Biopotential electrodes A complex interface Basics of Instrumentation

Biopotential electrodes A complex interface Basics of Instrumentation, Measurement and Analysis 2011, 2012
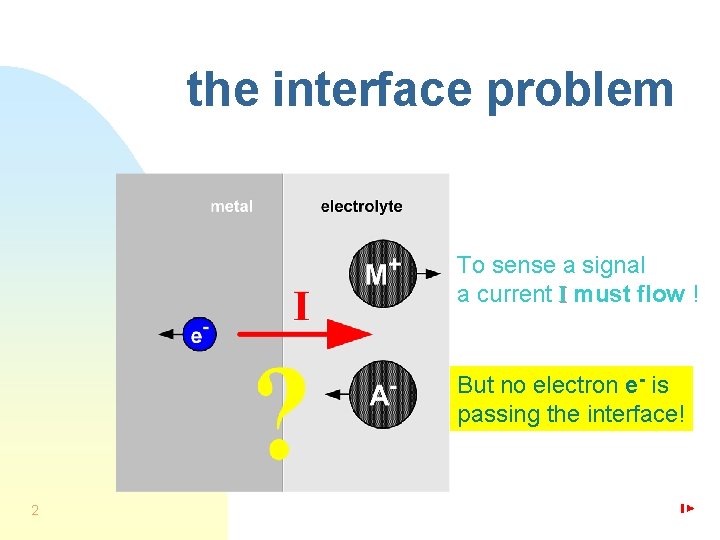
the interface problem To sense a signal a current I must flow ! But no electron e- is passing the interface! 2
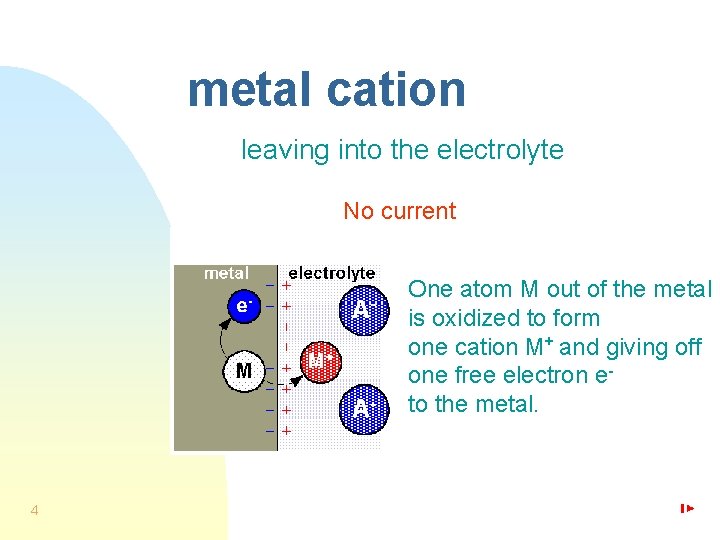
metal cation leaving into the electrolyte No current One atom M out of the metal is oxidized to form one cation M+ and giving off one free electron eto the metal. 4
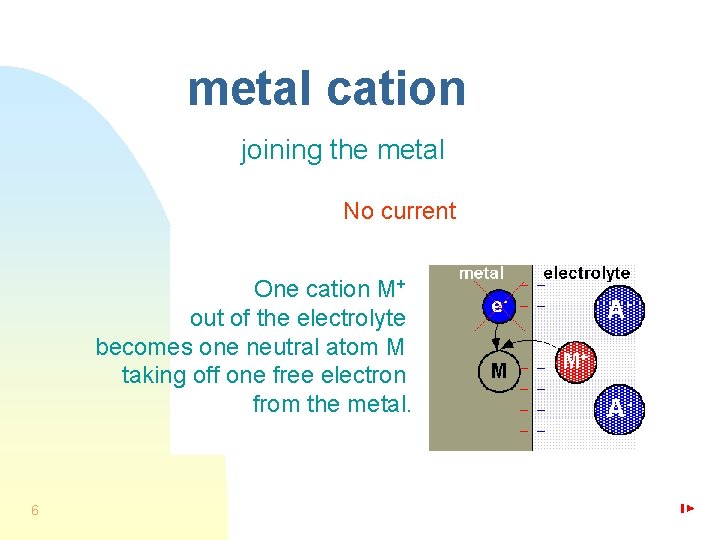
metal cation joining the metal No current One cation M+ out of the electrolyte becomes one neutral atom M taking off one free electron from the metal. 6
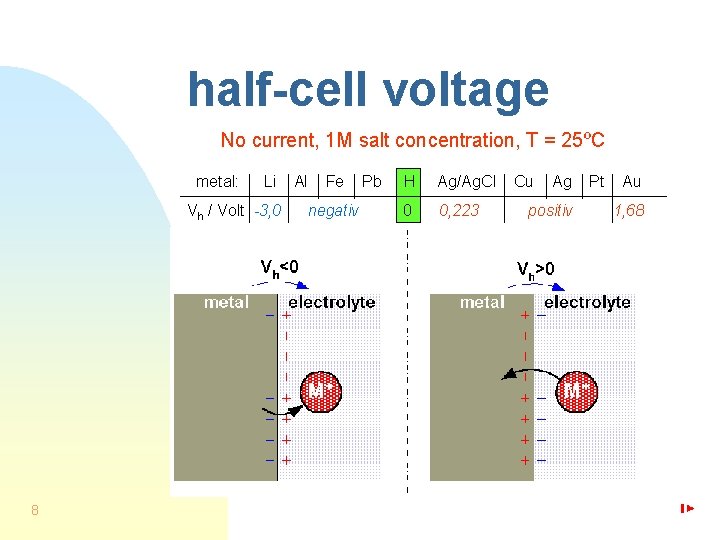
half-cell voltage No current, 1 M salt concentration, T = 25ºC metal: Li Vh / Volt -3, 0 8 Al Fe negativ Pb H Ag/Ag. Cl 0 0, 223 Cu Ag positiv Pt Au 1, 68

