BIOPHARMACEUTICS 1 Introduction to bipharmaceutics Biopharmaceutics the study

BIOPHARMACEUTICS 1

Introduction to bipharmaceutics: Biopharmaceutics: the study of how the physicochemical properties of drugs, dosage forms and routes of administeration affect the rate and extent of the drug absorption. Thus, biopharmaceutics involves factors that influence the: 1) protection and stability of the drug within the product; 2) the rate of drug release from the product; 3) the rate of dissolution of the drug at the absorption site; and 4) the availability of the drug at its site of action. 2
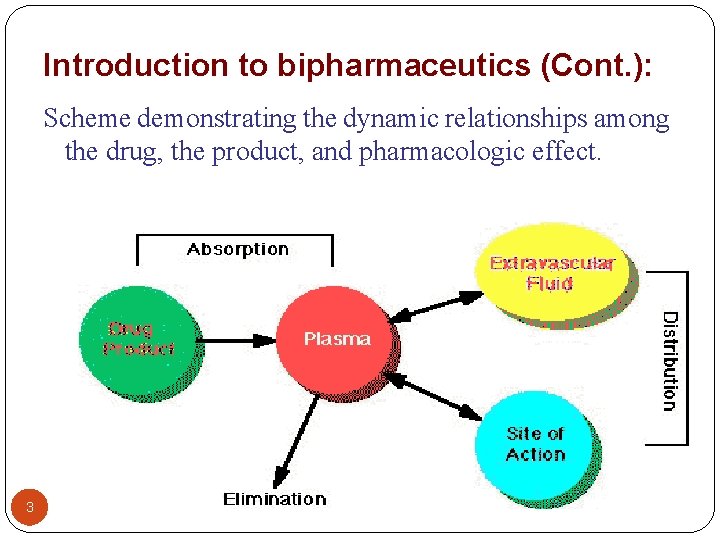
Introduction to bipharmaceutics (Cont. ): Scheme demonstrating the dynamic relationships among the drug, the product, and pharmacologic effect. 3

