Biopolymers and Bioplastics August 10 12 2015 San

Biopolymers and Bioplastics, August 10 -12, 2015 San Francisco, USA Utilization of microalgal-derived ash as a mineral reinforcement material in biocomposite formulation with poly(vinyl alcohol) TRAN DANG THUAN, Ph. D Advanced Biomass R&D Center, Republic of Korea

Contents I. Introduction II. Experimental III. Characterization of materials IV. Properties of composite materials V. Summary & conclusions
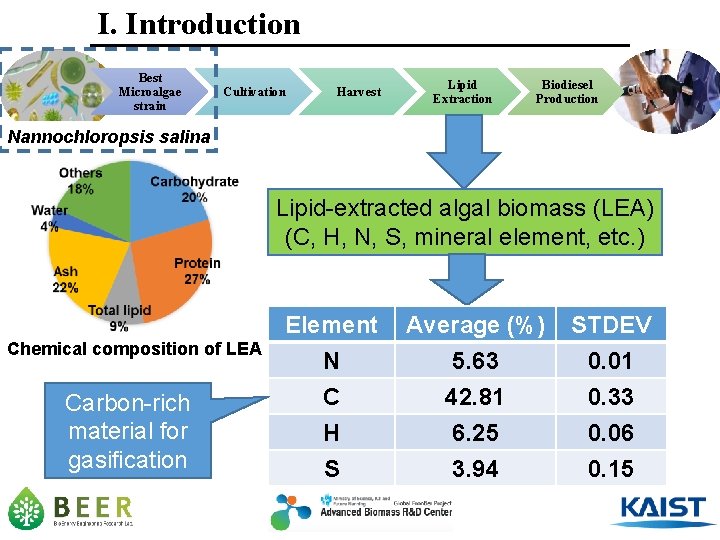
I. Introduction Best Microalgae strain Cultivation Harvest Lipid Extraction Biodiesel Production Nannochloropsis salina Lipid-extracted algal biomass (LEA) (C, H, N, S, mineral element, etc. ) Chemical composition of LEA Carbon-rich material for gasification Element N C H Average (%) 5. 63 42. 81 6. 25 STDEV 0. 01 0. 33 0. 06 S 3. 94 0. 15
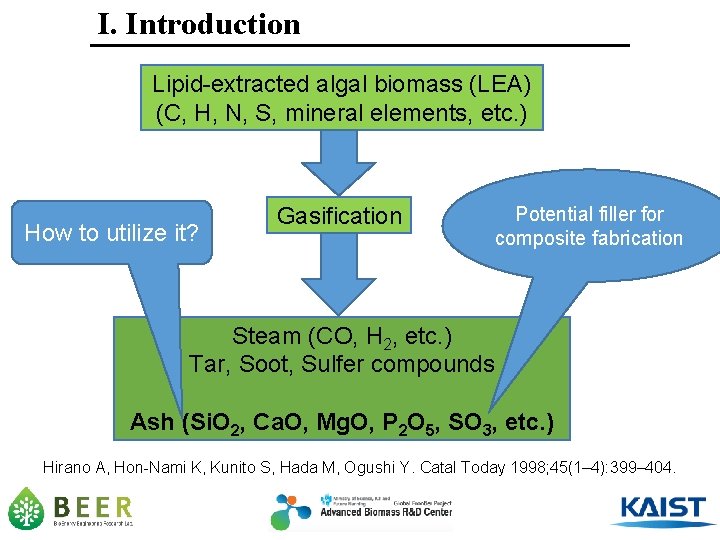
I. Introduction Lipid-extracted algal biomass (LEA) (C, H, N, S, mineral elements, etc. ) How to utilize it? Gasification Potential filler for composite fabrication Steam (CO, H 2, etc. ) Tar, Soot, Sulfer compounds Ash (Si. O 2, Ca. O, Mg. O, P 2 O 5, SO 3, etc. ) Hirano A, Hon-Nami K, Kunito S, Hada M, Ogushi Y. Catal Today 1998; 45(1– 4): 399– 404.