Nernst equation For arbitrary concentration and temperature E = RT/(z. F)·ln(c/K) E – electrode potential R = 8. 314 J /(mol*K) – molar gas constant T – absolute temperature z – valence F = 96485 C/mol – Faraday’s constant c – concentration of metal ion in solution K – “metal solution pressure”, or tendency to dissolve 9
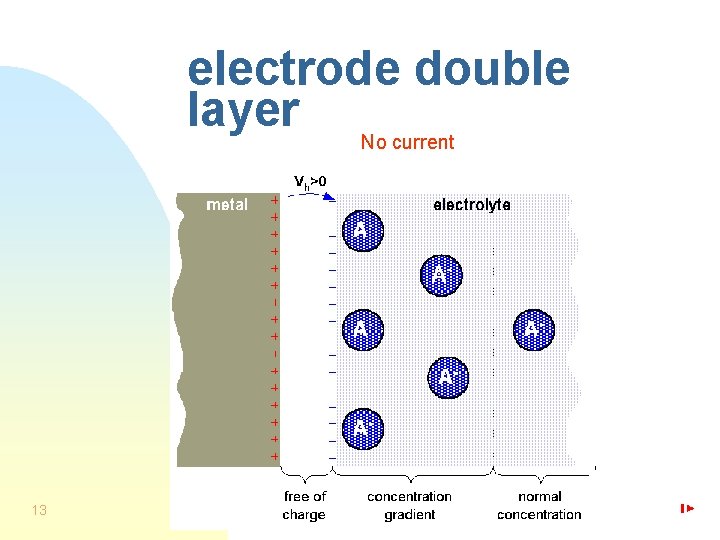
electrode double layer No current 13

current influence n n n with current flowing the half-cell voltage changes this voltage change is called overpotential or polarization: Vp = V r + V c + V a activation, depends on direction of reaction concentration (change in double layer) ohmic (voltage drop) 16

polarizable electrode n n 18 “perfectly” polarizable electrode: - only displacement current, electrode behave like a capacitor example: noble metals like platinum Pt

