Biophysics II By AProf Xiang Yang Liu Biophysics

Biophysics II By A/Prof. Xiang Yang Liu Biophysics & Micro/nanostructures Lab Department of Physics, NUS

Outline n n Review of Energy, Enthalpy and Entropy and the correlation with Q and W Equilibrium and Equilibrium constant

Energy, Enthalpy and Entropy The Change of Enthalpy and Entropy in Different Processes n As d. E = Qrev- Pd. V, At const V, d. E = Qrev For single phase E = Qrev= m. Cvd. T n As d. H = d. E + Pd. V + Vdp = Qrev- Pd. V + Vdp = Qrev+ Vdp At const P, d. H = Qrev For single phase H = Qrev= m. Cpd. T n d. S = Qrev/T, single phase Qrev= m. Cp(or Cv)d. T n Isothermal reversible expansion ( E = 0, H = (PV), S = Wrev/T; for ideal gas, S = n. Rln(Vf/Vi)) n At constant T and P (such as melting and evaporation) H = Q, S= H/T

Chemical potential n n n μ measures the availability of a particle species Goal: Understand how both concentration and internal energy of a molecular species enter its chemical availability. At equilibrium A, = B, matching role for macroscopic systems in equilibrium The corresponding standard thermodynamic quantities of Ho, So, Go Go = Ho - TSo, G o = Ho - T S o A solution with multi-solutes- similar to that of mixtures of gases, but instead of partial pressure of 1 atm, the concentrations for each solute are defined at 1 M (or mole fraction = 1, etc. depending on the unit of concentration used. ) n

Ideal mixing + A B AB What does it mean AB -1/2 ( AA + BB) = 0 E = Efin – Eini = 4(1/2 AB) – [2 (1/2 AA) + 2 (1/2 BB)] = 4[ AB -1/2 ( AA + BB)] = 0 No volume change ( V = 0) H = E + (PV). At const P, H = E + P (V) = 0

Ideal mixing n In the mixing of a multi components solution, Emix = Hmix = 0, smix-i = ni. Rlnxi (i = 1, 2, …, ), Smix = smix-i = R (nilnxi) Gmix-i = Hmix + T Smix= RT (nilnxi) n An ideal solution or ideal gases n G = Go + RT (ni. Rlnxi) ¨ Chemical potential at const T, P, i = [ G/ Ni]T, P, Nj, j I ¨ i = io + RTlnxi ( io: Standard Chemical potential) ¨ n The above expressions hold for a mixture of ideal gases, where xi = Pi/P.

