Biomolecules Carbon Compounds The Element Carbon back side

Biomolecules: Carbon Compounds

The Element Carbon (back side) v. Carbon is the most abundant element found in living things. v. Carbon has 4 valence electrons which enable it to form strong covalent bonds with other atoms. v. It can bond with other carbon atoms to form rings and very long chains which can be twisted and folded into millions of different, very large, and complex shapes.
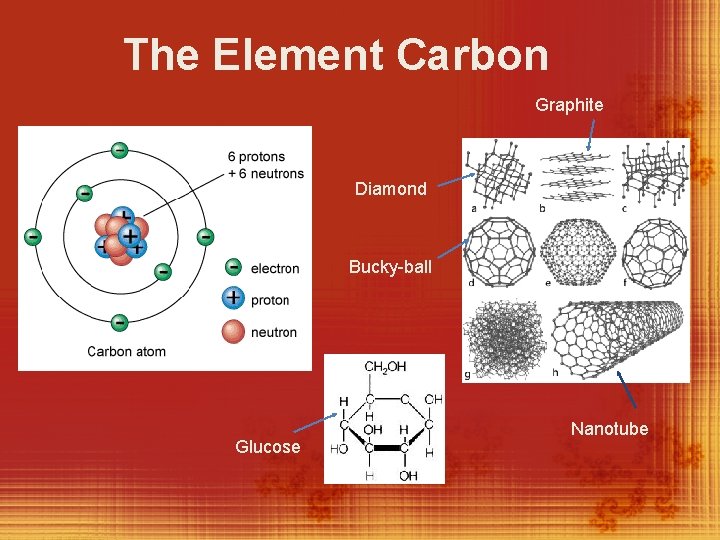
The Element Carbon Graphite Diamond Bucky-ball Glucose Nanotube

Macromolecules (back of book) v. All living things are made up carbon and a combination of other elements referred to as biomolecules § Biomolecules are very large molecules called macromolecules. § Each macromolecule is made up of small individual units called monomers §The macromolecules are formed by a process known as polymerization.
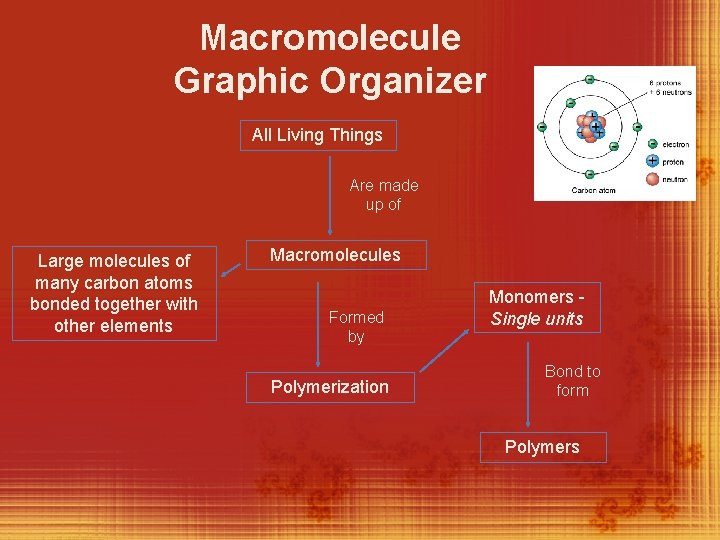
Macromolecule Graphic Organizer All Living Things Are made up of Large molecules of many carbon atoms bonded together with other elements Macromolecules Formed by Polymerization Monomers Single units Bond to form Polymers