Introduction to bipharmaceutics (Cont. ): ADME: is an acronym in pharmacokinetics and pharmacology for absorption, distribution, metabolism, and excretion, and describes the disposition of a pharmaceutical compound within an organism. Pharmacokinetics: The study and characterization of the time course (kinetics) of drug absorption, distribution, metabolism and elimination (ADME). 4
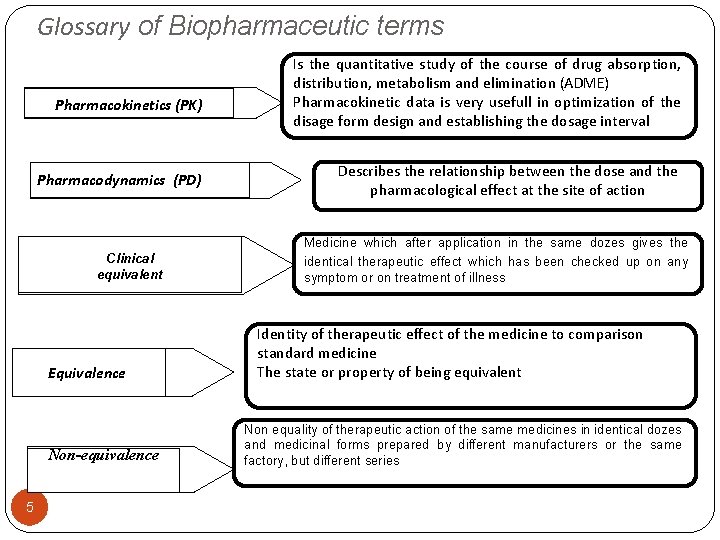
Glossary of Biopharmaceutic terms Pharmacokinetics (PK) Pharmacodynamics (PD) Clinical equivalent Equivalence Non-equivalence 5 Is the quantitative study of the course of drug absorption, distribution, metabolism and elimination (ADME) Pharmacokinetic data is very usefull in optimization of the disage form design and establishing the dosage interval Describes the relationship between the dose and the pharmacological effect at the site of action Medicine which after application in the same dozes gives the identical therapeutic effect which has been checked up on any symptom or on treatment of illness Identity of therapeutic effect of the medicine to comparison standard medicine The state or property of being equivalent Non equality of therapeutic action of the same medicines in identical dozes and medicinal forms prepared by different manufacturers or the same factory, but different series
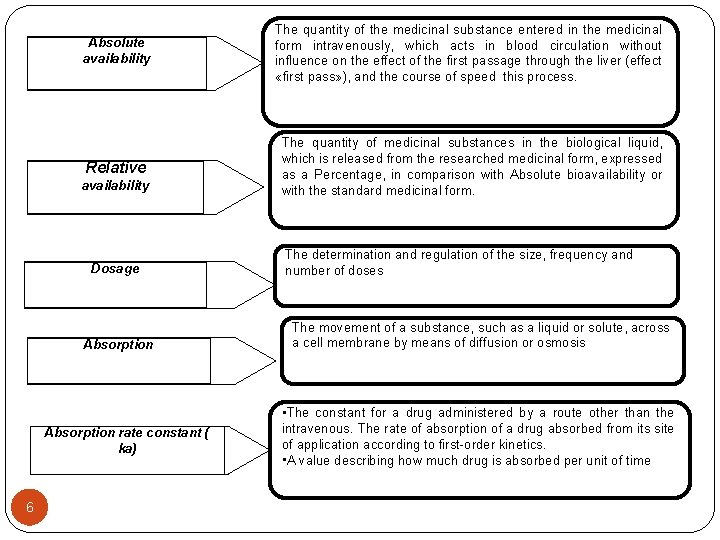
Absolute availability The quantity of the medicinal substance entered in the medicinal form intravenously, which acts in blood circulation without influence on the effect of the first passage through the liver (effect «first pass» ), and the course of speed this process. Relative The quantity of medicinal substances in the biological liquid, which is released from the researched medicinal form, expressed as a Percentage, in comparison with Absolute bioavailability or with the standard medicinal form. availability Dosage Absorption rate constant ( ka) 6 The determination and regulation of the size, frequency and number of doses The movement of a substance, such as a liquid or solute, across a cell membrane by means of diffusion or osmosis • The constant for a drug administered by a route other than the intravenous. The rate of absorption of a drug absorbed from its site of application according to first-order kinetics. • A value describing how much drug is absorbed per unit of time
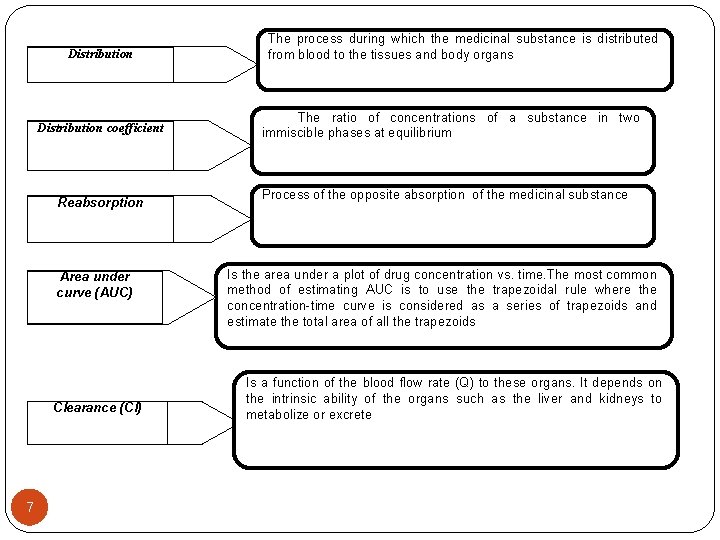
Distribution coefficient Reabsorption Area under curve (AUC) Clearance (Cl) 7 The process during which the medicinal substance is distributed from blood to the tissues and body organs The ratio of concentrations of a substance in two immiscible phases at equilibrium Process of the opposite absorption of the medicinal substance Is the area under a plot of drug concentration vs. time. The most common method of estimating AUC is to use the trapezoidal rule where the concentration-time curve is considered as a series of trapezoids and estimate the total area of all the trapezoids Is a function of the blood flow rate (Q) to these organs. It depends on the intrinsic ability of the organs such as the liver and kidneys to metabolize or excrete
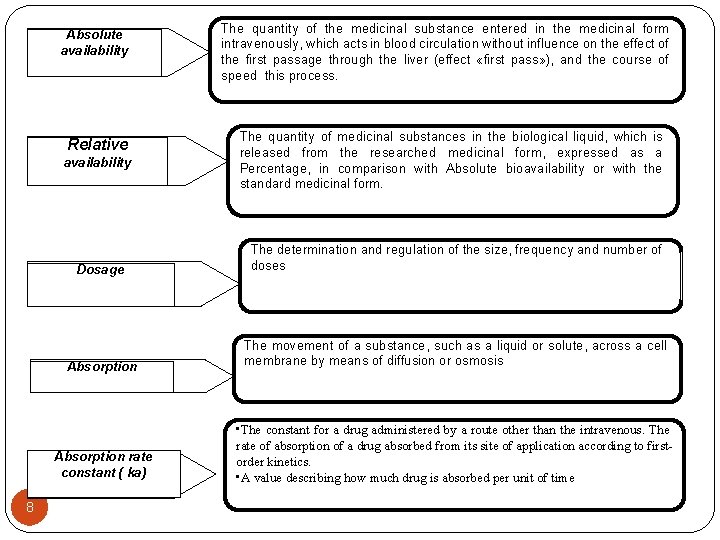
Absolute availability Relative availability 8 The quantity of the medicinal substance entered in the medicinal form intravenously, which acts in blood circulation without influence on the effect of the first passage through the liver (effect «first pass» ), and the course of speed this process. The quantity of medicinal substances in the biological liquid, which is released from the researched medicinal form, expressed as a Percentage, in comparison with Absolute bioavailability or with the standard medicinal form. Dosage The determination and regulation of the size, frequency and number of doses Absorption The movement of a substance, such as a liquid or solute, across a cell membrane by means of diffusion or osmosis Absorption rate constant ( ka) • The constant for a drug administered by a route other than the intravenous. The rate of absorption of a drug absorbed from its site of application according to firstorder kinetics. • A value describing how much drug is absorbed per unit of time
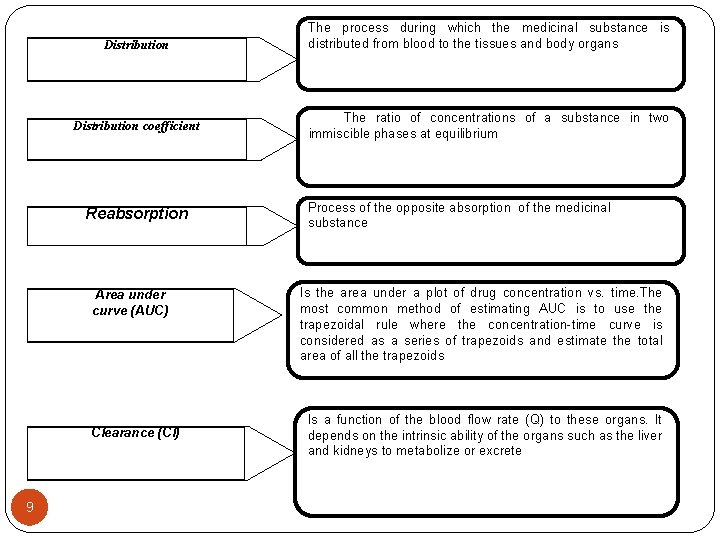
Distribution coefficient Reabsorption Area under curve (AUC) Clearance (Cl) 9 The process during which the medicinal substance is distributed from blood to the tissues and body organs The ratio of concentrations of a substance in two immiscible phases at equilibrium Process of the opposite absorption of the medicinal substance Is the area under a plot of drug concentration vs. time. The most common method of estimating AUC is to use the trapezoidal rule where the concentration-time curve is considered as a series of trapezoids and estimate the total area of all the trapezoids Is a function of the blood flow rate (Q) to these organs. It depends on the intrinsic ability of the organs such as the liver and kidneys to metabolize or excrete
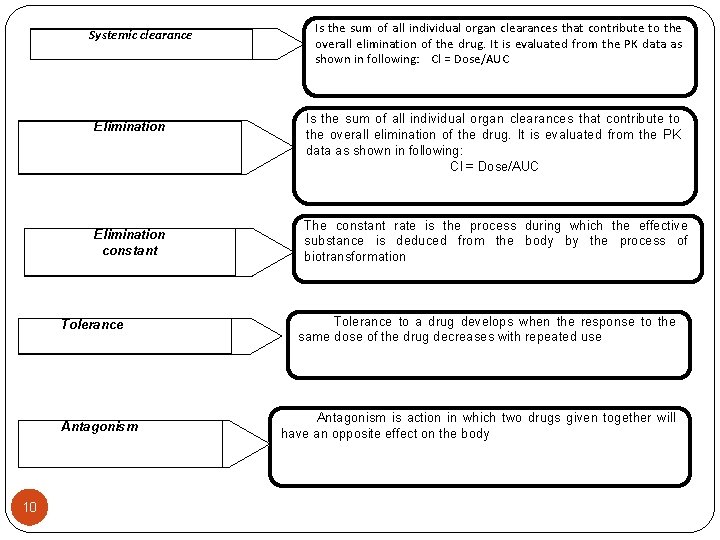
Systemic clearance Elimination constant Tolerance Antagonism 10 Is the sum of all individual organ clearances that contribute to the overall elimination of the drug. It is evaluated from the PK data as shown in following: Cl = Dose/AUC The constant rate is the process during which the effective substance is deduced from the body by the process of biotransformation Tolerance to a drug develops when the response to the same dose of the drug decreases with repeated use Antagonism is action in which two drugs given together will have an opposite effect on the body

