BIOMEMS Optics background Winter 2011 Contents u u
BIOMEMS Optics background, Winter 2011

Contents u u u u u Properties of Light Absorption and Light Emission Spectrophotometry: Absorbance Spectroscopy: Luminescence Chemiluminescence Bioluminescence Phosphoresence Applications Spectroscopy: Overview

Properties of Light as a wave. • The wave-particle duality. A principle of quantum mechanics which implies that light (and, indeed, all other subatomic particles) sometimes act like a wave, and sometimes act like a particle, depending on the experiment you are performing. E = amplitude of electric field (J) n = frequency (Hz) l = wavelength (m) c = speed of light (2. 998 x 108 m/s in vacuum) Light is also viewed as particles or packets of energy called photons. Energy of a photon: Also written as:
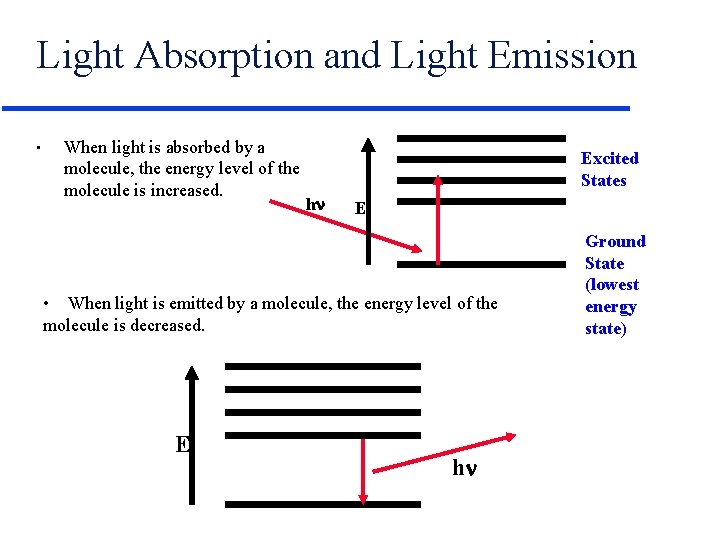
Light Absorption and Light Emission • When light is absorbed by a molecule, the energy level of the molecule is increased. Excited States hn E • When light is emitted by a molecule, the energy level of the molecule is decreased. E hn Ground State (lowest energy state)

Spectrophotometry u u u The wavelength of light determines how it interacts with matter (see also lithography). We use these interactions as a probe to obtain chemical information about samples. Spectrophotometry is the use of “light” in chemical measurements UV 400 nm IR 750 nm IR typically referred to by wavenumber (10 -12, 500 cm-1)

