Biophysics of Breathing Jan Jaku Breathing is a

Biophysics of Breathing Jan Jakuš

Breathing is a vital function of the body, a periodic and rhythmic process of inspiration and expiration that covers the metabolic demands of body for O 2 and CO 2. - must assure in adults the intake of O 2 250 ml / min, and the expenditure of CO 2 200 ml / min. - is governed involuntarily by “ a respiratory centre’’, localized within the brainstem - - can be interrupted or increased voluntarily (from the cortex) (for more info look at a book: Jakus, Tomori Stransky: Neuronal Determinants of Breathing, Coughing and Related Motor Behaviours, 2004, Wist, 335 p. )
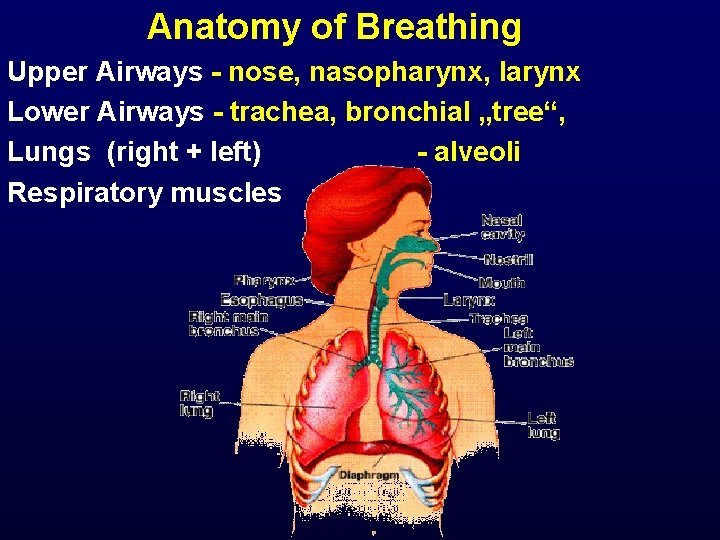
Anatomy of Breathing Upper Airways - nose, nasopharynx, larynx Lower Airways - trachea, bronchial „tree“, Lungs (right + left) - alveoli Respiratory muscles
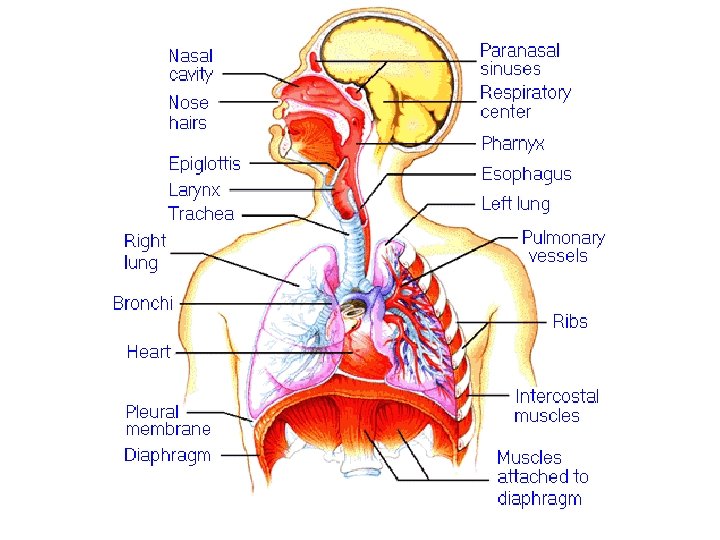