Equilibrium constant of two states K Nn Nde
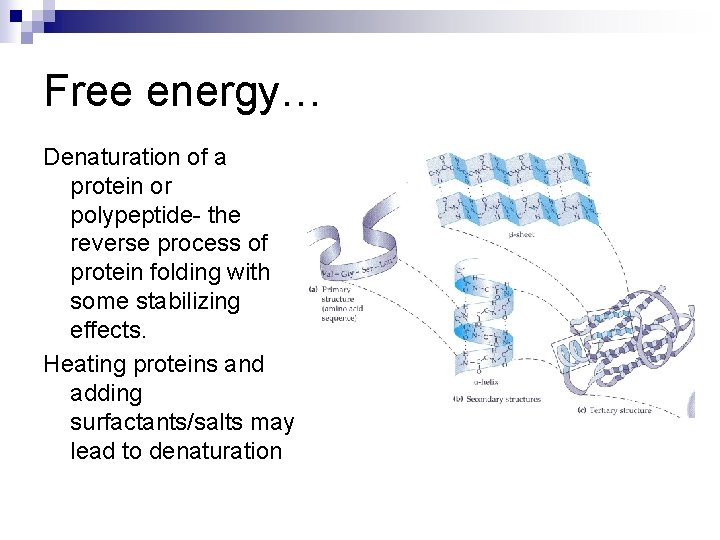
Free energy… Denaturation of a protein or polypeptide- the reverse process of protein folding with some stabilizing effects. Heating proteins and adding surfactants/salts may lead to denaturation
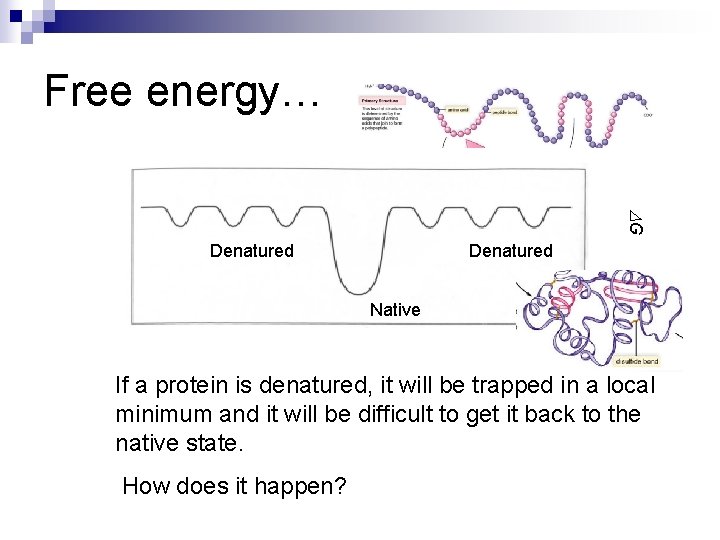
Free energy… G Denatured Native If a protein is denatured, it will be trapped in a local minimum and it will be difficult to get it back to the native state. How does it happen?

Free energy… Denaturation of a protein or polypeptide: ¨ Gden = Hden – T Sden ¨ S = R ln (Wden/Wnative) Since Wden/Wnative >> 1, H > 0 (require E) ¨ low T, G > 0, The breaking of the favorable interactions that hold the native ¨ high T , G < 0. conformation will surely require the input of energy, so H >0.

Free energy… n n The ratio of molecules at equilibrium For the special case, nd/nn is the ratio of molecules at equilibrium, G = 0
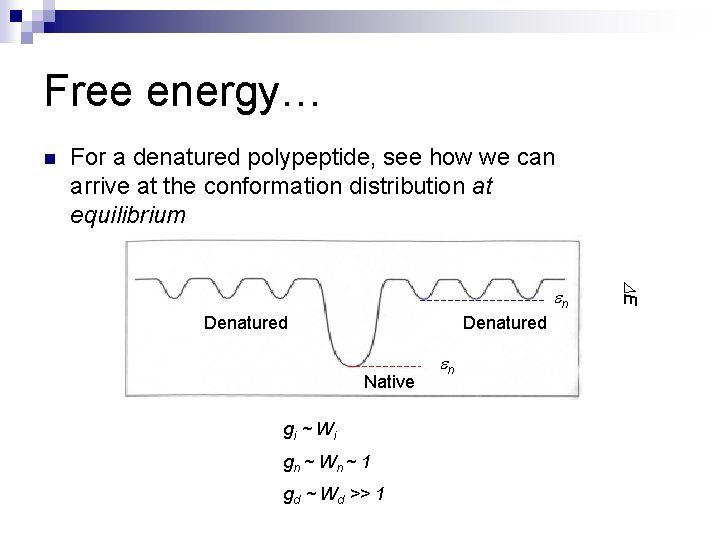
Free energy… n For a denatured polypeptide, see how we can arrive at the conformation distribution at equilibrium Denatured Native gi ~ W i gn ~ W n ~ 1 gd ~ Wd >> 1 n E n

Free energy… Boltzmann distribution Actual distribution

Free energy… Putting quantities on a molar basis: n K is the equilibrium o n = noconstant At equilibrium, G = 0, n = n d, n n Go is dthe standard free energy