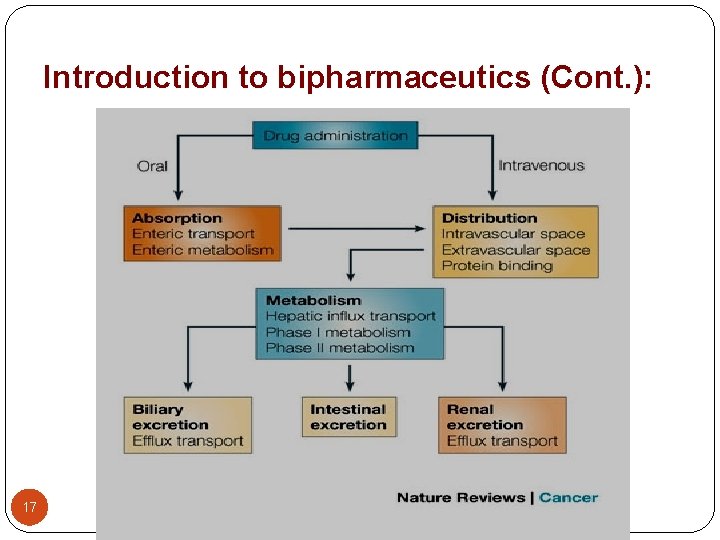
ADME Process Blood concentration of a drug are the result of four simultaneously occurring processes: Absorption Distribution Metabolism Excretion Besides the ADME process, an important factor of drug concentration is how drugs move through biological membranes by diffusion. 11

Absorption Once a drug is released from its dosage formulation, the process that transfers it into the blood is called absorption. One of the primary factors affecting oral drug absorption is the gastric emptying time. This the time that the drug remains in the stomach before it is emptied into the small intestine Most absorption occurs in the small intestine. Some factors increase the gastric emptying time, but most slow it. If a drug remains in the stomach too long, it can be degraded or destroyed, and its effect decreased. 12

Absorption Gastric emptying time can be affected by: Amount and type of food in the stomach The presence of other drugs The person’s body position The person’s emotional state 13

