BIOSAFETY BIORISK ASSESSMENT Dr Attya Bhatti Biosafety definition

BIOSAFETY BIORISK ASSESSMENT Dr Attya Bhatti

Biosafety definition • All measures that prevent accidentally exposure at biological agents (micro-organisms and toxins) and infections and their released in environment Biological agent Any microorganism including those which have been genetically modified, cell cultures and endoparasites, which may be able to provoke any infection, allergy or toxicity in humans, animals or plants

Bio-risk definition Combination of the probability of occurrence of harm and the severity of that harm where the source of harm is a biological agent or toxin

Biohazard definition Potential source of harm caused by biological agents or toxins The source of harm may be an unintentional exposure, accidental release or loss, theft, misuse, diversion, unauthorized access or intentional unauthorized release

Biorisk Assessment (adapted from OHSAS 18001: 2007) Process of evaluating the biorisk(s) arising from a biohazard(s), taking into account the adequacy of any existing controls, and deciding whether or not the biorisk(s) is acceptable As defined by Kaplan and Garrick, risk analysis consists of answering three specific questions: • what can happen? • what is the chance that it will happen? • if it happens, what are the consequences? .

Characteristics • Risk assessment is first step to control (reduce or eliminate) the hazards • Assessments can be qualitative, semi-quantitative or quantitative • Risk assessment methodology should be: – identified – implemented – maintained • Provide a guide for the selection of appropriate - biosafety levels needs - microbiological practices - safety equipment - facility safeguards
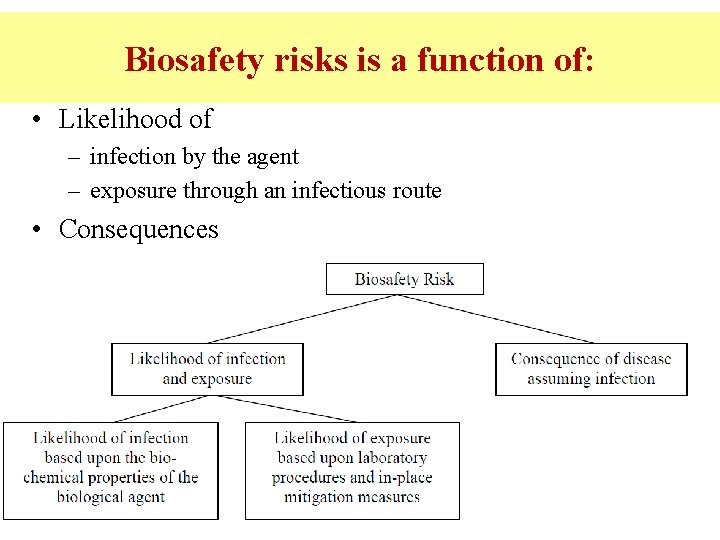
Biosafety risks is a function of: • Likelihood of – infection by the agent – exposure through an infectious route • Consequences

Scenarios • Risk to individuals in the laboratory • Risk to an individual outside the laboratory (the human community) • Risk to animals outside the laboratory (the animal community) • Risks to humans and animals resulting from a secondary exposure

Routes of exposure • droplets or droplet nuclei (upper or lower respiratory tract) • compromised skin or direct injection into the blood stream • the mucosal membranes • the gastrointestinal tract

Consequences of disease in humans assuming infection (1) • Does this agent or one of its by-products cause a carcinogenic or mutagenic reaction in a human host? • Does this agent have toxin or enzyme production which has a negative impact in a healthy human host? • Does this agent suppress a human host’s immune system? (E. g. cause dramatic suppression which renders the host susceptible other infections) • Does this agent have the ability to mutate once in a host or in the natural environment to become infectious through new route or new hosts, or to cause increased consequences? • What is the duration of illness (the average length of time of clinical signs of infection) in a normally healthy human host? • What is the severity of illness (the average severity of illness, ranging from no signs of illness to hospitalize in critical condition) in a normal health human host? • What is the duration of infection (the length of time the host is infected with the organism) in a normal healthy human host?

Consequences of disease in humans assuming infection (2) • Does this disease cause any long-term conditions in a normal healthy human host? • What is the frequency of death in humans caused by this disease in a defined population during a specified interval of time (Mortality Rate)? • What level of national / international reporting is required for outbreaks of this disease? • Do effective diagnostic tests exist for humans? • Do post exposure treatments (including immuno-globulin, vaccines and antimicrobials) exist for humans? • Do preventative measures (vaccines) exist for humans?