Breathing (Respiration) : - External (the air exchange at the level of lungs) - Internal (the O 2 and CO 2 exchange at the tissue level)
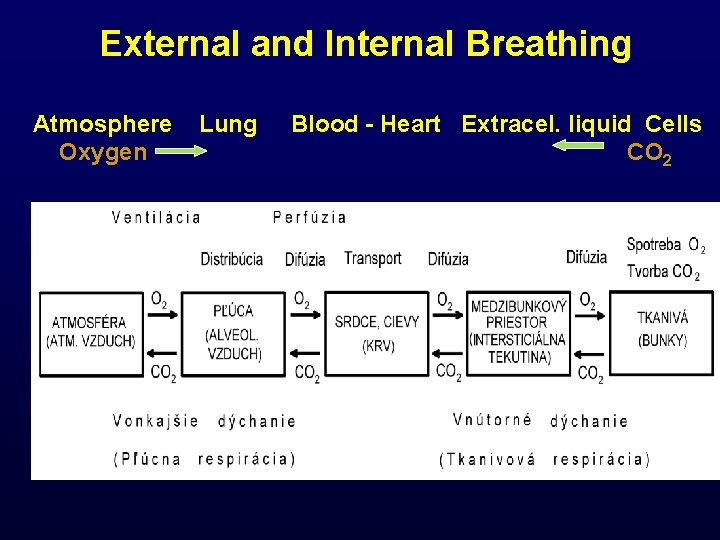
External and Internal Breathing Atmosphere Lung Blood - Heart Extracel. liquid Cells Oxygen CO 2
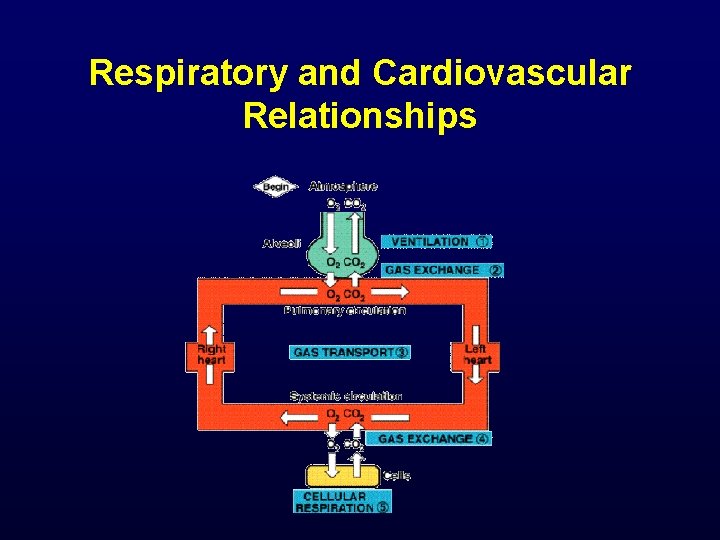
Respiratory and Cardiovascular Relationships