Distribution Blood carries the drug throughout the body and to its sites of action. Drugs are rapidly distributed to organs having high blood flow rates such as the heart, liver and kidneys. Distribution to the muscle, fat, and skin is slower because they have lower blood flow rates. 14

Metabolism Drug metabolism refers to the body’s process of transforming drugs. The transformed drug is called a metabolite. The primary site of drug metabolism is in the liver. When transformed in the liver a drug is broken down into inactive or active molecules. Inactive are excreted through the kidneys and active produce effects and excreted later. 15

Excretion Most drugs are excreted in the urine by the kidneys. The functional unit of the kidney is the nephron. Some oral drugs that are difficult to break down can be excreted in the feces (poop). The job of the kidney is to filter the blood and remove waste products. 16
Introduction to bipharmaceutics (Cont. ): 17
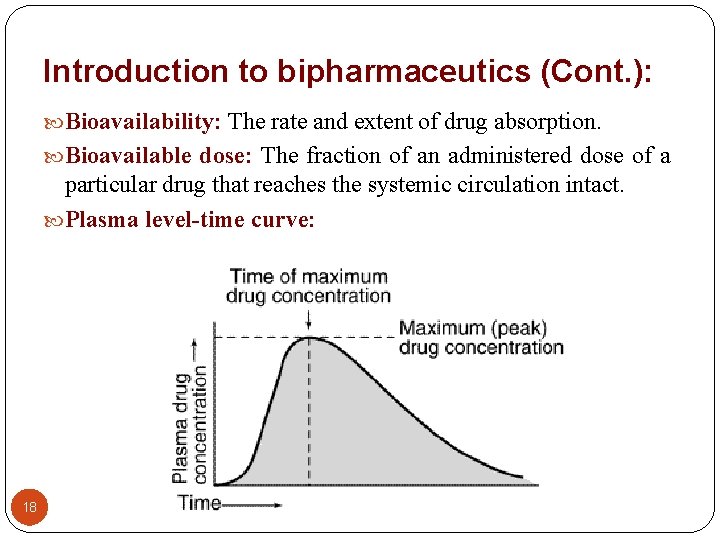
Introduction to bipharmaceutics (Cont. ): Bioavailability: The rate and extent of drug absorption. Bioavailable dose: The fraction of an administered dose of a particular drug that reaches the systemic circulation intact. Plasma level-time curve: 18

Introduction to bipharmaceutics (Cont. ): The plasma level-time curve is generated by measuring the drug concentration in plasma samples taken at various time intervals after a drug product is administered. The concentration of drug in each plasma sample is plotted against the corresponding time at which the plasma sample was removed. 19
Introduction to bipharmaceutics (Cont. ): 20

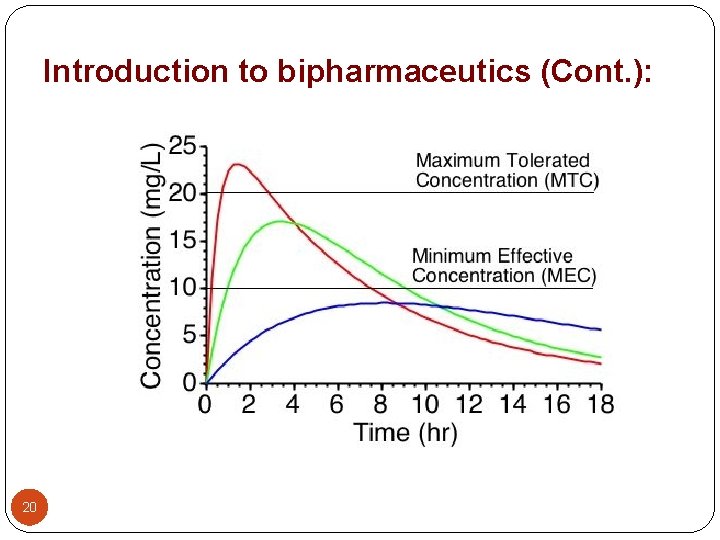
Introduction to bipharmaceutics (Cont. ): Drug Product Performance Parameters: 1 - Minimum effective concentration (MEC): The minimum concentration of drug needed at the receptors to produce the desired pharmacologic effect. 2 - Minimum toxic concentration (MTC): The drug concentration needed to just produce a toxic effect. 3 - Onset time: The time required for the drug to reach the MEC. 4 - Duration of action: The difference between the onset time and the time for the drug to decline back to the MEC. 21

Introduction to bipharmaceutics (Cont. ): 5 - The time of peak plasma level: The time of maximum drug concentration in the plasma and is proportional to the rate of drug absorption. 6 - The peak plasma level: The maximum drug concentration, usually related to the dose and the rate constants for absorption and elimination of the drug. 7 - Area under the curve: It is related to the amount of drug absorbed systemically. 22
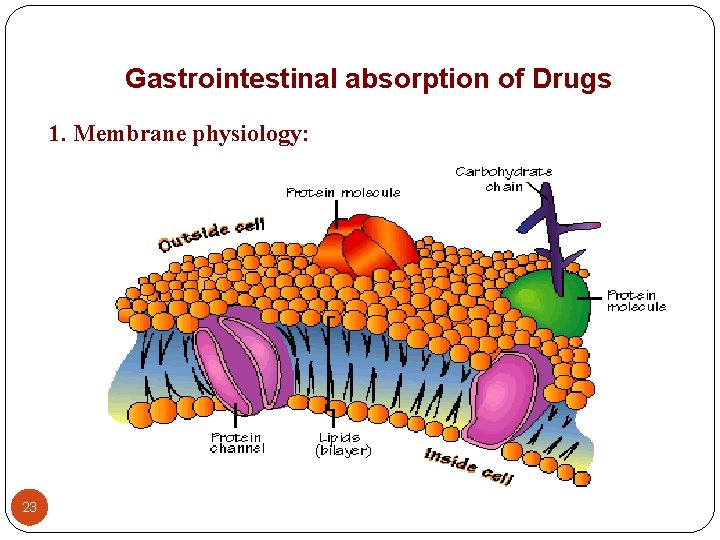
Gastrointestinal absorption of Drugs 1. Membrane physiology: 23