Consequences of disease in animals assuming infection • If the agent infects animals, what is the expected morbidity rate to a naive but otherwise healthy animal population? • What level of national or international reporting is required for outbreaks of this disease? • What species of animals can this agent infect? • Do effective diagnostic tests exist for animals? • Do post exposure treatments (including immuno-globulin, vaccines and antimicrobials) exist for animals? • Do preventative measures (vaccines) exist for animals?

The risk assessment should categorize risks • Use of a risk matrix identifying likelihood and consequence categories – e. g. high, moderate and low zones • Acceptability of risk levels should be defined • Inherent risk from the biological agents and toxins – e. g. from risk grouping descriptions, material safety data sheets etc.

Risk assessment CAREFUL judgment biosafety levels ≠ risk group A risk assessment should be conducted in a manner which is standardized and systematic allowing it to be repeatable and comparable. Determine the degree of correlation between an agent’s risk group classification and biosafety level

Classification of infective microorganisms by risk groups • Risk Group 1 (no or very low individual and community risk) A microorganism that is unlikely to cause disease • Risk Group 2 (moderate individual risk, low community risk) A pathogen that can cause human or animal disease but is unlikely to be a serious hazard to laboratory workers, the community, livestock or the environment. – Effective treatment and preventive measures are available and the risk of spread of infection is limited.

Classification of infective microorganisms by risk groups • Risk Group 3 (high individual risk, low community risk) A pathogen that usually causes serious human or animal disease but does not ordinarily spread from one infected individual to another. – Effective treatment and preventive measures are available. • Risk Group 4 (high individual and community risk) A pathogen that usually causes serious human or animal disease and that can be readily transmitted from one individual to another, directly or indirectly. – Effective treatment and preventive measures are not usually available.
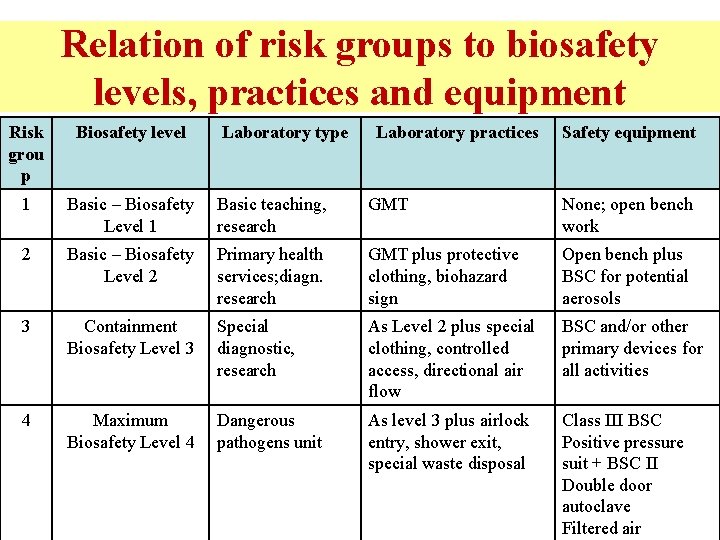
Relation of risk groups to biosafety levels, practices and equipment Risk grou p Biosafety level Laboratory type Laboratory practices Safety equipment 1 Basic – Biosafety Basic teaching, Level 1 research GMT None; open bench work 2 Basic – Biosafety Primary health Level 2 services; diagn. research GMT plus protective clothing, biohazard sign Open bench plus BSC for potential aerosols 3 Containment Biosafety Level 3 Special diagnostic, research As Level 2 plus special clothing, controlled access, directional air flow BSC and/or other primary devices for all activities 4 Maximum Biosafety Level 4 Dangerous pathogens unit As level 3 plus airlock entry, shower exit, special waste disposal Class III BSC Positive pressure suit + BSC II Double door autoclave Filtered air
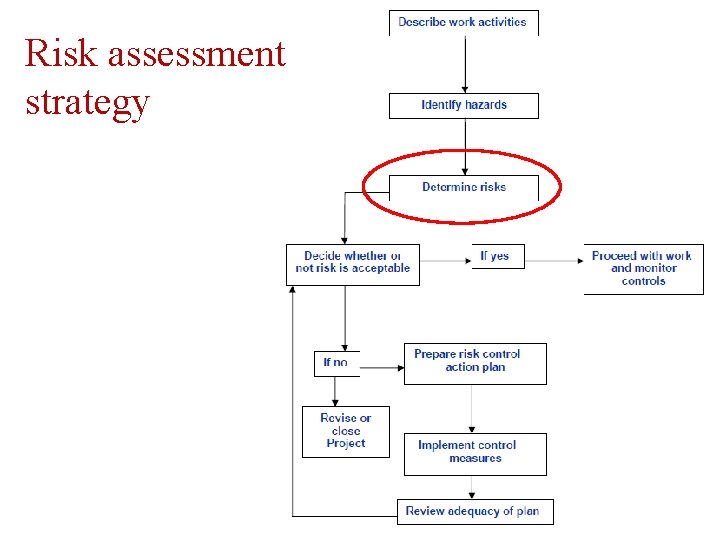
Risk assessment strategy