External Breathing : 1. VENTILATION - cyclic air exchange during breathing caused by the respiratory „pump“ muscles - diaphragm external and internal intercostals, abdominal, and auxiliary muscles (Jakus et al. book) 2. DISTRIBUTION - mixing of inhaled air with an air that remains within the airways after expiration (150 ml-death volume). 3. DIFFUSION - transfer of O 2 and CO 2 through the alveolar-capilĺary membrane along the partial pressure gradients (Fick´s Law) 4. PERFUSION- gas transport in blood between lungs and tissues by heart and vessels
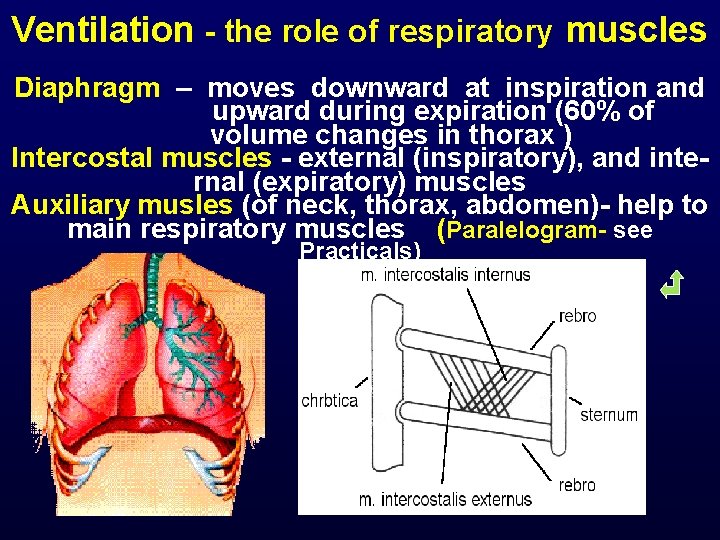
Ventilation - the role of respiratory muscles Diaphragm – moves downward at inspiration and upward during expiration (60% of volume changes in thorax ) Intercostal muscles - external (inspiratory), and internal (expiratory) muscles Auxiliary musles (of neck, thorax, abdomen)- help to main respiratory muscles (Paralelogram- see Practicals)
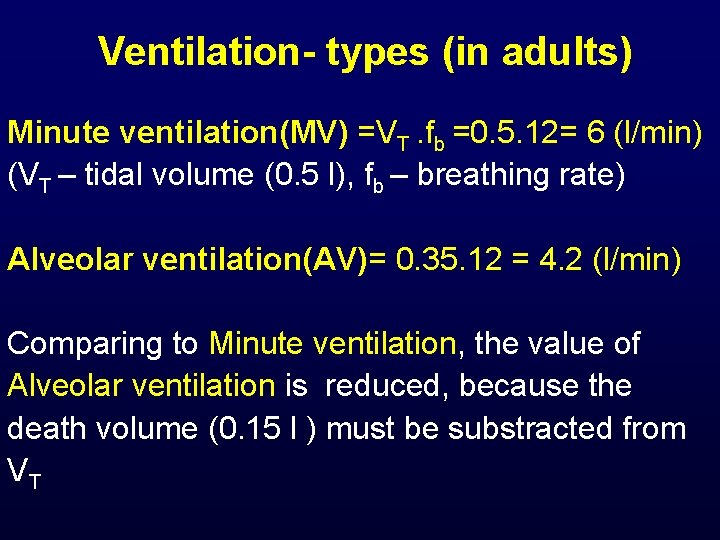
Ventilation- types (in adults) Minute ventilation(MV) =VT. fb =0. 5. 12= 6 (l/min) (VT – tidal volume (0. 5 l), fb – breathing rate) Alveolar ventilation(AV)= 0. 35. 12 = 4. 2 (l/min) Comparing to Minute ventilation, the value of Alveolar ventilation is reduced, because the death volume (0. 15 l ) must be substracted from VT

Origine of Breathing. Action potentials from respiratory centre drive respiratory muscles. These, in turn are contracted and create pressure changes. Pressure changes enable pressure gradient and this leads to a flow of air. Then lungs are filled (or emptied) with air volumes. (Hering´s model of breathing -see practicals) Remember these changes: A/ AT REST: QUIET INSPIRATION (active process): contraction of diaphragm + external intercostals fall of pleural pressure (PPl = - 0. 8 k. Pa) fall of intrapulmonary (Pp = - 0. 1 k. Pa ) Pressure gradient inspiratory airflow (VI = + 0. 4 l/s) inspiratory tidal volume (VT = 0. 5 l)

OUIET EXPIRATION ( mostly passive process) : recoil forces (i. e elasticity of the thoracic wall and lung tissue + passive movement of the diaphragm upward slightly negative Ppl = - 0. 1 k. P, and to slightly positive intrapulmonay pressure PP =+ 0. 5 k. Pa pressure gradient expiratory airflow (VE= - 0. 4 l/s) expiratory volume ( VT = 0. 5 l ) empties the lungs B/ AT WORK: FORCEFUL INSPIRATION consists of the same processes as shown above + contraction of external intercostals + auxiliary muscles result in higher pressure gradients, and to higher values of Ppl, PP, VE and VT