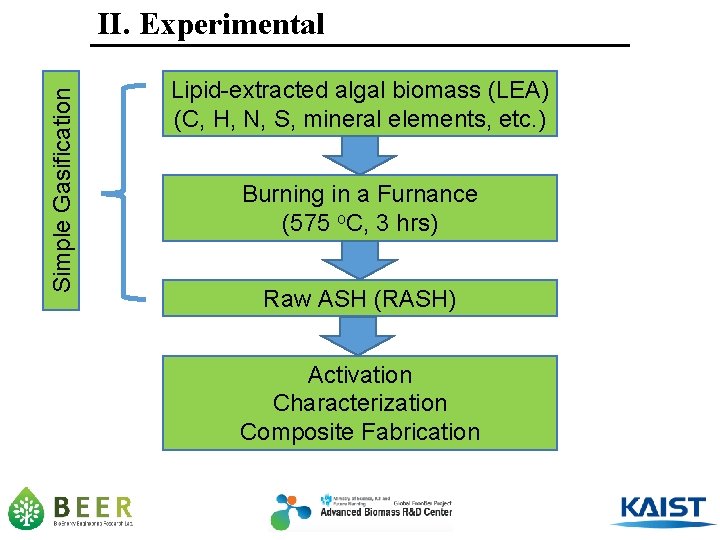
Simple Gasification II. Experimental Lipid-extracted algal biomass (LEA) (C, H, N, S, mineral elements, etc. ) Burning in a Furnance (575 o. C, 3 hrs) Raw ASH (RASH) Activation Characterization Composite Fabrication
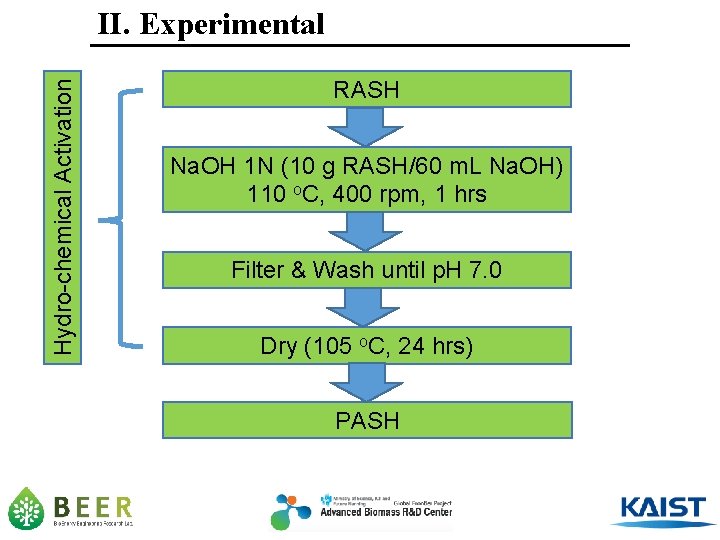
Hydro-chemical Activation II. Experimental RASH Na. OH 1 N (10 g RASH/60 m. L Na. OH) 110 o. C, 400 rpm, 1 hrs Filter & Wash until p. H 7. 0 Dry (105 o. C, 24 hrs) PASH
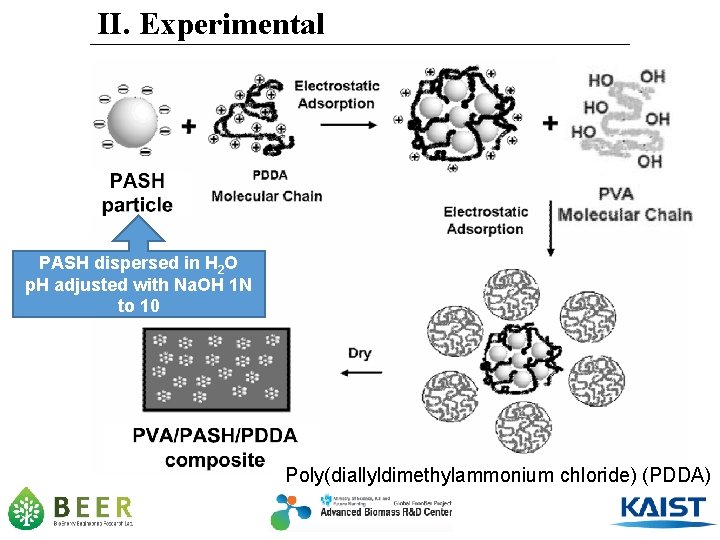
II. Experimental PASH dispersed in H 2 O p. H adjusted with Na. OH 1 N to 10 Poly(diallyldimethylammonium chloride) (PDDA)
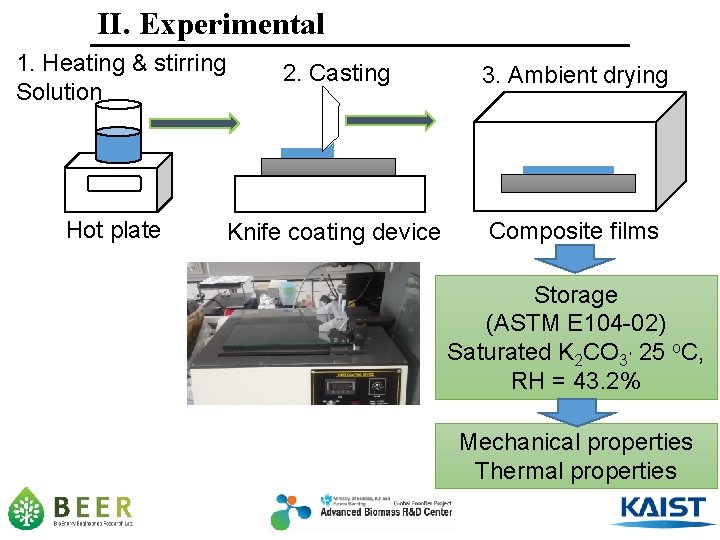
II. Experimental 1. Heating & stirring Solution Hot plate 2. Casting 3. Ambient drying Knife coating device Composite films Storage (ASTM E 104 -02) Saturated K 2 CO 3, 25 o. C, RH = 43. 2% Mechanical properties Thermal properties 8