Chemical Reactions
![Chemical Potential n n i = io+ RTln[i ]: concentration of ; io: chemical Chemical Potential n n i = io+ RTln[i ]: concentration of ; io: chemical](http://slidetodoc.com/presentation_image_h2/11d1bc0d88a53b5cb75b4f2b4a0a309a/image-16.jpg)
Chemical Potential n n i = io+ RTln[i ]: concentration of ; io: chemical potential at P = 1 atm, T = 298 K and [i ] 1. io depends on the unit of concentration selected, ie the unit of [i ] is mole fraction, xi, io: is the chemical potential at P = 1 atm, T = 298 K and xi 1; ¨ If the unit of [i ] is “molar” (moles per liter), Mi, io: is the chemical potential at P = 1 atm, T = 298 K and Mi 1. ¨ The same applied to other concentration units ¨ If
![Chemical Potential i = io+ RTln[i ] i: describing the availability of particles just Chemical Potential i = io+ RTln[i ] i: describing the availability of particles just](http://slidetodoc.com/presentation_image_h2/11d1bc0d88a53b5cb75b4f2b4a0a309a/image-17.jpg)
Chemical Potential i = io+ RTln[i ] i: describing the availability of particles just as T n n describes the availability of (internal) energy. The chemical potential is greater for molecules with more internal energy as they are more eager to dump that energy into the world as heat thereby increasing the world’s disorder). The chemical potential goes up when the concentration increases (more molecules available)

Chemical Potential A molecular species will be highly available for chemical reactions if its concentration is big or its internal energy is big.

Chemical Reactions n Biomineralization & Demineralization Ca 2+ + CO 32 - H+ 5 Ca 2+ + 3 (PO 4)3 - + OH- Ca. CO 3 ↓ Ca 5(PO 4)3 OH (HAP)↓ H+
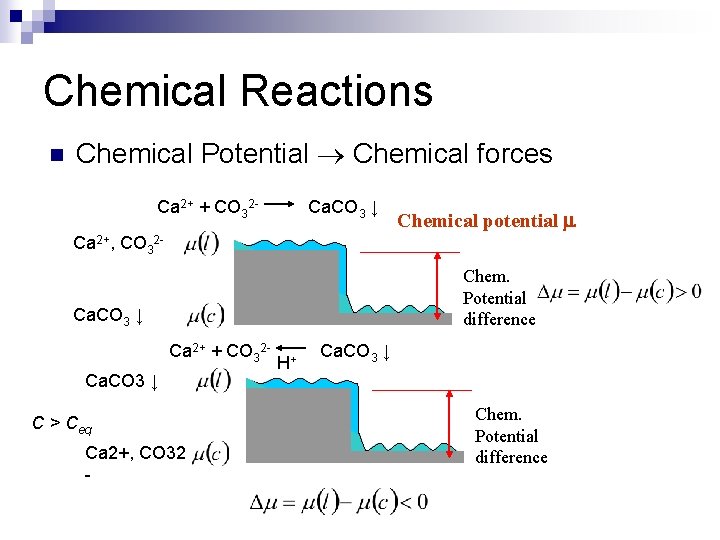
Chemical Reactions n Chemical Potential Chemical forces Ca 2+ + CO 32 - Ca. CO 3 ↓ Chemical potential Ca 2+, CO 32 Chem. Potential difference Ca. CO 3 ↓ Ca 2+ + CO 32 Ca. CO 3 ↓ C > Ceq Ca 2+, CO 32 - H+ Ca. CO 3 ↓ Chem. Potential difference

Chemical Reactions The reaction will stall when i. B = i. B or = 0. Chemical equilibrium is the point where the chemical forces balance.