FORCEFUL EXPIRATION (e. g. in cough, sneeze, strong voluntary expiration) It starts sudenly with contraction of abdominal muscles (expiratory), creating high abdominal pressure (PAB), very high PPl and PP pressures, also very high pressure gradient , and thus extremely strong expiratory airflow (velocity like tornado) and very high expiratory volume

Mechanics of breathing - means concomitant changes of respiratory muscles (diaphragm, intercostal and auxiliary muscles) creating particular Ppl and PP, pressures, inspiratory and expiratory airflows (V), and tidal volumes (VT), resulting in some Work of breathing (during inspiration and expiration). Work of breathing is affected by: Lung compliance, Airway resistance

1/ Lung compliance – distensibility (C) - is the ratio between Volume of air (VT) / Pressure( P). N = 2 (l. k. Pa-1) Lung fibrosis C - the lung tissue is thicker and thus its compliance (distensibility) is lower Lung emphysema C- lung tissue is thinner, and thus compliance (distensibility) of the lungs is higher. LOOP OF LUNG COMPLIANCE

2/ Airway resistance Raw - is the relationship between PRESSURE (P) / AIR FLOW (V) (Unit is k. Pa / l / s) In a disease like bronchial asthma the airway resistance is high, because the contraction of smooth muscles within the lower airways decreases the diameter of airways. Thus, the airflow is low, but Work of muscles and breathing is high. LOOP OF AIRWAY RESISTANCE
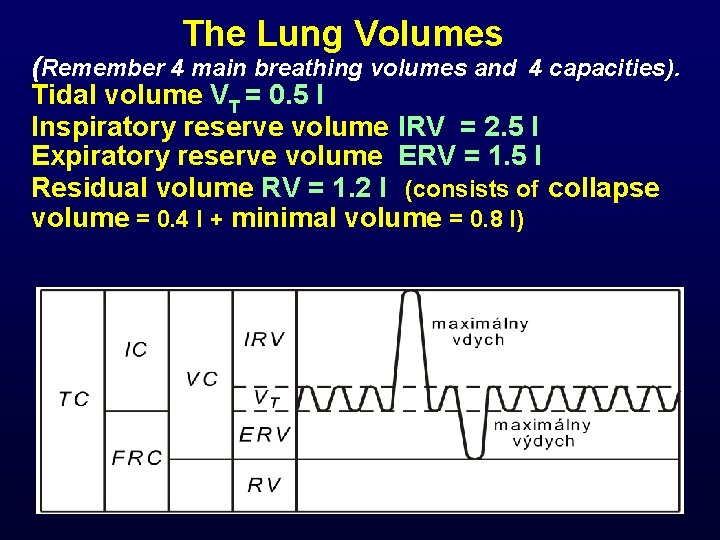
The Lung Volumes (Remember 4 main breathing volumes and 4 capacities). Tidal volume VT = 0. 5 l Inspiratory reserve volume IRV = 2. 5 l Expiratory reserve volume ERV = 1. 5 l Residual volume RV = 1. 2 l (consists of collapse volume = 0. 4 l + minimal volume = 0. 8 l)
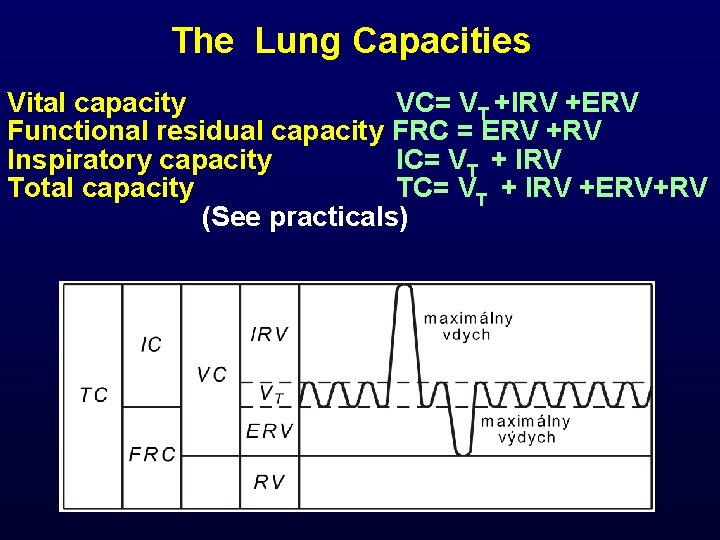
The Lung Capacities Vital capacity VC= VT +IRV +ERV Functional residual capacity FRC = ERV +RV Inspiratory capacity IC= VT + IRV Total capacity TC= VT + IRV +ERV+RV (See practicals)
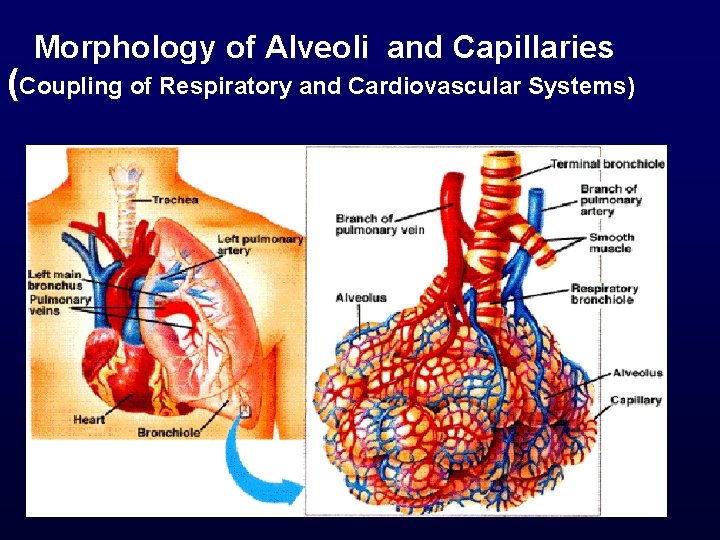
Morphology of Alveoli and Capillaries (Coupling of Respiratory and Cardiovascular Systems)