Membrane physiology (Cont. ): - The cell membrane is the barrier that separates the - 24 inside of the cell from the outside. The cell membrane is made up of phospholipids, proteins, and other macromolecules. The phosopholipids make up a bilayer. It contains hydrophilic and hydrophobic molecules. The proteins in the cell membrane are located within the phospholipid bilayer. So, the biologic membrane is mainly lipid in nature but contains small aqueous channels or pores.
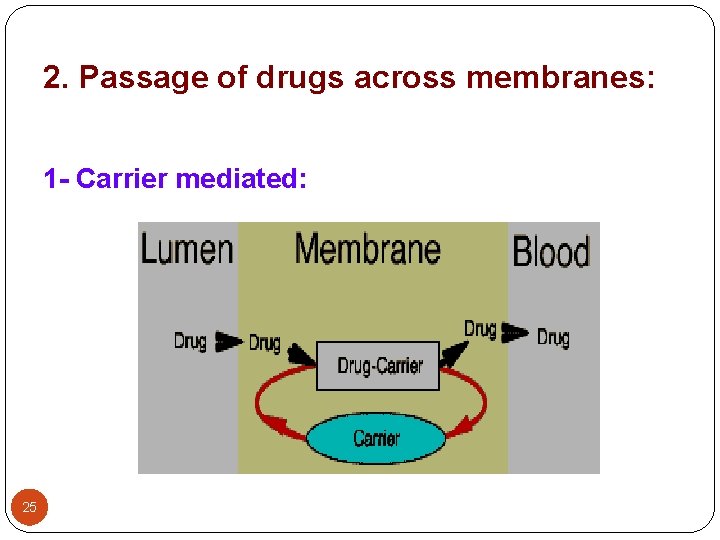
2. Passage of drugs across membranes: 1 - Carrier mediated: 25
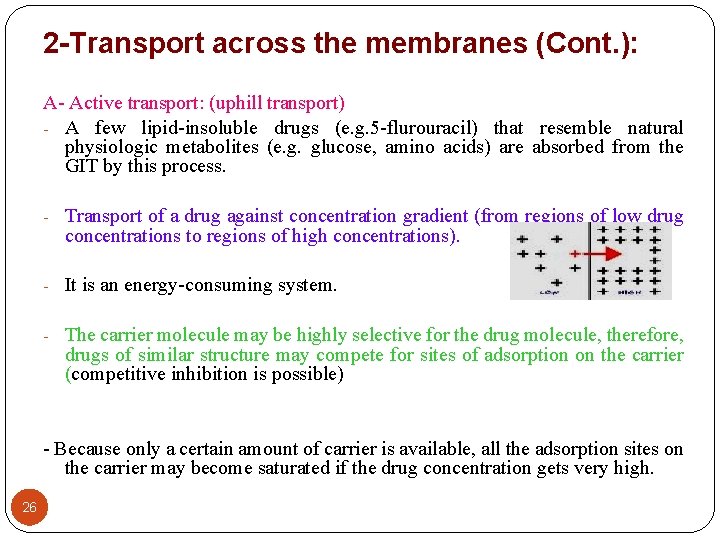
2 -Transport across the membranes (Cont. ): A- Active transport: (uphill transport) - A few lipid-insoluble drugs (e. g. 5 -flurouracil) that resemble natural physiologic metabolites (e. g. glucose, amino acids) are absorbed from the GIT by this process. - Transport of a drug against concentration gradient (from regions of low drug concentrations to regions of high concentrations). - It is an energy-consuming system. - The carrier molecule may be highly selective for the drug molecule, therefore, drugs of similar structure may compete for sites of adsorption on the carrier (competitive inhibition is possible) - Because only a certain amount of carrier is available, all the adsorption sites on the carrier may become saturated if the drug concentration gets very high. 26
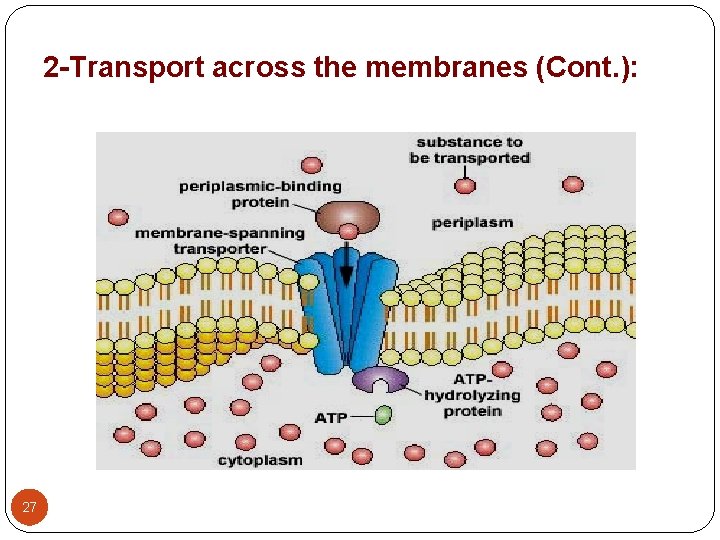
2 -Transport across the membranes (Cont. ): 27