Four Groups of Organic Compounds v Biomolecules are also known as organic compounds. v There are four groups of organic compounds found in living things: § § Carbohydrates Lipids Nucleic Acids Proteins
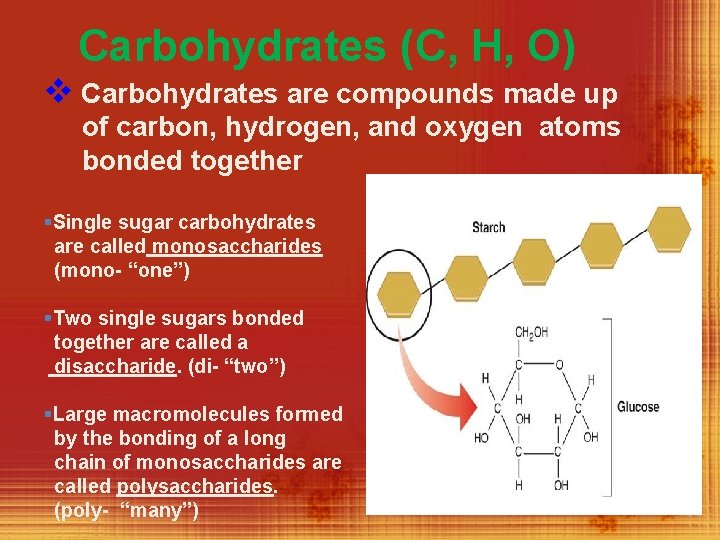
Carbohydrates (C, H, O) v Carbohydrates are compounds made up of carbon, hydrogen, and oxygen atoms bonded together §Single sugar carbohydrates are called monosaccharides (mono- “one”) §Two single sugars bonded together are called a disaccharide. (di- “two”) §Large macromolecules formed by the bonding of a long chain of monosaccharides are called polysaccharides. (poly- “many”)

Carbohydrates v Functions of carbohydrates include: § Living things use carbohydrates as their main source of energy § Living things store carbohydrates as complex sugars known as starches § Plants also use carbohydrates for structural purposes

Carbohydrates v An indicator is a chemical that changes color in the presence of specific biomolecules. v. Iodine is an indicator used to identify complex carbohydrates (starches). v. Benedict’s is an indicator used to identify simple carbohydrates (sugars). Did you know? ? Sugars end with the suffix: -ose. Glucose, sucrose, fructose, etc.

Carbohydrates on a Food label v There are three types of carbs that we eat: v. Dietary fiber v. Sugar v. Starch v. On a food label, there is a total # of carbs. Below that, it lists the # of grams of dietary fiber (if this is missing, there is no fiber in that food) and # of grams of sugar. v. Starches are not listed on food labels. To figure out the amount of starch in a food, subtract fiber and sugar from the total carbohydrates.
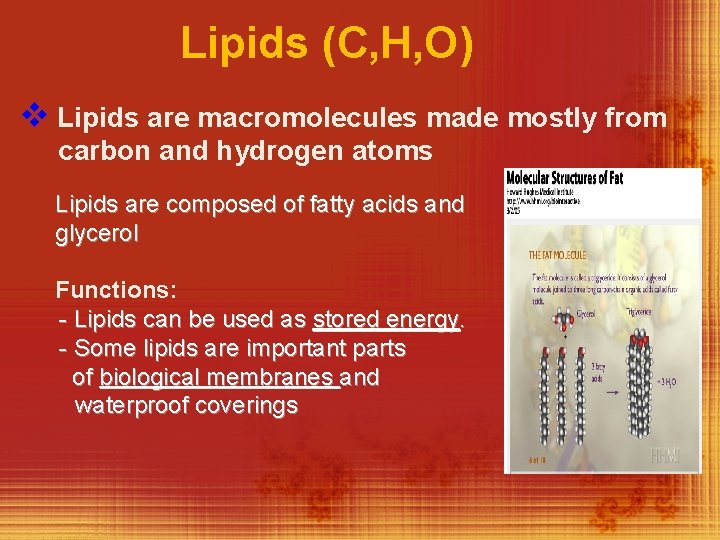
Lipids (C, H, O) v Lipids are macromolecules made mostly from carbon and hydrogen atoms Lipids are composed of fatty acids and glycerol Functions: - Lipids can be used as stored energy. - Some lipids are important parts of biological membranes and waterproof coverings

