Biopharma IP Issues in China NJIPLA NJCCA Joint

Biopharma IP Issues in China NJIPLA & NJCCA Joint Conference Hyatt Regency, New Brunswick, NJ Yong Zhao Merck IP Group May 9, 2013

Disclaimer Not views of Merck 2

Current IP Environment in China § IP Protection in China § Chinese Government’s 12 th Five Year Plan: Transform Chinese economy from made-in-China to created-in. China; build up Biopharma ecosystem with the following goals: Ø 30 NCEs in 5 years Ø Improve R&D output (No. 3 in patents; 300, 000 scientists, …) Ø Improve IP system, shorten patent exam period, incentivize employees and companies Ø Cooperate with foreign entities and talents, absorb foreign technology and re-innovate 3
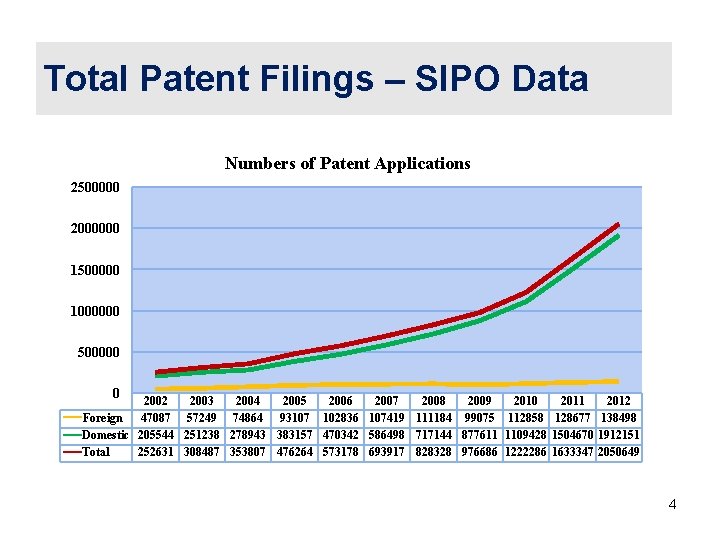
Total Patent Filings – SIPO Data Numbers of Patent Applications 2500000 2000000 1500000 1000000 500000 0 2002 2003 2004 2005 2006 2007 2008 2009 2010 2011 2012 Foreign 47087 57249 74864 93107 102836 107419 111184 99075 112858 128677 138498 Domestic 205544 251238 278943 383157 470342 586498 717144 877611 1109428 1504670 1912151 Total 252631 308487 353807 476264 573178 693917 828328 976686 1222286 1633347 2050649 4
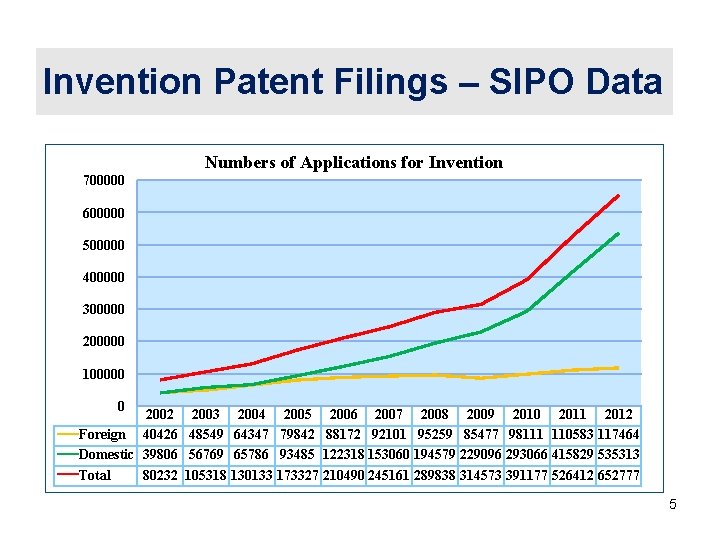
Invention Patent Filings – SIPO Data Numbers of Applications for Invention 700000 600000 500000 400000 300000 200000 100000 0 2002 2003 2004 2005 2006 2007 2008 2009 2010 2011 2012 Foreign 40426 48549 64347 79842 88172 92101 95259 85477 98111 110583 117464 Domestic 39806 56769 65786 93485 122318 153060 194579 229096 293066 415829 535313 Source: SIPO Total 80232 105318 130133 173327 210490 245161 289838 314573 391177 526412 652777 5
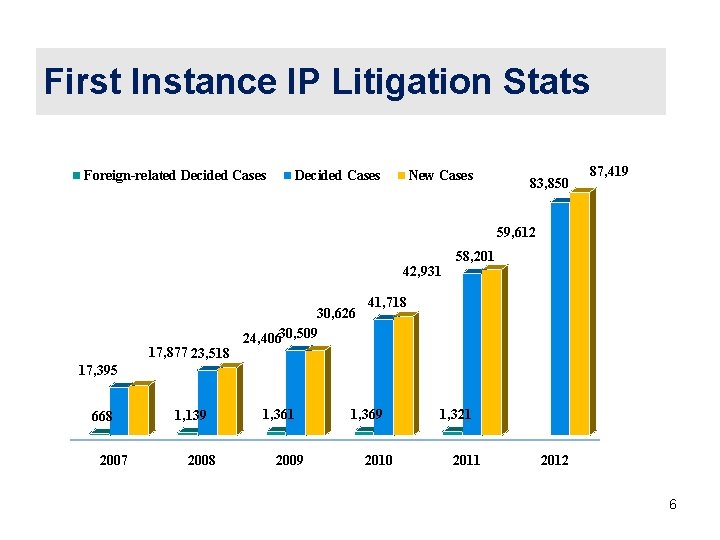
First Instance IP Litigation Stats Foreign-related Decided Cases New Cases 83, 850 87, 419 59, 612 42, 931 17, 877 23, 518 30, 626 24, 40630, 509 58, 201 41, 718 17, 395 668 2007 1, 139 2008 1, 361 2009 1, 369 2010 1, 321 2012 6