nonpolarizable electrode n n 20 “perfectly” nonpolarizable electrode: - current passes freely across interface, - no overpotential examples: - silver/silver chloride (Ag/Ag. Cl), - mercury/mercurous chloride (Hg/Hg 2 Cl 2) (calomel)
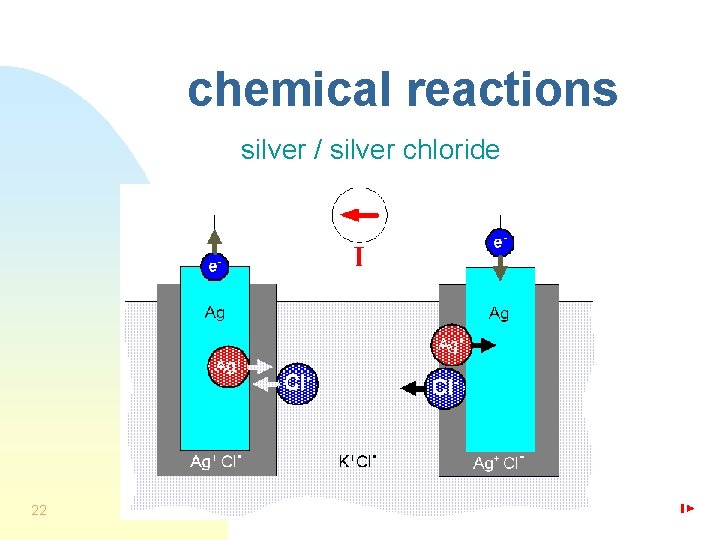
chemical reactions silver / silver chloride 22
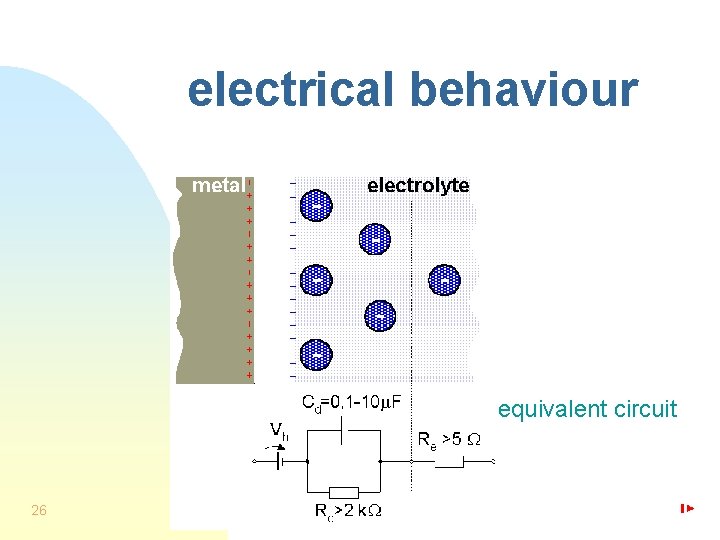
electrical behaviour equivalent circuit 26
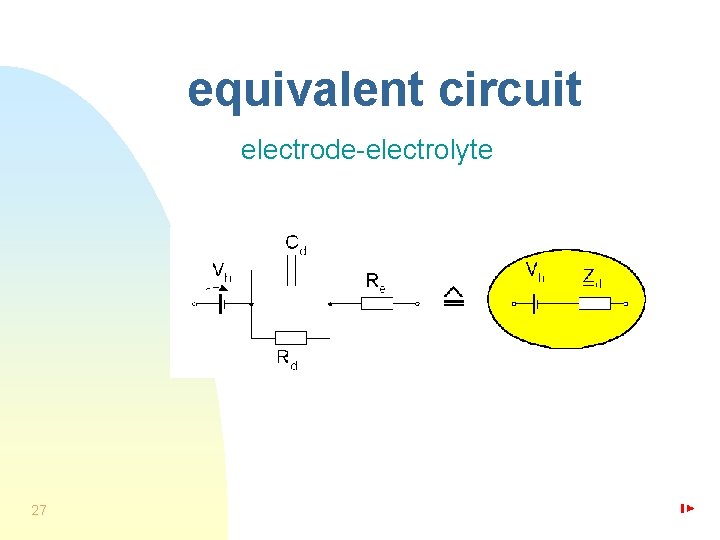
equivalent circuit electrode-electrolyte 27
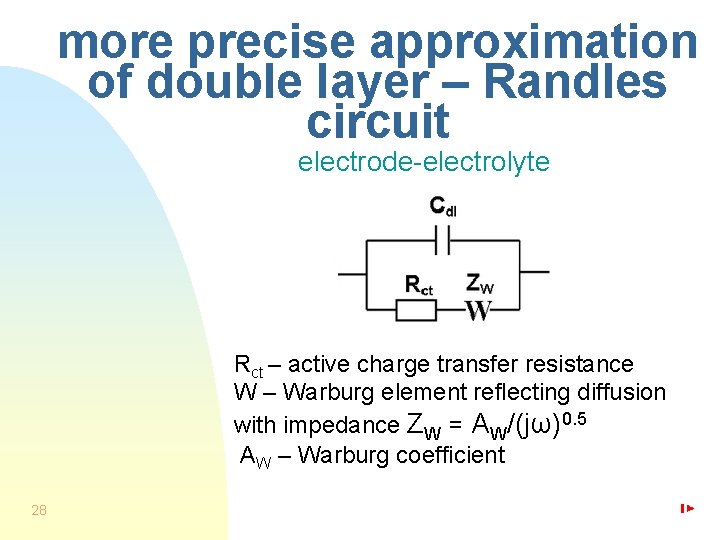
more precise approximation of double layer – Randles circuit electrode-electrolyte Rct – active charge transfer resistance W – Warburg element reflecting diffusion with impedance ZW = AW/(jω)0. 5 AW – Warburg coefficient 28
- Slides: 14