Partial Pressure of Gases- a drive for diffusion ATHMOSPHERIC AIR is a mixture of 21% of O 2 + 0. 04 % CO 2 + 78% of N 2 , and other residual gases (e. g. Hellium, Neon, Argon) Partial pressures of particular gases depend on their % concentration within the air. (DALTON´S LAW). The higher is % of a gas within a gas mixture, the higher is its partial pressure (and vice versa). At normal value of barometric pressure = 101. 3 k. Pa (760 torr, 1 atm) the partial pressure of P 02 is approx. 21 k. Pa and PCO 2 is 0. 04 k. Pa

Remember: Using DALTON’S LAW one can count partial pressure of a gas according to formula: PO 2 = V% O 2 x ( PB - PH 2 O ) / 100 PO 2 = 20. 93 x (101. 3 – 0. 8) / 100 = 21. 03 (k. Pa) PCO 2 = 0. 04 (k. Pa) PN 2 = 79 (k. Pa) This formula can be used for calculations of particular pressures of gases in the air, in the airways or within the arterial and venous blood. See next table.
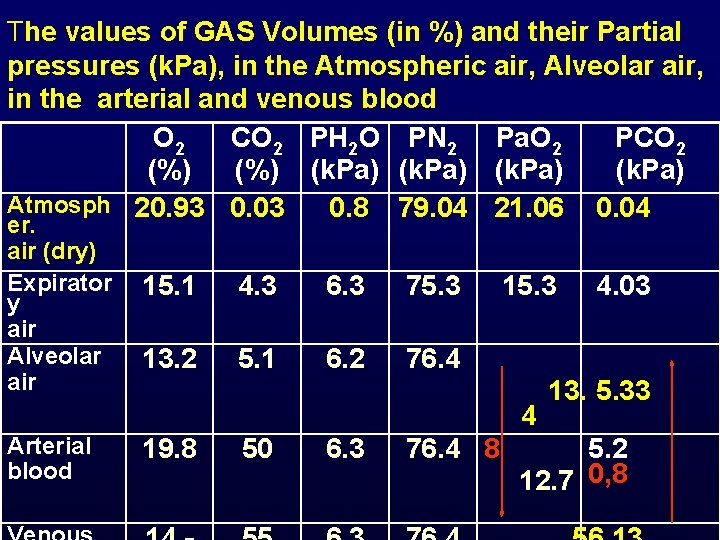
The values of GAS Volumes (in %) and their Partial pressures (k. Pa), in the Atmospheric air, Alveolar air, in the arterial and venous blood O 2 CO 2 PH 2 O PN 2 Pa. O 2 PCO 2 (%) (k. Pa) Atmosph 20. 93 0. 03 0. 8 79. 04 21. 06 0. 04 er. air (dry) Expirator y air Alveolar air Arterial blood 15. 1 4. 3 6. 3 75. 3 13. 2 5. 1 6. 2 76. 4 19. 8 50 6. 3 15. 3 4. 03 13. 5. 33 4 76. 4 8 5. 2 12. 7 0, 8