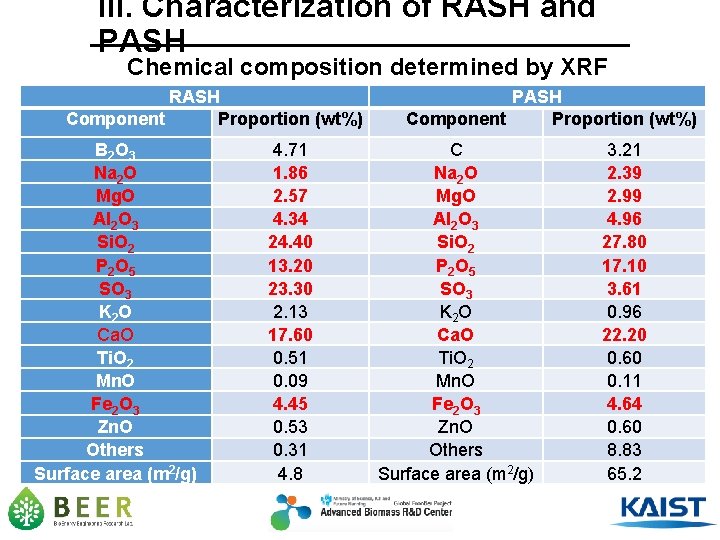
III. Characterization of RASH and PASH Chemical composition determined by XRF RASH Component Proportion (wt%) B 2 O 3 Na 2 O Mg. O Al 2 O 3 Si. O 2 P 2 O 5 SO 3 K 2 O Ca. O Ti. O 2 Mn. O Fe 2 O 3 Zn. O Others Surface area (m 2/g) 4. 71 1. 86 2. 57 4. 34 24. 40 13. 20 23. 30 2. 13 17. 60 0. 51 0. 09 4. 45 0. 53 0. 31 4. 8 PASH Component Proportion (wt%) C Na 2 O Mg. O Al 2 O 3 Si. O 2 P 2 O 5 SO 3 K 2 O Ca. O Ti. O 2 Mn. O Fe 2 O 3 Zn. O Others Surface area (m 2/g) 3. 21 2. 39 2. 99 4. 96 27. 80 17. 10 3. 61 0. 96 22. 20 0. 60 0. 11 4. 64 0. 60 8. 83 65. 2
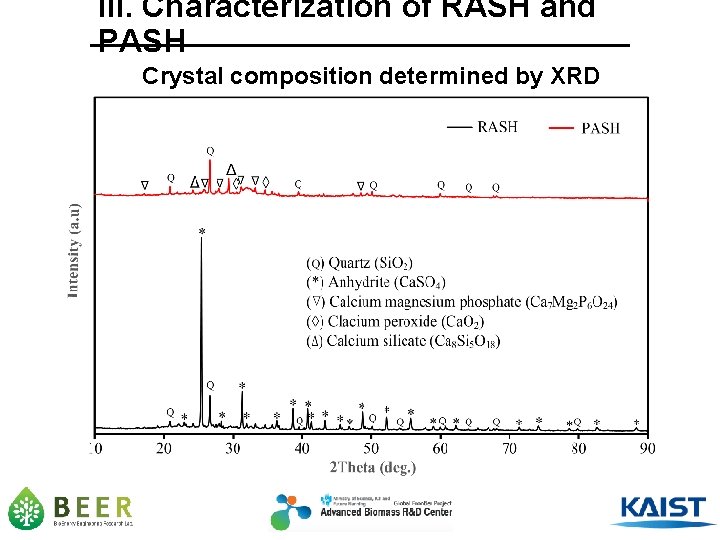
III. Characterization of RASH and PASH Crystal composition determined by XRD
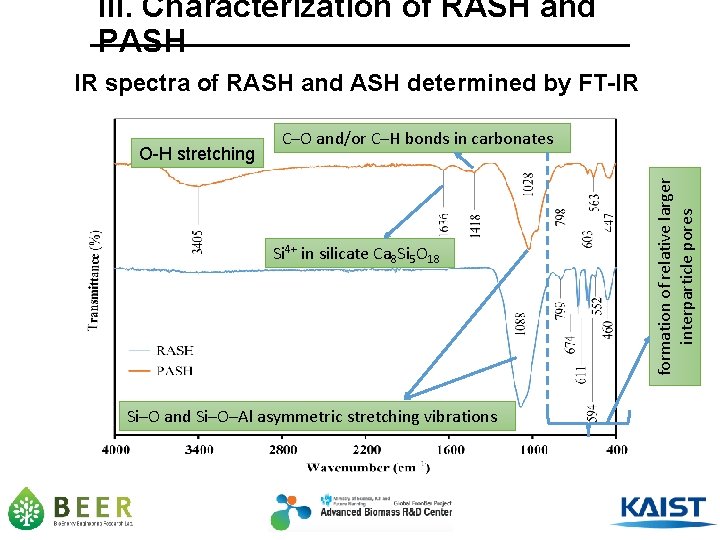