Biorisk Assessment Sheet

Risk Characterization The description of the nature and magnitude of the human risk, including attendant uncertainty.

Biological agents at work There are three ways in which you might be exposed to biological agents at work: • Exposure as a result of working with biological agents, eg in a microbiology laboratory; • Exposure which does not result from the work itself but is incidental to it, mainly because biological agents are present as contaminants, eg farming, refuse collection, sewage treatment. • Exposure which is not a result of the work that you do, e. g catching flu from a work colleague.

Assessing the Risks Risk: • The likelihood that a specified undesired event will occur due to the realisation of a hazard by, or during work activities or by the products and services created by work activities. • Risk is a measure of future uncertainties in achieving program performance goals and objectives within defined cost, schedule and performance constraints.

The risk assessment must consider : � the biological agent(s) that are/may be present; � what hazard groups they belong to; � what form they are in (for example, do the agents form spores or cysts that are resistant to disinfection? ); � the diseases they may cause (for example, infections, allergies or toxic effects); � how and where they are present and how they are transmitted; � the likelihood of exposure and consequent disease; � people who may be particularly susceptible; for example, children, pregnant women or anyone with low immunity;

Measures needed to control (reduce) exposure a) keeping the number of employees exposed or likely to be exposed to the biological agent as low as possible; b) designing work processes and engineering control measures so as to prevent or minimise the release of biological agents into the place of work; c) displaying the biohazard sign and other relevant warning signs; d) drawing up plans to deal with accidents involving biological agents; e) specifying appropriate decontamination and disinfection procedures; f) instituting means for the safe collection, storage and disposal of contaminated waste, including the use of secure and identifiable containers, after suitable treatment where appropriate; g) making arrangements for the safe handling and transport of biological agents, or materials that may contain such agents, within the workplace; h) specifying procedures for taking, handling, processing and disposing of samples that may contain biological agents;

Measures needed to control (reduce) exposure i) providing collective protection measures and, where exposure cannot be adequately controlled by other means, individual protection measures including, in particular, the supply of appropriate protective clothing or other special clothing; j) where appropriate, making available effective vaccines for those employees who are not already immune to the biological agent to which they are exposed or are liable to be exposed; k) introducing hygiene measures including provision of adequate washing and toilet facilities and the prohibition of eating, drinking, smoking and applying cosmetics in working areas.

Assessing the Risks Quantitative risk assessment (QRA) • Commonly used in the high technology industries • QRA tends to deal with the avoidance of low probability events with serious consequences to the human / animal/ plant and the surrounding environment.

Assessing the Risks Subjective risk assessment • Qualitative risk assessment involves making a formal judgement on the consequence and probability using: Risk = Severity x Likelihood

Assessing the Risks Example: The likely effect of a hazard may for example be rated: 1. Major Death or major injury or illness causing long term disability 2. Serious Injuries or illness causing short-term disability 3. Slight All other injuries or illnesses

Assessing the Risks The likelihood of harm may be rated 1. High Where it is certain that harm will occur 2. Medium Where harm will often occur 3. Low Where harm will seldom occur

Assessing the Risks Risk = Severity of Harm x Likelihood of occurrence

Creating Risk Assessments • Steps – Identify the hazards – Assess the risks (are they high, medium or low? ) – Determine control measures (how do you reduce high or medium to low) – Implement an action plan to reduce the risks – Monitor, maintain and review the plan (e. g. check the plan after a suitable time to see if it is still OK because circumstances may have changed)

Controlling the risks • Eliminating risks: eg by substituting a hazardous biological agent with something less/non-hazardous, eg using a non-toxigenic strain of a biological agent when carrying out laboratory quality control (QC) tests;

Controlling the risks Controlling risks at source: by using engineering controls and giving collective protective measures priority, eg using a microbiological safety cabinet when work could create an infectious aerosol, or using needle safety devices to prevent and control needle stick injuries

Controlling the risks Minimising risks by designing suitable systems of working: eg having an effective hand hygiene policy in place in laboratory or healthcare settings. , includes the use of personal protective clothing and equipment (PPE).