DIFFUSION – is a transfer of gases (O 2. . . ) through the Alveolar-capillary membrane along the partial pressure gradients of O 2 and CO 2 or N 2 being governed by : FICK’S LAW Diffusion rate: V = (P 1 – P 2). A. k s P 1 P 2 partial pressures A = diffusion surface (70 m 2) s = thickness of Alv. -capillary membrane (0. 8 um) k = diffusive constant -depends on a membrane and gas properties Diffusion rate : VO 2 = 15 – 20 ml / min. Diffusion rate for VCO 2 is 20 -times higher than for O (300 -400 ml/ min)
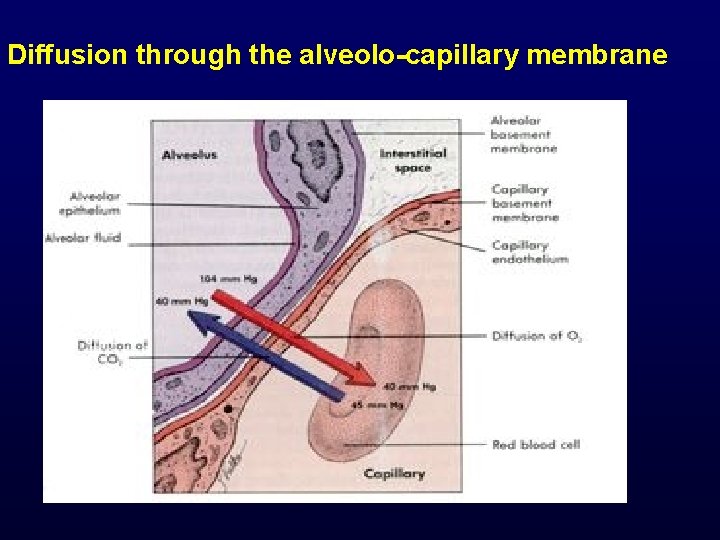
Diffusion through the alveolo-capillary membrane
Dynamics of Diffusion

PHYSICAL SOLUBILITY of O 2 and CO 2 within the blood plasma is under HENRY’S LAW: VO 2 = x PO 2 x 1000 = 3 ml O 2 /1 l arterial blood PB VCO 2 = x PCO 2 x 1000 = 27 ml CO 2 / 1 l arter. blood 101 , - coefficients for O 2, and CO 2 (respectively) PB - atmospheric (barometric) pressure Solubility of gases in liquids depends on their partial pressures. Gases in liquids are in two forms: physically disolved in blood plasma, and chemically bounded on Hemoglobine of the red blood cells. 1 l of arterial blood takes 200 ml of O 2. From this only 3 ml of O 2 is physically dissolved in plasma, and 197 ml O 2 binds chemically on Hemoglobine.

Wishing You Pleasant Day
- Slides: 27