Spectrophotometry Blue - Orange Blue-green - Red Green - Purple How is EMR used in analysis? i) Sample absorbs EMR (Absorption) - absorbed wavelengths qualitative analysis - extent of absorption quantitative analysis. ii) Sample emits (or can be forced to emit) EMR (Luminescence) - emission wavelength qualitative - emission intensity quantitative. iii) Sample scatters EMR - sometimes qualitative & quantitative What we see is not the color absorbed, but the complementary color. u The absorbing species is called a chromophore-Contrast with ionophore u e. g. Blue jeans absorb orange wavelengths u
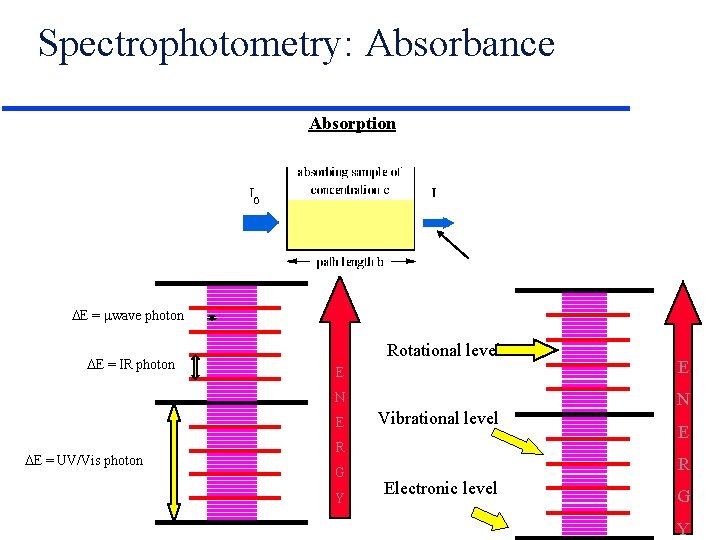
Spectrophotometry: Absorbance Absorption E = wave photon E = IR photon Rotational level E E N N E E = UV/Vis photon Vibrational level R G Y E R Electronic level G Y

Spectrophotometry: Absorbance Jablonski Diagram
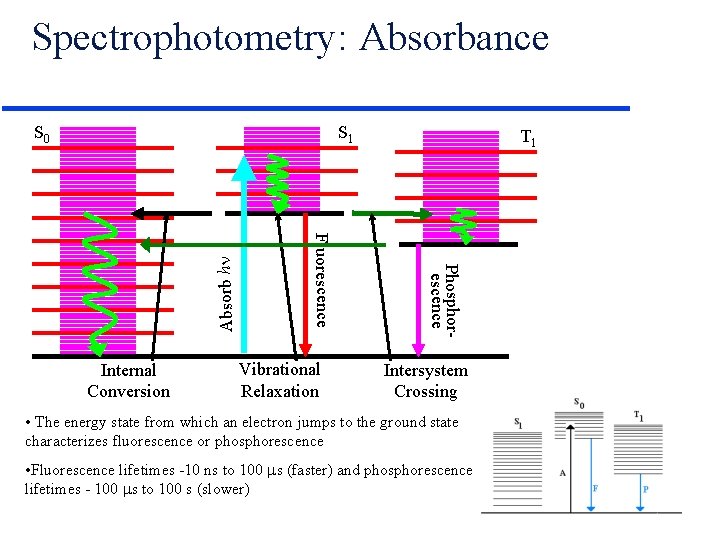
Spectrophotometry: Absorbance S 1 Vibrational Relaxation T 1 Phosphorescence Internal Conversion Fluorescence Absorb h S 0 Intersystem Crossing • The energy state from which an electron jumps to the ground state characterizes fluorescence or phosphorescence • Fluorescence lifetimes -10 ns to 100 s (faster) and phosphorescence lifetimes - 100 s to 100 s (slower)
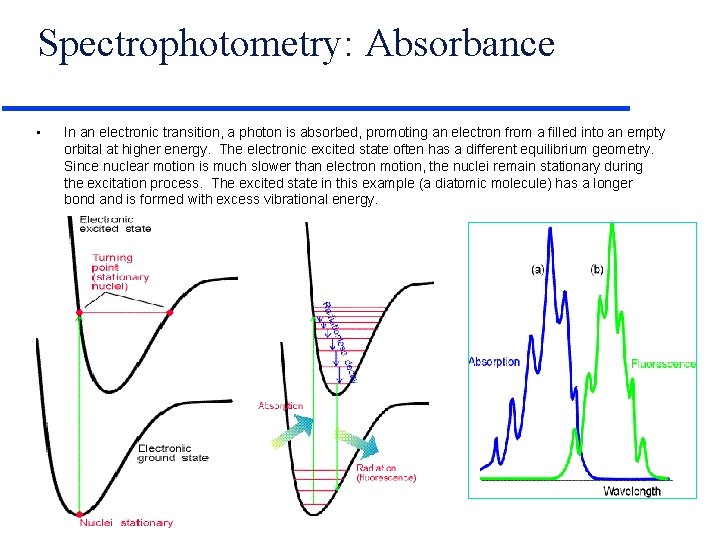
Spectrophotometry: Absorbance • In an electronic transition, a photon is absorbed, promoting an electron from a filled into an empty orbital at higher energy. The electronic excited state often has a different equilibrium geometry. Since nuclear motion is much slower than electron motion, the nuclei remain stationary during the excitation process. The excited state in this example (a diatomic molecule) has a longer bond and is formed with excess vibrational energy.