Lipids v. The common categories of lipids are: fats oils waxes steroids
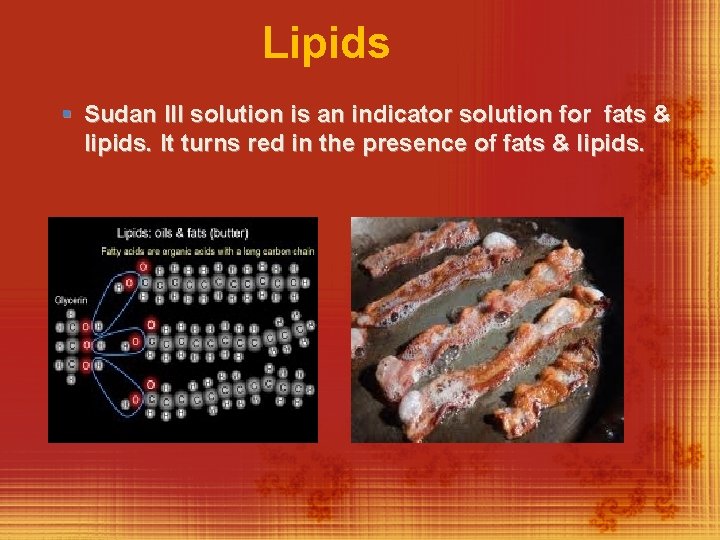
Lipids § Sudan III solution is an indicator solution for fats & lipids. It turns red in the presence of fats & lipids.

Proteins (C, H, O, N) sometimes (C, H, O, N, S) v. Proteins are macromolecules that contain nitrogen, carbon, hydrogen, and oxygen. § Proteins are large molecules (polymers) made up of monomers called amino acids. § Biuret Solution is an indicator solution for proteins. It turns blue-violet in the presence of proteins.
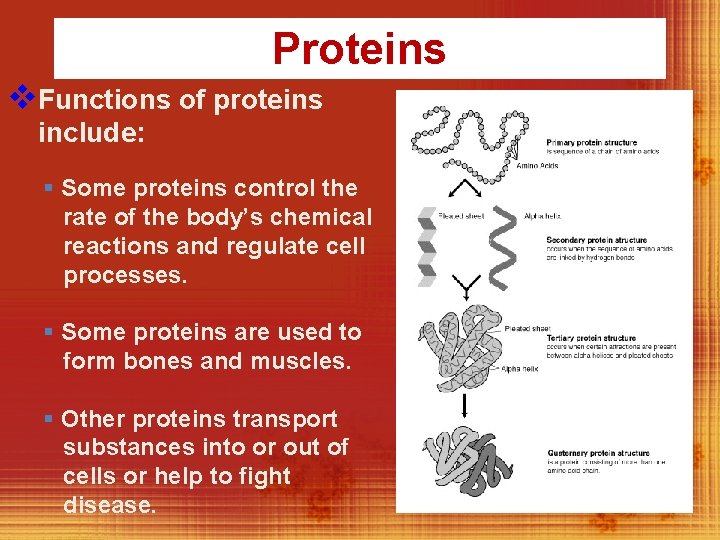
Proteins v. Functions of proteins include: § Some proteins control the rate of the body’s chemical reactions and regulate cell processes. § Some proteins are used to form bones and muscles. § Other proteins transport substances into or out of cells or help to fight disease.

Biological Catalyst • A catalyst is a substance that speeds up the rate of a chemical reaction. • Catalysts work by lowering a reaction’s activation energy.

Enzymes • Enzymes are proteins that act as biological catalysts. • Enzymes speed up chemical reactions by lowering activation energies. • Did you know? ? Most enzymes end with the suffix –ase. Lactase, transaminase, phosphatase, etc.

Enzymes • Proteins make efficient catalysts because their shapes are very specific.