IP Issues Related to Collaborations with Chinese Partners 7

Collaborating with Chinese Partners § Drivers for Biopharma contracts in China: Ø Generating returns with low cost Ø Accessing local talents and expertise Ø Market access and penetration Ø Leveraging partner’s familiarity and experience with Chinese Government » Seeking new pipeline and innovation » Desire to improve Biopharma R&D prowess 8

Collaborating with Chinese Partners § Typical Standard Form MNC Biopharma Contract: Ø Arbitration and/or choice of law and venue provisions (e. g. , designating a US state law and a US court) Ø Grant-back of IP rights to improvements Ø Standard warrantee/indemnification clauses Ø Written in English Potential issues when used for China? 9

Legal Framework for Technology Contract § Relevant Chinese Laws Include: Ø Contract Law (1999) Ø Technology Import and Export Regulation (2002) Ø Foreign Trade Law (2004) Ø Antimonopoly Law (2008) Ø Patent Law, 3 rd Amendment (2009) Ø Supreme People’s Court’s (SPC) Judicial Interpretation on Technology Contract Disputes (2005) 10

Consequences of Violating Relevant Laws § Any contract that violates mandatory Chinese laws or regulations is null and void (partial or full invalidity) (Art. 52 of Contract Law) Ø Difficult to circumvent Art. 52 v Chinese courts are unlikely to enforce such contracts v Public policy is to curb anti-competitive provisions 11

Technology Import & Export Regulation § Scope of the Regulation Ø Covers assignments of patent rights, patent licensing, collaboration contracts, exchange of data, etc. Ø Technology import/export amounts to foreign trade and requires foreign trade license § Three categories of technologies under the Regulation Ø Prohibited, Restricted & Permitted § Most pharmaceuticals fall into Permitted category Ø Registration of contract is required for Permitted tech contract Ø Registration may also have litigation implications 12

Common Pitfalls in Biopharma Contracts § Limiting counterparty from making improvements to a transferred technology § Anti-competitive Provisions Ø Prohibiting counterparty from challenging validity of patent; Ø Royalties for patent beyond patent term; Ø Imposing conditions unnecessary for utilizing the transferred technology Any technology contract which illegally monopolizes technologies, impedes technological progress, or infringes upon technological achievement of others shall be void. (Art. 329 of Contract Law) 13