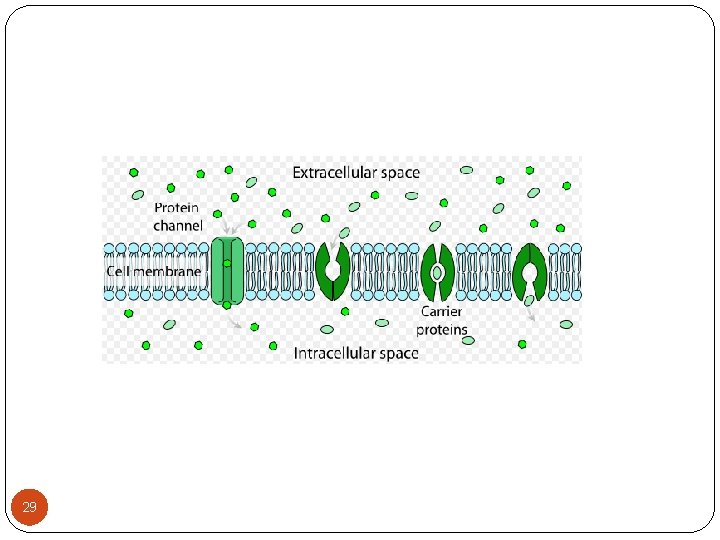
2 -Transport across the membranes (Cont. ): B- Facilitated diffusion: (downhill transport) - Play a very minor role in absorption. - A drug carrier is required but no energy is necessary. e. g. vitamin B 12 transport. - Saturable if not enough carrier and structurally selective for the drug and shows competition kinetics for drugs of similar structure. - No transport against a concentration gradient only downhill but faster. 28
29

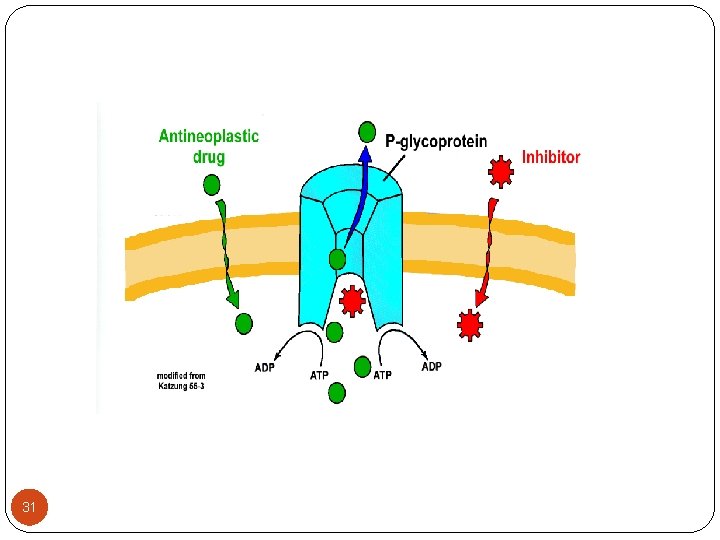
2 -Transport across the membranes (Cont. ): C- P-glycoprotein: (permeability glycoprotein) - P-glycoprotein transporters (PGP) are present throughout the body including liver, brain, kidney and the intestinal tract epithelia. - Act as reverse pump generally inhibiting absorption. - This is an active, ATP-dependent process. 30
31

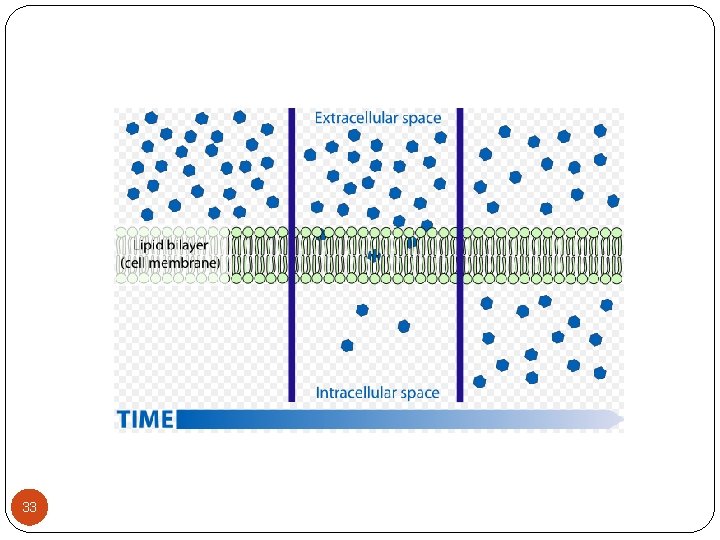
2 -Transport across the membranes (Cont. ): 2 - Passive diffusion: - Most drugs cross biologic membranes by passive diffusion. - Diffusion occurs when the drug concentration on one side of the membrane is higher than that on the other side. - The process is passive because no external energy is expended. - The driving force for passive diffusion is the difference in drug concentrations on either side of the cell membrane. 32
33
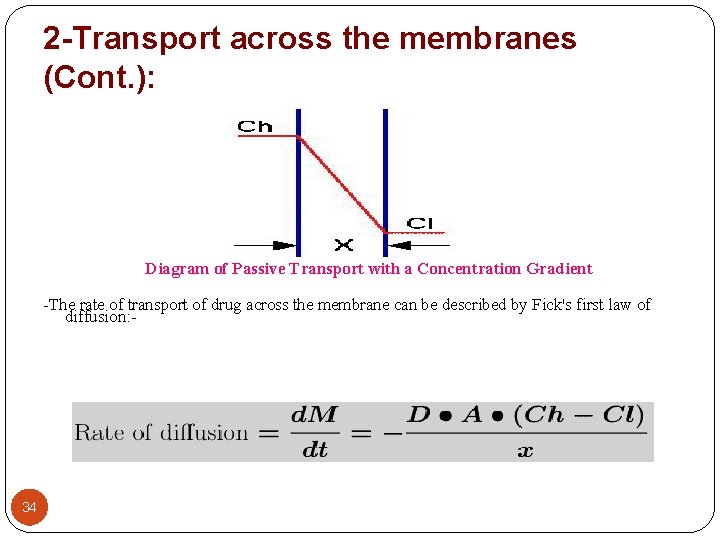
2 -Transport across the membranes (Cont. ): Diagram of Passive Transport with a Concentration Gradient -The rate of transport of drug across the membrane can be described by Fick's first law of diffusion: - Fick's First Law, Rate of Diffusion 34