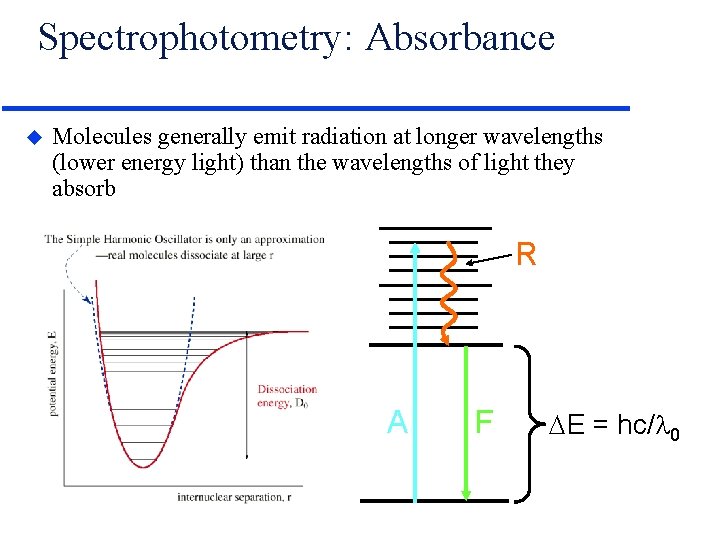
Spectrophotometry: Absorbance u Molecules generally emit radiation at longer wavelengths (lower energy light) than the wavelengths of light they absorb R S 1 A S 0 F E = hc/ 0
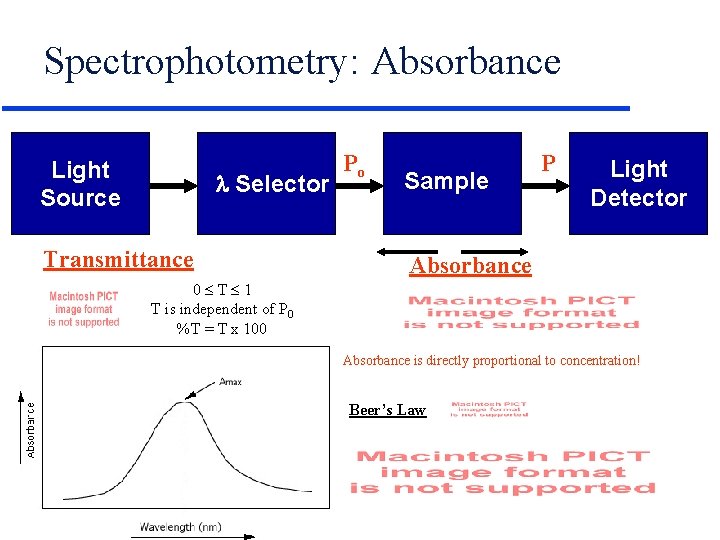
Spectrophotometry: Absorbance Light Source l Selector Transmittance Po Sample P Light Detector Absorbance 0 T 1 T is independent of P 0 %T = T x 100 Absorbance is directly proportional to concentration! Beer’s Law
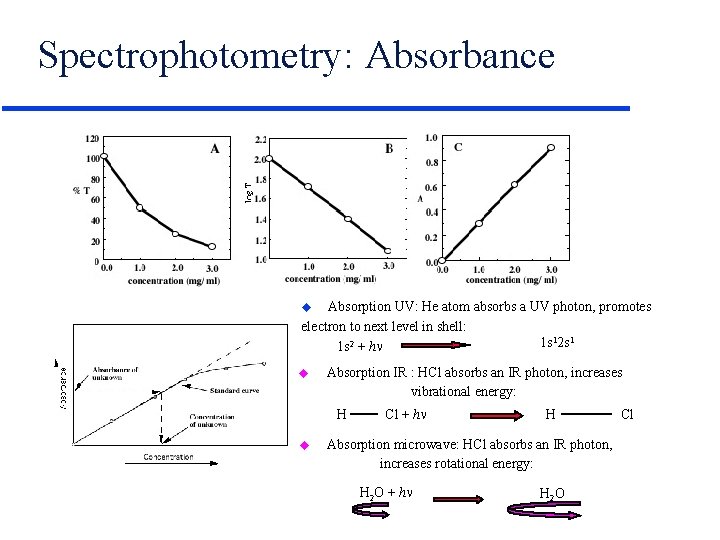
Spectrophotometry: Absorbance Absorption UV: He atom absorbs a UV photon, promotes electron to next level in shell: 1 s 12 s 1 1 s 2 + h u u Absorption IR : HCl absorbs an IR photon, increases vibrational energy: H u Cl + h H Absorption microwave: HCl absorbs an IR photon, increases rotational energy: H 2 O + h H 2 O Cl

Spectrophotometry: Absorbance Po P P-d. P dx A = bc d. P = -Pc dx c = concentration of analyte probability of a photon being absorbed
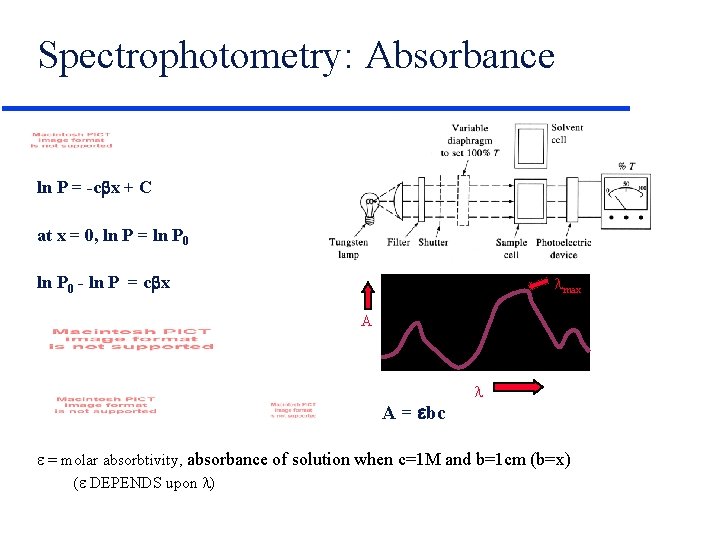