Common Pitfalls in Biopharma Contracts § IP Ownership on improvements made by counterparty requires payment Ø Art. 354 of Contract Law: Improvement is owned by the party who developed it unless specifically stipulated in the contract Ø Art. 10 of SPC Interpretation on Technology Disputes: Improvement should be paid for otherwise tech contract is void v Technology contract should make it explicit that improvements have been paid for 14

Common Pitfalls in Biopharma Contracts § Have proper IP warranty provisions in contract Ø Non-infringement warranty – under default rule, Licensor is liable for 3 rd party infringement claim v Licensor should specify a limit for total liability as Chinese law does not have a cap on such liability Ø Under default rule, Licensor is liable if the licensed technology is not correct, complete, effective or cannot reach the specified technological target 15

Re-visit Standard Form Contract § Standard Form Contract May Not Be Effective Ø Grant-back of IP rights to improvements – needs payment Ø Standard warrantee clauses – may lack IP warrantee Ø Arbitration and/or choice of law and venue provisions - may be ineffective Ø Written in English – needs Chinese translation before dispute arises v Chinese courts often consider issues according to relevant Chinese laws regardless of a foreign court’s decision or choice of law provisions in contract 16

Collaborating with Chinese Universities § Identify the right legal entity as contractual party § Direct government funding may complicate IP Ownership § Address scope of collaboration and avoid misuse of transferred technology § Employee has pre-emptive right to acquire technology within 1 year after leaving Univ. (Art. 326 of Contract Law) § If joint ownership, provide manner of exploitation in agreement (Art. 15 of 3 rd Amendment) 17

Biopharma Patent Procurement Issues in China 18

Patent Prosecution – First Filing Requirement § Where any entity or individual intends to file an application in a foreign county for a patent for an invention … made in China, it or he shall first apply for a security examination. (Art. 20, 3 rd Amendment) § Failure to comply is ground for rejection/invalidation § Three options before filing outside China: Ø File security exam request (in Chinese) Ø File Chinese national application (in Chinese) Ø File PCT (can be in English) in SIPO as RO 19
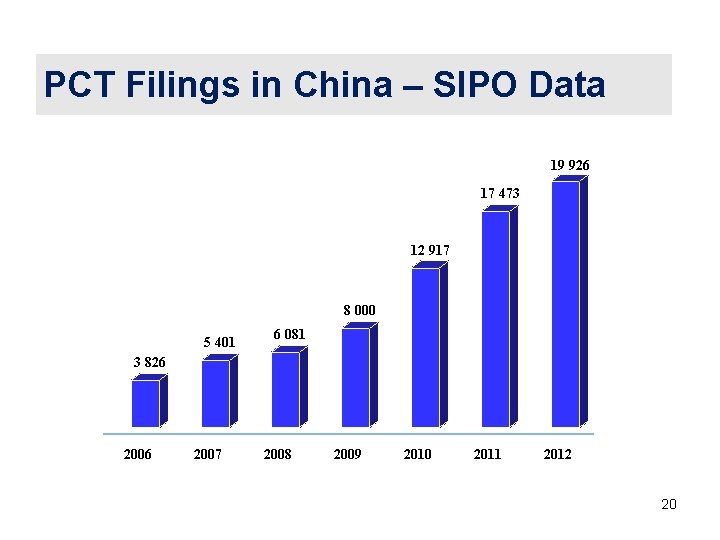
PCT Filings in China – SIPO Data 19 926 17 473 12 917 8 000 5 401 6 081 3 826 2007 2008 2009 2010 2011 2012 20

Inventor Reward & Remuneration Relevant Patent Laws and Regulations § An employer shall reward and remunerate its employeeinventors of a service invention (Art. 16, 3 rd Amendment) § Implementing Regulation to the 3 rd Amendment Ø Rule 76. An employer may enter into an agreement with its inventor, or provide in its policy formulated according to relevant laws, on the manner and amount of reward and remuneration. Ø Rule 77. In the absence of an agmt or a policy, the reward must be at least RMB 3, 000 for an invention patent. Ø Rule 78. In the absence of an agmt or a policy, the remuneration must be at least 2% of profit upon exploitation of an invention patent; or at least 10% income from patent license. 21