Nucleic Acids (C, H, O, N, P) v. Nucleic acids are macromolecules containing hydrogen, oxygen, nitrogen, carbon, and phosphorus. § Nucleic acids are polymers assembled from individual monomers known as nucleotides. § Nucleotides include of three parts: - a 5 -carbon sugar - a phosphate group - a nitrogenous base
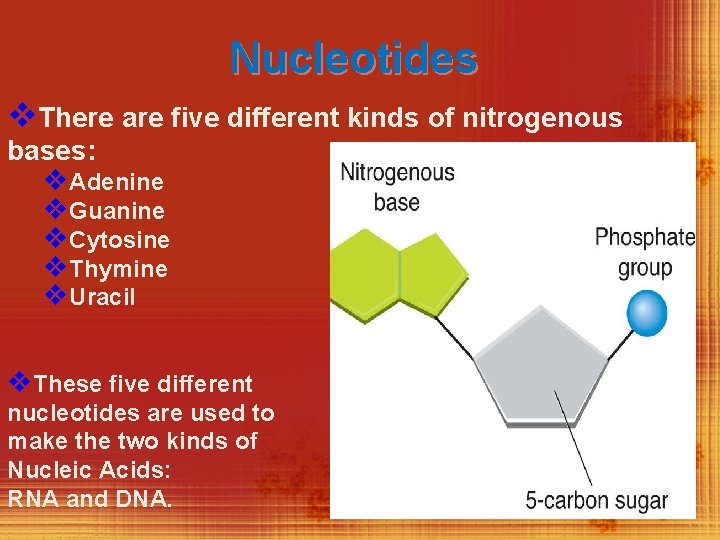
Nucleotides v. There are five different kinds of nitrogenous bases: v. Adenine v. Guanine v. Cytosine v. Thymine v. Uracil v. These five different nucleotides are used to make the two kinds of Nucleic Acids: RNA and DNA.
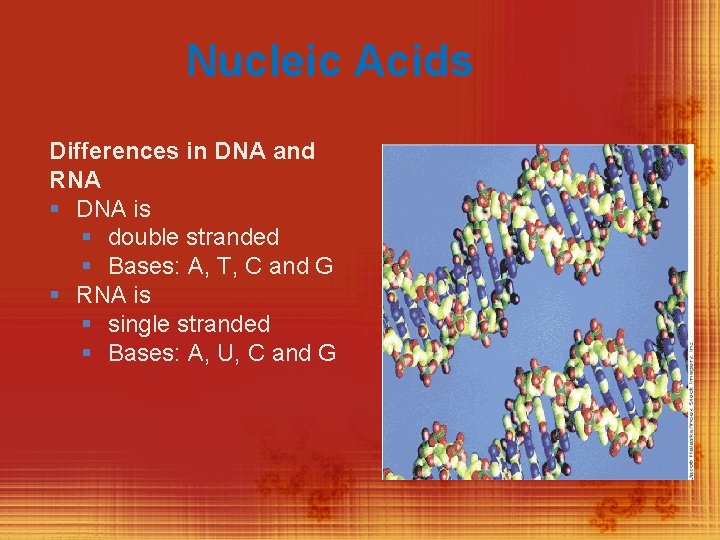
Nucleic Acids Differences in DNA and RNA § DNA is § double stranded § Bases: A, T, C and G § RNA is § single stranded § Bases: A, U, C and G

Nucleic Acids v. Functions: § Nucleic acids store and transmit genetic information. § There are two kinds of nucleic acids, -ribonucleic acid (RNA) -deoxyribonucleic acid (DNA).

Indicators (write on the page of biomolecule measured) Sudan III solution is an indicator solution for fats & lipids. It turns red in the presence of fats & lipids. Benedict’s solution is an indicator solution for simple sugars. It changes from blue to yellow, orange or red. Iodine solution is an indicator solution for complex sugars. It changes from brown to blue /purple. Biuret Solution is an indicator solution for
- Slides: 23