Activity: Point to be look into • Eliminate – do not use the hazardous thing or do the hazardous activity • Substitute – do or use something safer • Reduce – use less of the hazardous thing or moderate the activity • Isolate – keep the hazard away from people • Contain – use a container or screen off the activity • Engineering Controls – use of a cover, screen, brake, restrictor etc • Administrative Controls – e. g. authorised users, mandatory training • Personal Protective Equipment (PPE) – safety glasses, lab coat, gloves, hard hat etc.
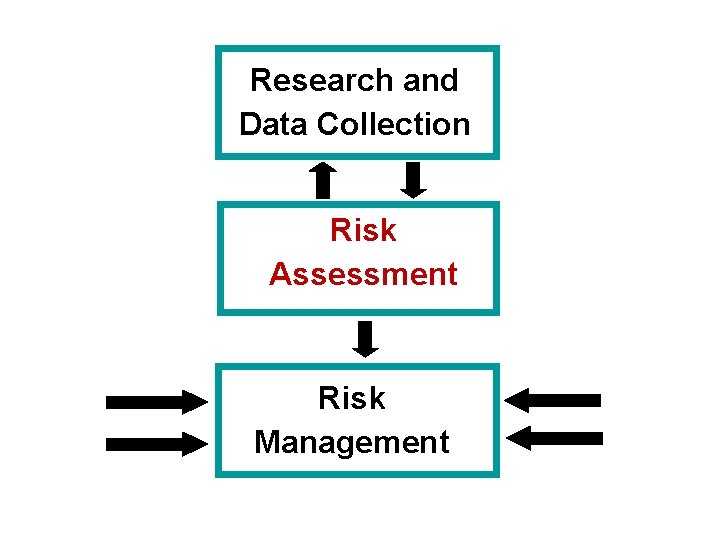
Research and Data Collection Risk Assessment Risk Management

Problems and Limitations: Risk Assessment 1. The process by which policy and science are mixed together in a risk assessment is poorly understood. 2. Its ability to provide a “Bright Line” is overstated by risk assessors and overused by regulators and lawmakers.

Problems and Limitations: Risk Assessment 3. The data quality objective for the different goals and types of risk assessment is poorly understood. 4. of the decision maker and the degree of complexity of the assessment, with unnecessary analyses confusing and delaying response.

Problems and Limitations: Risk Assessment 5. Risk assessment often obscures the substantial gap between the data needs for good public health decision making and the lack of available data. 6. Risk assessment is secondary prevention rather than primary prevention.

Scientists manipulate formulae to match the real world. Policymakers manipulate the real world to match formulae.

Risk Assessment Guidelines: Use and Intent • • • Provide consistency Establish data quality objectives Assure technical quality Clarify scientific assumptions Allow flexibility to be transparent Provide public with “Road Map”

Precautionary Principle Described in the Rio Declaration: • Nations shall use the precautionary approach to protect the environment. • Where there are threats of serious or irreversible damage, scientific uncertainty shall not be used to postpone cost-effective measures to prevent environmental degradation.

RISK ASSESSMENT AND THE PRECAUTIONARY PRINCIPLE • The Precautionary Principle is already incorporated in Risk Assessment. • The Precautionary Principle should be incorporated into Risk Assessment. • The Precautionary Principle and Risk Assessment are completely antithetical.

Specific guidelines Available on www. cantacuzino. ro

Declaration of International Federation of Biosafety Association on Advancing Global Biosafety and Biosecurity 2011, the year of building international biosafety communities Longer-term goals • Promoting biosafety education, particularly of the younger generation, involving curriculum development within a common framework and a common methodology for evaluation. • Supporting appropriate and practical legislative framework development. • Developing a strategy to obtain funding for applied biosafety research programs

Declaration of International Federation of Biosafety Association on Advancing Global Biosafety and Biosecurity Short term goals • Raising awareness from the political and public perspectives, and encouraging collaboration among the human and animal health communities • Developing sustainable biosafety associations and mapping gaps and needs • Providing advice to the competent national authorities and services for the improvement of efficient waste management (through disseminating knowledge of appropriate systems, tools and technologies for public and veterinary health environments). • Promoting human and laboratory capacity building with proper identification of gaps and needs (using the best available concepts, principles and practices)

Principals of Risk Management Risk management The eradication or minimisation of the adverse affects of risks to which an organisation is exposed.