Spectrophotometry: Absorbance ln P = -c x + C at x = 0, ln P = ln P 0 - ln P = c x max A A = bc = molar absorbtivity, absorbance of solution when c=1 M and b=1 cm (b=x) ( DEPENDS upon )
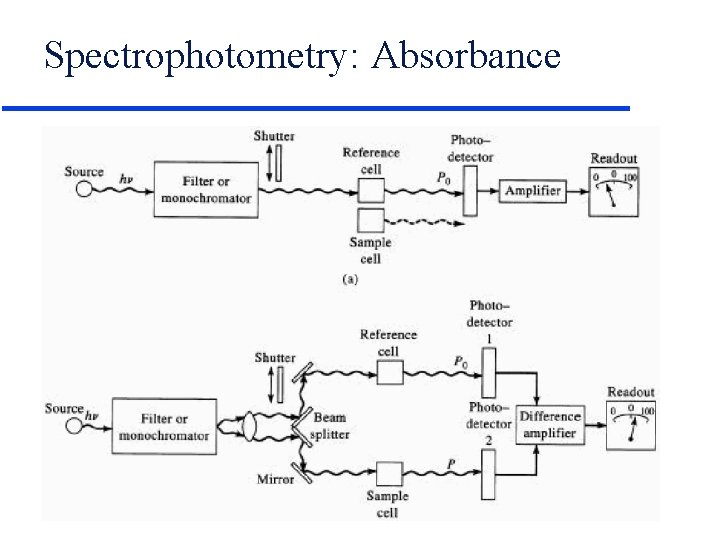
Spectrophotometry: Absorbance
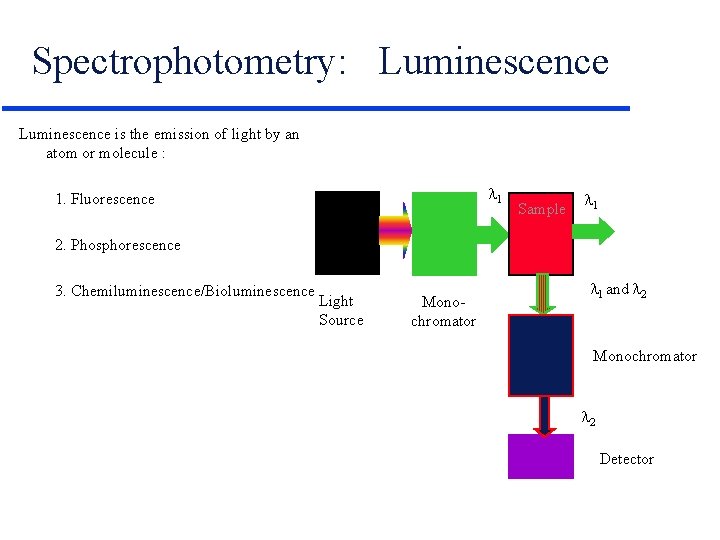
Spectrophotometry: Luminescence is the emission of light by an atom or molecule : 1 1. Fluorescence Sample 1 2. Phosphorescence 3. Chemiluminescence/Bioluminescence Light Source Monochromator 1 and 2 Monochromator 2 Detector

Spectrophotometry: Luminescence u For absorbance PA = P 0 - P – Measure transmission and determine A as a ratio of P 0 and P – A is INDEPENDENT of magnitude of P 0 u u For fluorescence the intensity is – Measure absolute number of photons – I is DEPENDENT on P 0 K depends upon: – efficiency of fluorescence – light collection efficiency

Spectrophotometry: Luminescence u I depends on: – (absorbtivity of sample) – b (path length of sample holder) – c (concentration of sample) – P 0 (intensity of incident radiation) – K I = KP 02. 