III. Characterization of RASH and PASH IR spectra of RASH and ASH determined by FT-IR Si 4+ in silicate Ca 8 Si 5 O 18 Si─O and Si─O─Al asymmetric stretching vibrations formation of relative larger interparticle pores O-H stretching C─O and/or C─H bonds in carbonates

III. Characterization of RASH and PASH FE-SEM micrographs of RASH and PASH RASH s e l ic e t r a fac p r e u k s li th t le oo e t m Pla ith s w r a l u ers g e PASH irr rat th nd c i w , a ce s le ugh rfa c i rt , ro s su a P pe it n a o sh
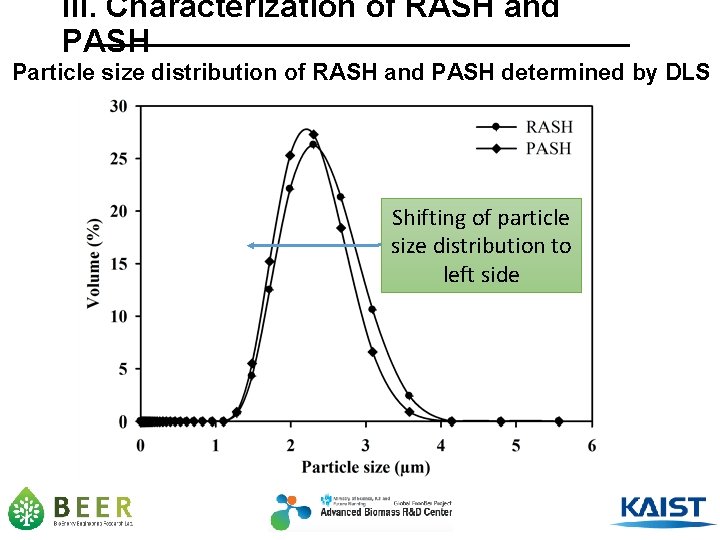
III. Characterization of RASH and PASH Particle size distribution of RASH and PASH determined by DLS Shifting of particle size distribution to left side
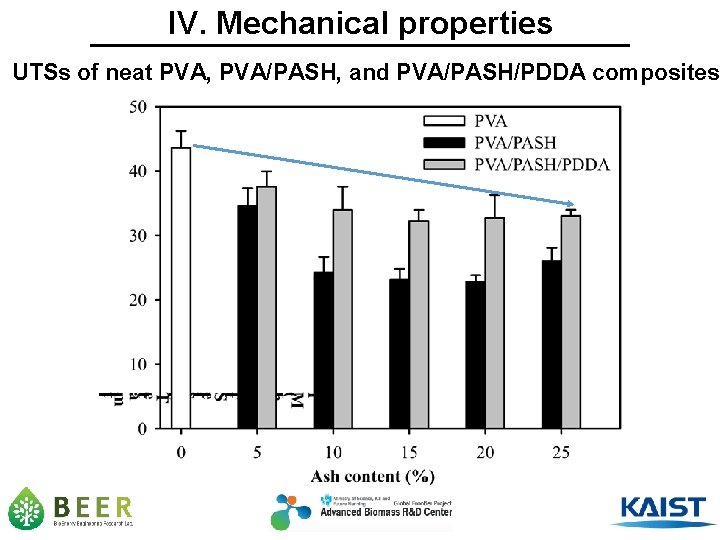
IV. Mechanical properties UTSs of neat PVA, PVA/PASH, and PVA/PASH/PDDA composites
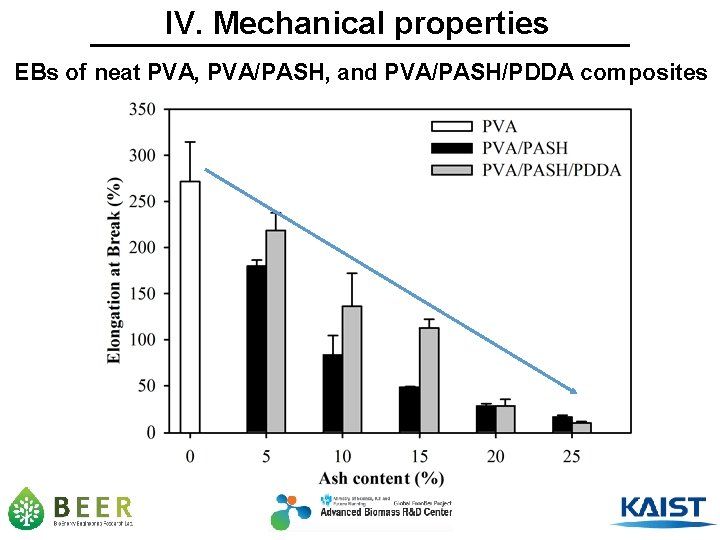
IV. Mechanical properties EBs of neat PVA, PVA/PASH, and PVA/PASH/PDDA composites
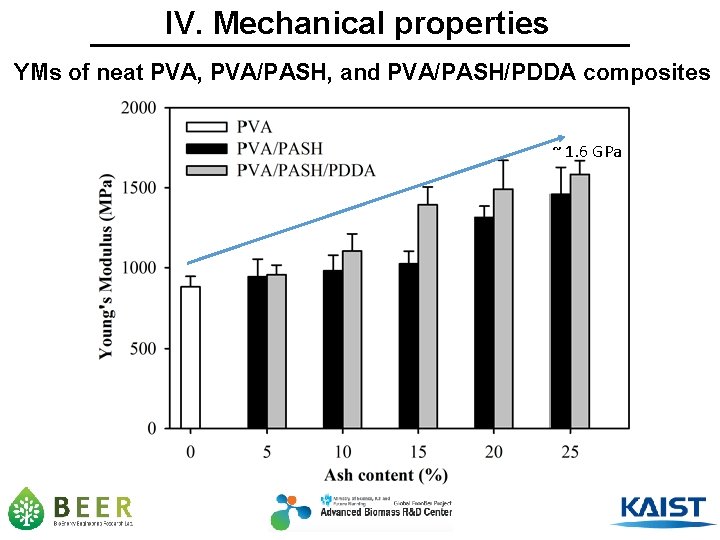