Inventor Reward & Remuneration § SIPO’s Draft Regulation on Service Inventions creates more questions than answers Ø Lacks clarity on “reasonableness” standard Ø IP rights reversal to employees and first refusal rights Ø “Technical secrets” as basis for reward and remuneration § Compliance Strategies Ø Have a reasonable company policy / IP agreement in place to avoid default Rules Ø Consider “reasonableness” factors when formulating policy Ø Comply with procedural requirements when implementing policy Ø Obtain informed waivers from employees 22

Patent Prosecution – Amendment § Limited flexibility in making amendments during prosecution Ø In general, voluntary amendments are not allowed after three months from the substantive examination stage. § Even less amendment flexibility during invalidation Ø Adding elements not present in the granted claims is not allowed 23

Patent Prosecution – Data Sufficiency The Law: The description shall set forth the invention … in a manner sufficiently clear and complete so as to enable a person skilled in the … technology to carry it out; . . . (Art. 26. 3, 3 rd Amendment) § Disclosure of the following required for NCE application Ø Chemical identification; Ø Preparation; and Ø Use and/or effect (biological activity data). v Post filing submission of data for enablement is not allowed 24

Patent Prosecution – Data Sufficiency Implications of SIPO’s 26. 3 practice § Lack of biological activity data is one of the major reasons for rejections in NCE applications § SIPO’s practice is inconsistent with other IP 5 jurisdictions despite similar laws § Current examination guidelines (effective 2006) retroactively applicable to patents granted pre-2006? § Implications – increased risk of invalidations? 25

Patent Invalidations at PRB § Invalidation proceedings occur at Patent Re-examination Board (PRB) of SIPO Ø Overall, about 50% invalidated and 50% maintained § About 20% of PRB decisions were appealed to courts Ø About 75% affirmed and 25% reversed § Straw man as invalidation requesting party Ø Potential misuse especially in light of 26. 3 practice and weak patent linkage system 26

Biopharma Patent Enforcement Issues in China 27

IP Litigation Headline Stats § Overall plaintiff win rate: 84%; foreign plaintiff win rate: 88% (Based on published > 15, 000 IP cases between 2006 – 2011) Ø China’s Current IP enforcement landscape is generally plaintiff-friendly § Average damage awards Ø Invention/utility model patent: ~ RMB 193, 830 (~ $30, 000) Ø Design patent: ~ RMB 56, 264 (~ $9, 000) Ø Trademark: ~ RMB 100, 275 (~ $15, 000) Ø Unfair competition: ~ RMB 94, 546 (~ $15, 000) Ø Copyright: ~ RMB 30, 034 (~ $5, 000) § Average time to get a first instance trial: 5. 3 months Source: Rouse & Co. & www. ciela. cn 28
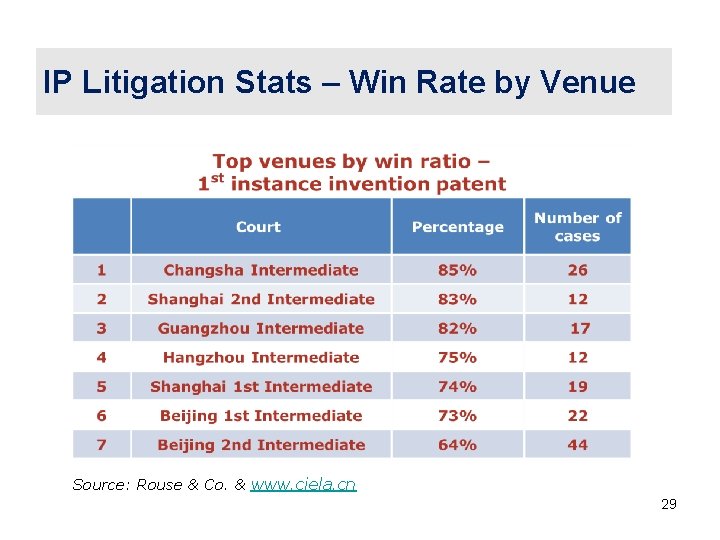
IP Litigation Stats – Win Rate by Venue Source: Rouse & Co. & www. ciela. cn 29