3 bc A major difference between luminescence and absorption is that the former is dependent upon number of incident photons. u Typically use lasers for fluorescence because of high photon flux u Another major difference between luminescence and absorption is that the former is based on a absolute measurement while the latter is relative. u Can measure much lower concentrations with fluorescence u
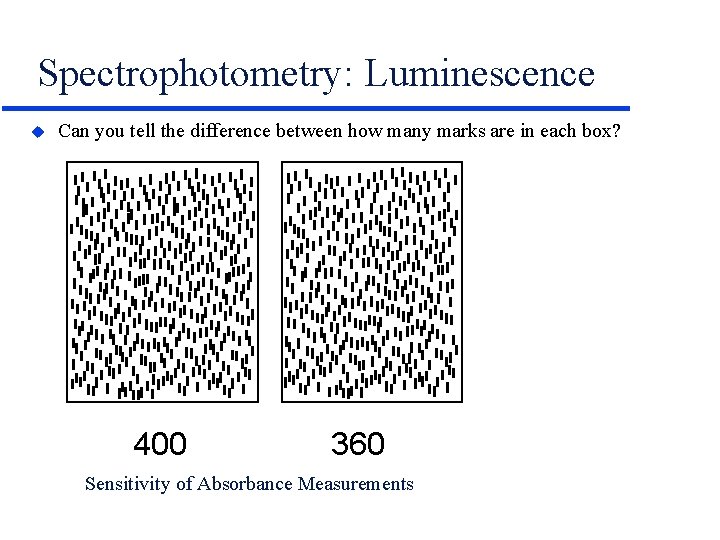
Spectrophotometry: Luminescence u Can you tell the difference between how many marks are in each box? 400 360 Sensitivity of Absorbance Measurements
Spectrophotometry: Luminescence u Can you tell the difference between how many marks are in each box? 0 Sensitivity of Luminescence Measurements 40
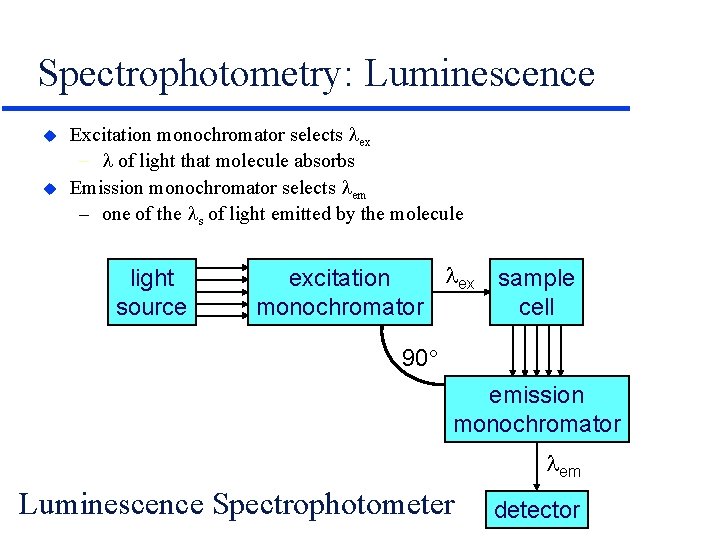
Spectrophotometry: Luminescence u u Excitation monochromator selects ex - of light that molecule absorbs Emission monochromator selects em – one of the s of light emitted by the molecule light source ex sample excitation cell monochromator 90 emission monochromator em Luminescence Spectrophotometer detector
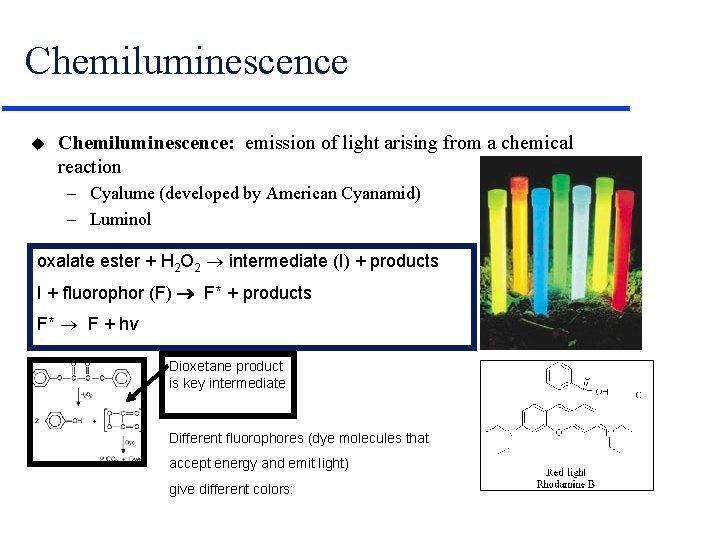
