biosafety levels Full-body, air-supplied, positive pressure personnel suit Secondary barriers: n n All physical barriers at BSL-3 isolated zone or a separate building; dedicated supply and exhaust, vacuum, decontamination systems; a recommended absence of windows (or sealed and resistant to breakage)

Risk Assessment In-Vitro In-Vivo Human Clinical Trial

Addressing Risk Assessments n n n What is the organism? Is it Wild-type, irradiated, or chemically treated? Look at kill data or kill curves. What is the max. concentration, volume, infectious dose? What is the work space like? Aerosolizing procedures? How do they contain their aerosols?


References 1. 2. 3. 4. US Department of Health and Human Services, Centers for Disease Control and Prevention and National Institutes of Health. Biosafety in Microbiological and Biomedical Laboratories. 5 th ed. Washington, DC: US Government Printing Office; 2007. http: //www. cdc. gov/od/ohs/ biosfty/bmbl 5 toc. htm. Accessed February 6, 2008. Clemson University Environmental Health and Safety. Biological Safety [online training]. http: //ehs. clemson. edu/training/biosafety/index. html. Accessed February 6, 2008. Centers for Disease Control and Prevention. Laboratory-Acquired West Nile Virus Infections — United States, 2002. MMWR Morb Mort Wkly Rep. 2002; 51: 1133 -1135. http: //www. cdc. gov/mmwr/preview/ mmwrhtml/mm 5150 a 2. htm. Accessed February 6, 2008. United States Government Accountability Office. High-Containment Biosafety Laboratories: Preliminary Observations on the Oversight of the Proliferation of BSL-3 and BSL-4 Laboratories in the United States. Publication GAO-08 -108 T. http: //www. gao. gov/docsearch/ abstract. php? rptno=GAO-08 -108 T. Published October 4, 2007.

n. Thanks
- Slides: 35