Prior Planning Prevents Poor Performance

Risk Management • Pathogen • Procedures • Personnel • Place • Protective Equipment

Ineffective Program Management • • • Morbidity/mortality Perceived lack of institutional control Regulatory visit/action Legal action (civil/criminal penalties) Bad press Low morale

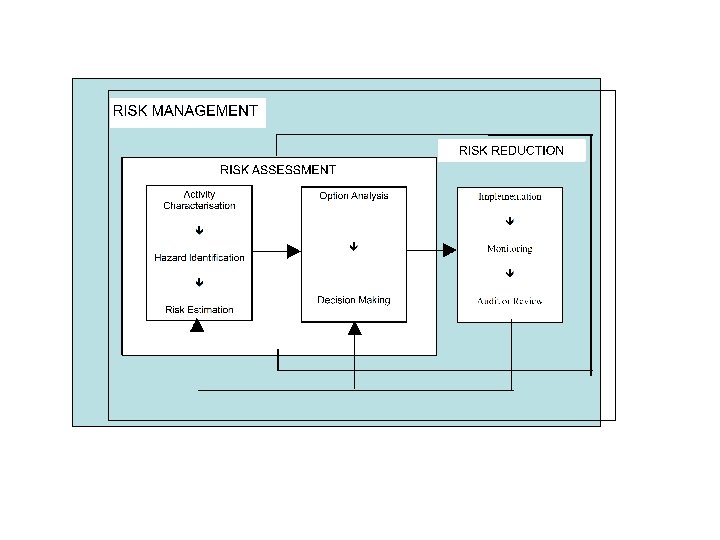
Stages in Risk Management • Identifying the hazards. • Evaluating the associated risks. • Controlling the risks.

Identify Hazards Broad categories of hazard To help with the process of identifying hazards it is useful to categorise hazards in different ways, for example by topic, e. g. : • • • Mechanical. Electrical. Radiation. Substances, Biological agents Fire and explosion.

Identification of Equipment • Autoclaves • Biosafety cabinets, clean benches, glove boxes • Eyewashes, showers • Centrifuge containment

Identification of Handlers • Name, employee ID Number • Campus address/contact information • Identify work experience • Observe work practices if necessary • Refresher training as needed

Determine risk The risk from the hazard should be determined by estimating the potential severity of harm and the likelihood that harm will occur.

Severity of harm • Information obtained about work activities is a vital input to risk assessment. To establish potential severity of harm, points should be considered: • Part(s) of the body likely to be affected; • Nature of the harm, ranging from slightly to extremely harmful: – 1) Slightly harmful, e. g. : • Superficial injuries; minor cuts and bruises; eye irritation from dust. • Nuisance and irritation (e. g. headaches); ill-health leading to temporary discomfort.