Current IP Enforcement Environment § Beyond the headlines, IP enforcement in China remains a challenge, especially for Biopharma patents Ø Biopharma patent plaintiff win rate ~50% (process cases ~20%) Ø PI in Biopharma patent cases rarely issued Ø No discovery - hard to prove infringement and/or damages Ø Statutory damages low (< ~ $150, 000; Art. 65 of 3 rd Amend. ) 30

Weak Patent Linkage System § In China, all drugs must be approved by State Food and Drug Administration (“SFDA”) before marketing § The Law on “patent linkage”: An applicant shall provide the information on patent and its ownership … in respect of the drug applied for registration, … Where another party owns the patent in China, the applicant shall provide a statement of noninfringement. The drug regulatory department shall publish the information … on its official website. (Art. 18, Measures for the Administration of Drug Registration (2007); SFDA’s official website: www. sfda. gov. cn) § Generic applicants can file for generic drug application only within two years of patent expiry (Art. 19, Measures (2007)) 31

Weak Patent Linkage System § Arts. 18 & 19 in Action Ø SFDA’s patent database lacks accuracy and transparency Ø No automatic stay upon filing of infringement suit by NDA holder Ø Generics often provide non-infringement statement in disregard of valid patents with little to no consequences Ø Secondary patents generally ineffective in preventing SFDA approvals (silver lining: compound patents effective) § No Orange Book type patent linkage system in China § Efforts to improve patent linkage system 32

Being Proactive and Working with SFDA § Preventing generic’s SFDA approval is the best option Ø Being proactive before approval and working with SFDA is key § Infringement action against generic after SFDA approval Ø Plaintiff’s win rate ~ 50%; permanent injunction generally granted § SFDA does not automatically revoke a drug approval even infringement is found Ø Should seek a court order to cancel drug approval as a remedy in each infringement action 33

Conclusions § The pending 4 th Amendment and SPC’s new Judicial Guidance may improve IP enforcement in China § Policy developments on improving patent linkage system, if successful, may enhance Biopharma IP environment § Developments in inventor reward and remuneration and 26. 3 issues counter the current overall positive direction § Biopharma R&D in China requires an IP strategy for compliance 34

History of Chinese Patent Laws § 1984: First Patent Law, effective April 1, 1985 § 1992: First Amendment, to implement Sino-US IPR Memorandum of Understanding; added pharmaceutical compounds as patentable subject matter § 2000: Second Amendment, to comply with TRIPS Agmt § 2008: Third Amendment, driven by internal considerations § 2012: Fourth Amendment, seeking public comments 35
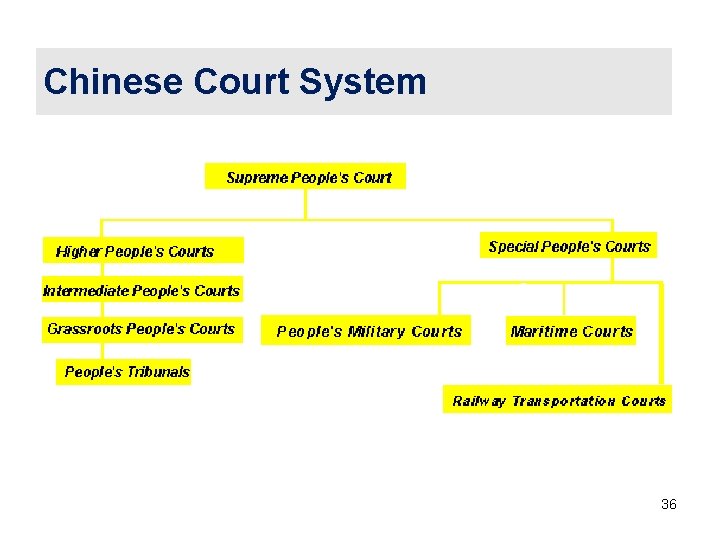
Chinese Court System 36
- Slides: 36