IV. Mechanical properties YMs of neat PVA, PVA/PASH, and PVA/PASH/PDDA composites ~ 1. 6 GPa
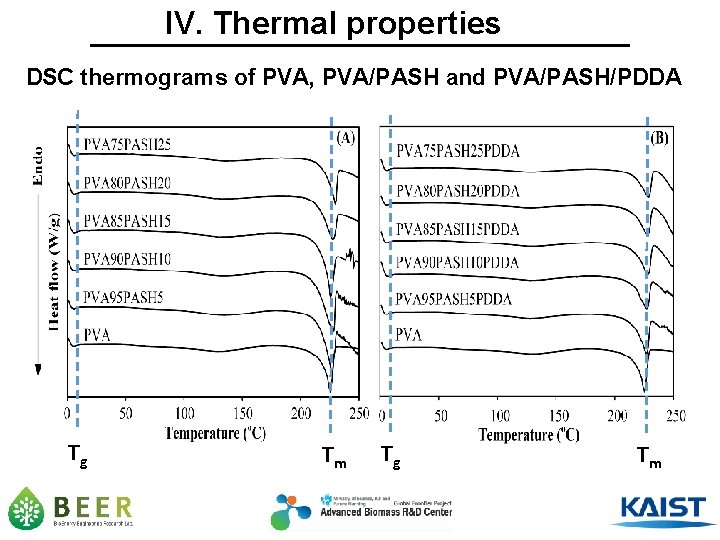
IV. Thermal properties DSC thermograms of PVA, PVA/PASH and PVA/PASH/PDDA Tg Tm
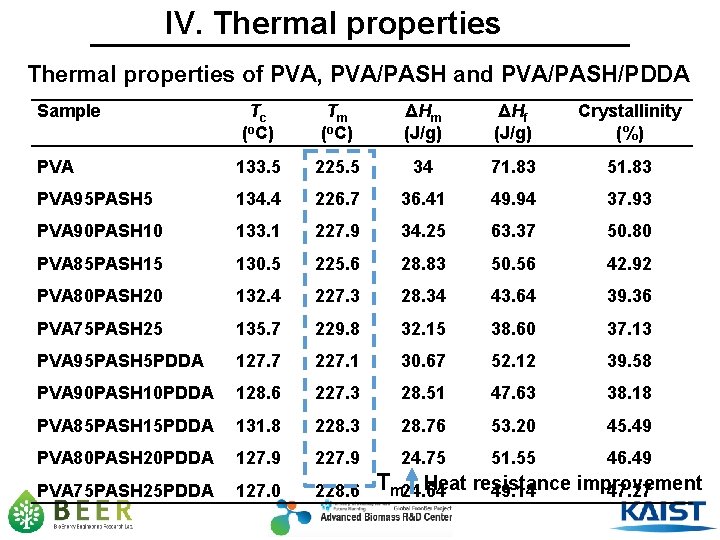
IV. Thermal properties of PVA, PVA/PASH and PVA/PASH/PDDA Sample Tc (o. C) Tm (o. C) ΔHm (J/g) ΔHf (J/g) Crystallinity (%) PVA 133. 5 225. 5 34 71. 83 51. 83 PVA 95 PASH 5 134. 4 226. 7 36. 41 49. 94 37. 93 PVA 90 PASH 10 133. 1 227. 9 34. 25 63. 37 50. 80 PVA 85 PASH 15 130. 5 225. 6 28. 83 50. 56 42. 92 PVA 80 PASH 20 132. 4 227. 3 28. 34 43. 64 39. 36 PVA 75 PASH 25 135. 7 229. 8 32. 15 38. 60 37. 13 PVA 95 PASH 5 PDDA 127. 7 227. 1 30. 67 52. 12 39. 58 PVA 90 PASH 10 PDDA 128. 6 227. 3 28. 51 47. 63 38. 18 PVA 85 PASH 15 PDDA 131. 8 228. 3 28. 76 53. 20 45. 49 PVA 80 PASH 20 PDDA 127. 9 24. 75 51. 55 46. 49 PVA 75 PASH 25 PDDA 127. 0 228. 6 Tm 24. 64 Heat resistance improvement 49. 14 47. 27