Severity of Harm – 2) Harmful, e. g. • Lacerations; burns; serious sprains; minor fractures. • Deafness; dermatitis; asthma; work related upper limb disorders; ill-health leading to permanent minor disability. – 3) Extremely harmful, e. g. • Amputations; major fractures; poisonings; multiple injuries; fatal injuries. • Occupational cancer; other severely life shortening diseases; acute fatal diseases.
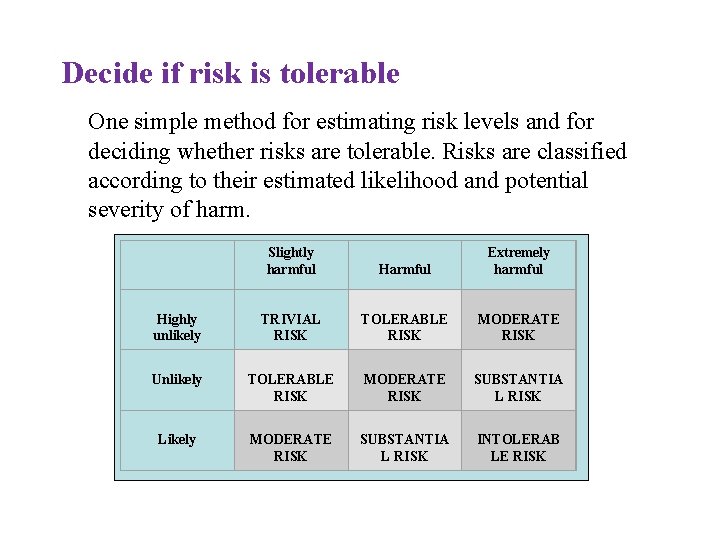
Decide if risk is tolerable One simple method for estimating risk levels and for deciding whether risks are tolerable. Risks are classified according to their estimated likelihood and potential severity of harm. Slightly harmful Harmful Extremely harmful Highly unlikely TRIVIAL RISK TOLERABLE RISK MODERATE RISK Unlikely TOLERABLE RISK MODERATE RISK SUBSTANTIA L RISK Likely MODERATE RISK SUBSTANTIA L RISK INTOLERAB LE RISK
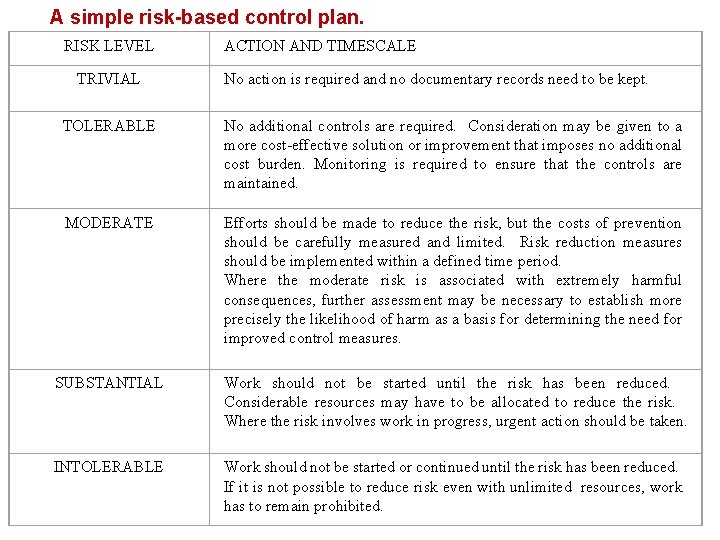
A simple risk-based control plan. RISK LEVEL TRIVIAL ACTION AND TIMESCALE No action is required and no documentary records need to be kept. TOLERABLE No additional controls are required. Consideration may be given to a more cost-effective solution or improvement that imposes no additional cost burden. Monitoring is required to ensure that the controls are maintained. MODERATE Efforts should be made to reduce the risk, but the costs of prevention should be carefully measured and limited. Risk reduction measures should be implemented within a defined time period. Where the moderate risk is associated with extremely harmful consequences, further assessment may be necessary to establish more precisely the likelihood of harm as a basis for determining the need for improved control measures. SUBSTANTIAL Work should not be started until the risk has been reduced. Considerable resources may have to be allocated to reduce the risk. Where the risk involves work in progress, urgent action should be taken. INTOLERABLE Work should not be started or continued until the risk has been reduced. If it is not possible to reduce risk even with unlimited resources, work has to remain prohibited.

Prepare risk control action plan The action plan should be reviewed before implementation, • Will the revised controls lead to tolerable risk levels? • Are new hazards created? • Has the most cost-effective solution been chosen? • What do people affected think about the need for, and practicality of, the revised preventive measures? • Will the revised controls be used in practice, and not ignored in the face of, for example, pressures to get the job done?

Prepare risk control action plan • Risk categories shown form the basis for deciding whether improved controls are required and the time scale for action. • The outcome of a risk assessment should be an inventory of actions, in priority order, to devise, maintain or improve controls.

Changing Conditions and Revising Risk assessment should be seen as a continuing process. Thus, the adequacy of control measures should be subject to continual review and revised if necessary.

ALARP • Legislation requires employers to reduce risks to a level that is as low as is reasonably practicable (sometimes abbreviated as ALARP). • To carry out a duty so far as is reasonably practicable means that the degree of risk in a particular activity or environment can be balanced against the time, trouble, cost and physical difficulty of taking measures to avoid the risk.

Uses of Risk Assessment for Management Decision Making • National Regulations: – Environmental standards (air, water, hazardous waste, etc. ) – Food safety (chemical contaminants, additives, pathogens) – Manufacturing and production (pharmaceuticals, pesticides, etc. ) • International Trade / WTO SPS Agreement: – Food products (Safety Assessment and Acceptable Daily Intake [ADI]) – Animals and animal products – Plants and plant products
- Slides: 66