2 -Transport across the membranes (Cont. ): The parameters of this equation are: - D: diffusion coefficient. This parameter is related to the size and lipid solubility of the drug and the viscosity of the diffusion medium. As lipid solubility increases or molecular size decreases then D increases and thus d. M/dt also increases. 35 A: surface area. As the surface area increases the rate of diffusion also increase. The surface of the intestinal lining (with villae and microvillae) is much larger than the stomach. This is one reason absorption is generally faster from the intestine compared with absorption from the stomach.

2 -Transport across the membranes (Cont. ): x: membrane thickness. The smaller the membrane thickness the quicker the diffusion process. As one example, the membrane in the lung is quite thin thus inhalation absorption can be quite rapid. (Ch -Cl): concentration difference. The drug concentration in blood or plasma will be quite low compared with the concentration in the GI tract. It is this concentration gradient which allows the rapid complete absorption of many drug substances. Normally Cl << Ch then: - 36
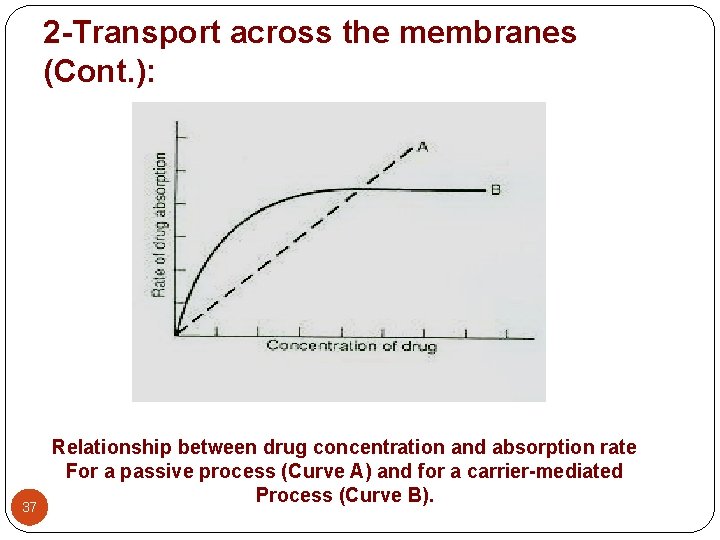
2 -Transport across the membranes (Cont. ): 37 Relationship between drug concentration and absorption rate For a passive process (Curve A) and for a carrier-mediated Process (Curve B).
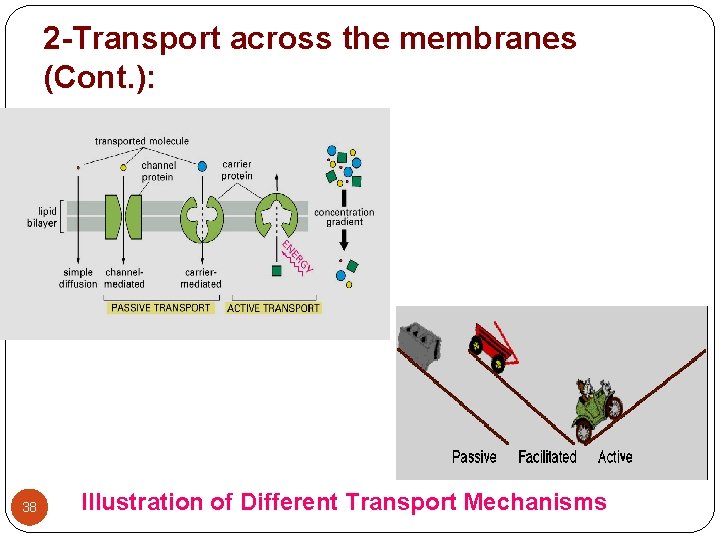
2 -Transport across the membranes (Cont. ): 38 Illustration of Different Transport Mechanisms