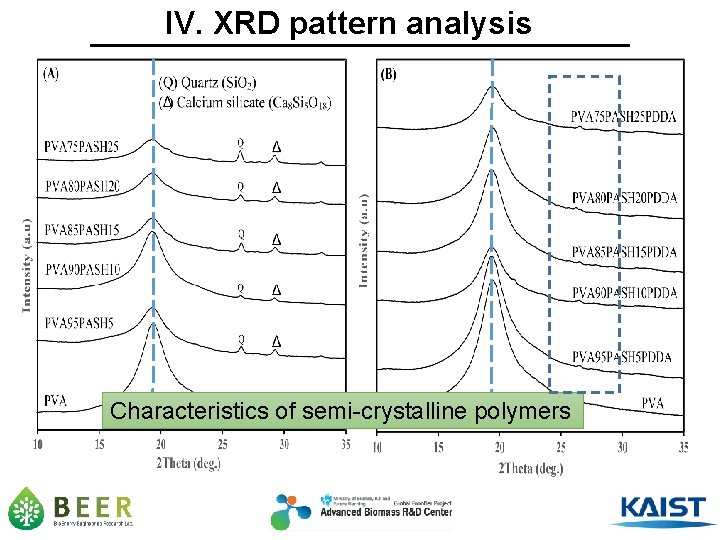
IV. XRD pattern analysis Characteristics of semi-crystalline polymers
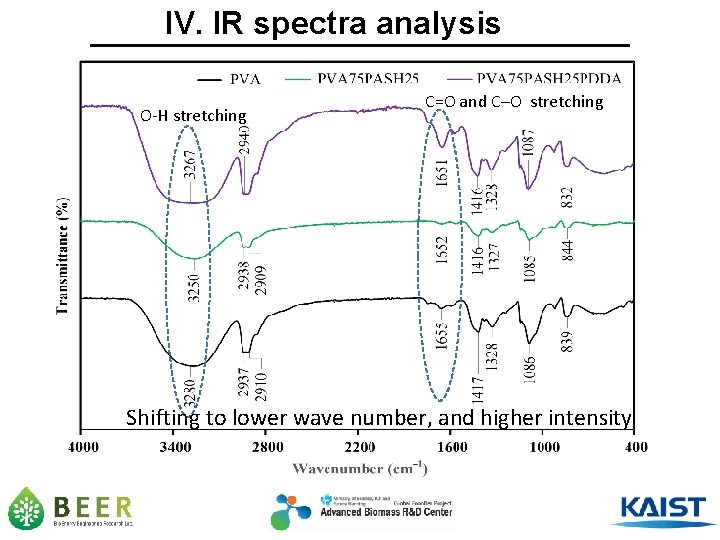
IV. IR spectra analysis O-H stretching C=O and C─O stretching Shifting to lower wave number, and higher intensity

PVA 95 PASH 5 bridge-like connection brittle failure, poor filler-matrix adhesion PVA 75 PASH 25 PDDA Full coverage Very little voids PVA 95 PASH 5 PDDA PVA 75 PASH 25 Abundance interfacial voids Partial full coverage IV. Morphological analysis (SEM) better polymer-reinforcement adhesion

V. Summaries & Conclusions § Hydrochemical activation made RASH become PASH with smaller size, rough and crater surface, and high surface area § Better dispersion and adhesion of PASH in PVA was observed in presence of polycations (PDDA) § Result in higher tensile strength of PVA/PASH/PDDA compared to PVA/PASH composites at every loading of the filler § The process of pretreatment of RASH, utilization of PASH by composite formulation with PVA and polycation is easy and efficient, which can be integrated in downstream processing of microalgae-based biorefinery § Value-added composites produced from microalgal ash can partially improve economical feasibility of microalgal industry

Acknowledgements § This research was financially supported by the Advanced Biomass R&D Center (ABC) of Korea Grant funded by the Ministry of Science, ICT and Future Planning (ABC-20100029728). § Supervised supports from Prof. Ji-Won Yang (CBE, KAIST) Prof. Min S. Park (CBE, ABC, KAIST) Prof. Yong Keun Chang (CBE, ABC, KAIST) § Many helps from my student Hyun-Ro Lee (M. S. , CBE, KAIST)

Thank you for your attention!
- Slides: 24