Chemiluminescence u Chemiluminescence: emission of light arising from a chemical reaction – Cyalume (developed by American Cyanamid) – Luminol oxalate ester + H 2 O 2 intermediate (I) + products I + fluorophor (F) F* + products F* F + hv Dioxetane product is key intermediate Different fluorophores (dye molecules that accept energy and emit light) give different colors:
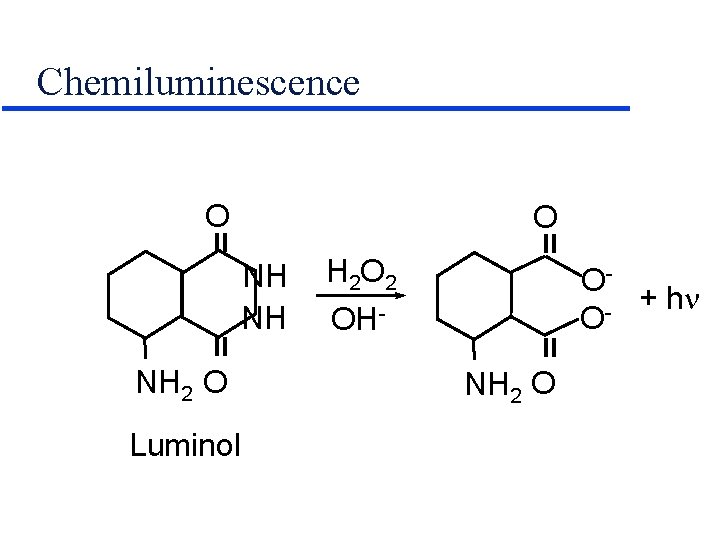
Chemiluminescence = NH NH NH 2 O Luminol H 2 O 2 OH- = O = = O NH 2 O O+ h O

Bioluminescence u Bioluminescence is the ability of living things to emit light. It is found in – – – u u many marine animals, both invertebrate (e. g. , some cnidarians, crustaceans, squid) and vertebrate (some fishes); some terrestrial animals (e. g. , fireflies, some centipedes); some fungi and bacteria The molecular details vary from organism to organism, but each involves – – u Green fluorescent protein found in some jellyfish causes bioluminescence. a luciferin, a light-emitting substrate a luciferase, an enzyme that catalyzes the reaction