2 -Transport across the membranes (Cont. ): 3 - Vesicular transport: (endocytosis) - It is the process of engulfing particles or dissolved materials by the cell. - Pinocytosis and phagocytosis are forms of vesicular transport that differ by the type of material ingested. Pinocytosis: refers to the engulfment of small molecules or fluid. Phagocytosis: refers to the engulfment of larger particles or macromolecules. - During pinocytosis or phagocytosis, the cell membrane invaginates to surround the material, and then engulfs the material into the cell. Subsequently, the cell membrane containing the material forms a vesicle or vacuole within the cell. - Vesicular transport is the proposed process for the absorption of Vitamin A, D, E, and K, peptides in new born. 39
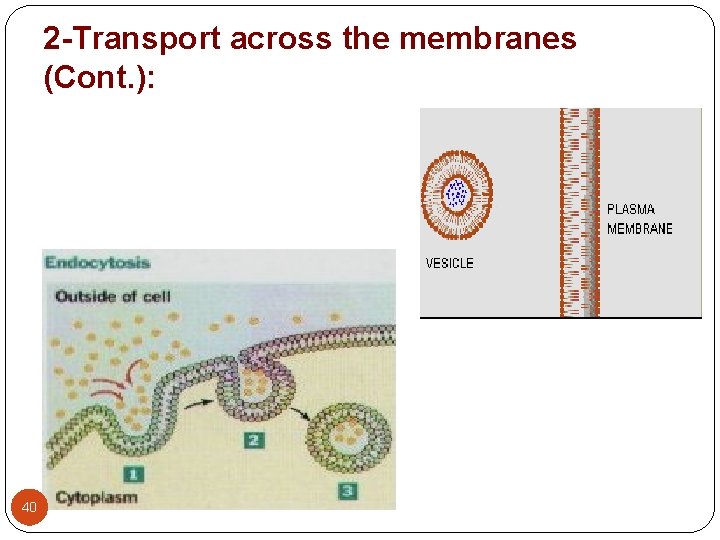
2 -Transport across the membranes (Cont. ): 40

2 -Transport across the membranes (Cont. ): 4 - Pore (convective) transport: - A certain type of protein called transport protein may form an open channel across the lipid membrane of the cell. - Very small molecules, such as urea, water and sugars are able to rapidly cross the cell membrane through these pores. 5 - Ion pair formation: - Strong electrolyte drugs are highly ionized or charged molecules, such as quaternary nitrogen compounds. - These drugs penetrate membranes poorly. When linked up with an oppositely charged ion, an ion pair is formed in which the overall charge of the pair is neutral. This neutral complex diffuses more easily across the membrane. - e. g. the formation of an ion pair for propranolol (basic drug) with oleic acid. 41
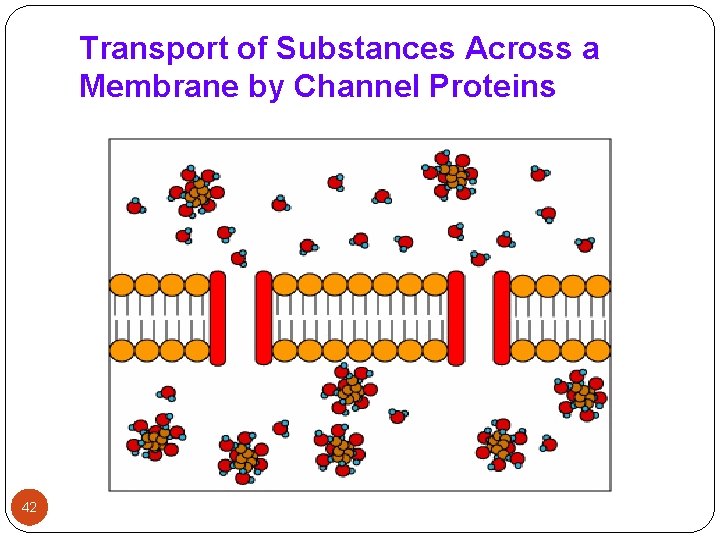
Transport of Substances Across a Membrane by Channel Proteins 42
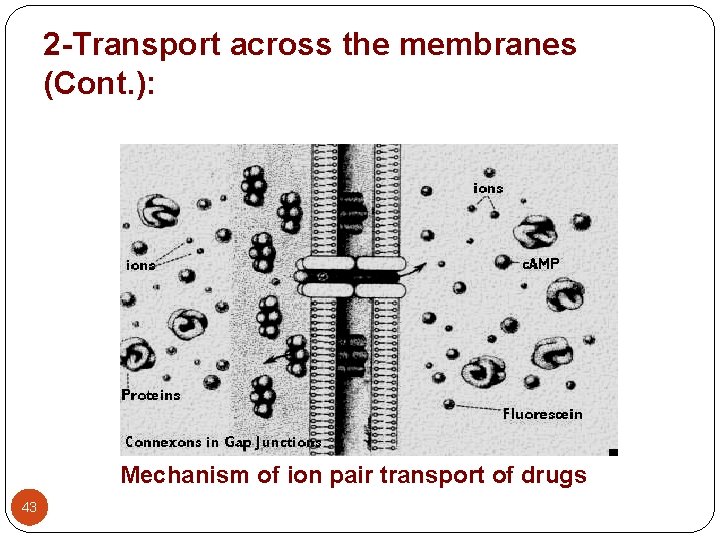
2 -Transport across the membranes (Cont. ): Mechanism of ion pair transport of drugs 43

Thank You 44
- Slides: 44