Chemical Reactions n n n G gives a universal criterion for the direction of a chemical reaction. Example Ca 2+ + CO 32 - → Ca. CO 3 , after reaction, how much Ca 2+ and CO 32 -. To find the condition for equilibrium, to find the Gibbs free energy change for between the final and initial states. G = ( )fin - ( )ini G = ( Ca. CO 3) - ( Ca(2+) + CO 3(2 -)) At equilibrium, the concentrations of all species should fulfill the eqs. Let be the equilibrium constant, then p. Keq = - log Keq

Chemical Reactions n Keq: ¨ temperature and Pressure dependent ¨ It depends on the unit of concentration 2 H 2 + O 2 2 H 2 O (8. 8)

Chemical Reactions n General reactions n vk: the stoichiometric coefficients n n G-the net chemical force driving the reaction A reaction will run forward if G < 0; or backward if G > 0. n Standard free energy change:

Chemical Reactions

Dissociation: Ionic and partially ionic bonds dissociate reality in water n n Association constant Ka and Dissociation constant Kd. . When Ka > 1 (log. Ka > 0, Kd < 1, log. Kd < 0), A+ and B- tends to associate to AB. When Kd > 1 (log. Kd > 0, Ka < 1, log. Ka < 0 or p Ka > 0), AB tends to dissociate to A+ and B-. The large p. Ka, the easier the dissociation of the protein will be. A+ + B- Ka Kd AB

Dissociation: Ionic and partially ionic bonds dissociate reality in water AB Kd Ka A+ + B ni: the number of species i.

Dissociation: Ionic and partially ionic bonds dissociate reality in water At equilibrium Let Standard Specific Gibbs Free change in association

Dissociation of water n n n The dissociation of water: H 2 O H+ + OHFor pure water [H+] = [OH-] = 10 -7 M Dissociation Const: Kd = [H+][OH-]/[H 2 O]. As [H 2 O] is constant at a given T, we have then Kw = Kd[H 2 O]. Kw = [H+][OH-] = (10 -7)2. Ion product of water at room temperature ¨ ¨ p. H = -log Kw p. H = 7 -neutral p. H < 7, acidic (an acidic solution). p. H > 7, basic (a basic solution).

Dissociation: Ionic and partially ionic bonds dissociate reality in water n A measure of the energy of single charges in a particular medium is its self-energy Es-the energy of a charge in the absence of its counter ion. rs: Stoke’s radius-the radius of charge distribution n D: Dielectric const.

Dissociation: Ionic and partially ionic bonds dissociate reality in water The interior of a globule protein n Hydrophobic interior It is difficult to bury a charge in the interior of a globular protein due to the hydrophobic environment. To estimate the effect of the self energy of amino acids in solution as opposed to being buried in the interior of a protein Association constant Ka: the large p. Ka, the easier the dissociation of protein will be. Globular protein A+ + B- AB
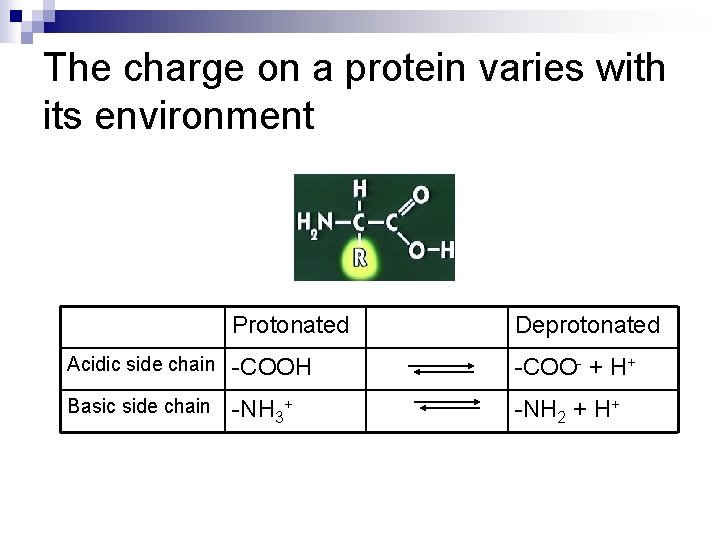
The charge on a protein varies with its environment Protonated Deprotonated Acidic side chain -COOH -COO- + H+ Basic side chain -NH 3+ -NH 2 + H+

The charge on a protein varies with its environment n Propobility of protonation

Review The measurement of the degree of disordering and the freedom n The direction of change in thermodynamic system 2 nd law n

Chapters in Textbook n Chapter 8 , in Biological Physics
- Slides: 35