ATP, the source of energy molecular oxygen, O 2 The more ATP available, the brighter the light. In fact, firefly luciferin and luciferase are commercially available for measuring the amount of ATP in biological materials. Fireflies use their flashes to attract mates. The pattern differs from species to species. In one species, the females sometimes mimic the pattern used by females of another species. When the males of the second species respond to these "femmes fatales", they are eaten!
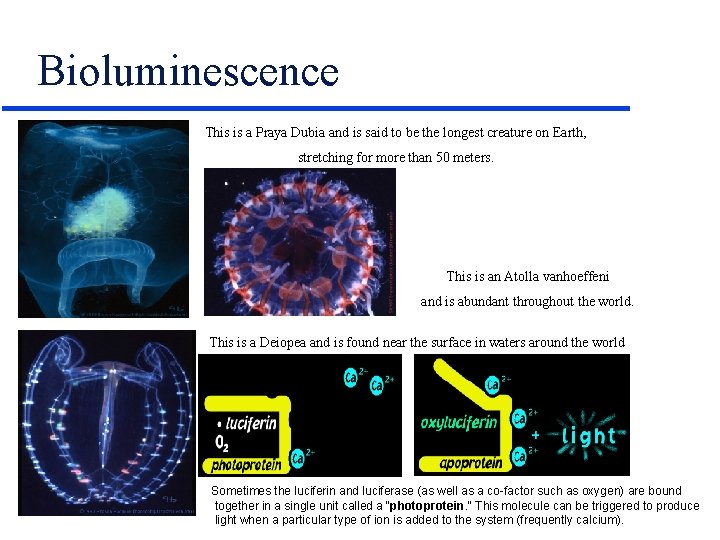
Bioluminescence This is a Praya Dubia and is said to be the longest creature on Earth, stretching for more than 50 meters. This is an Atolla vanhoeffeni and is abundant throughout the world. This is a Deiopea and is found near the surface in waters around the world Sometimes the luciferin and luciferase (as well as a co-factor such as oxygen) are bound together in a single unit called a "photoprotein. " This molecule can be triggered to produce light when a particular type of ion is added to the system (frequently calcium).
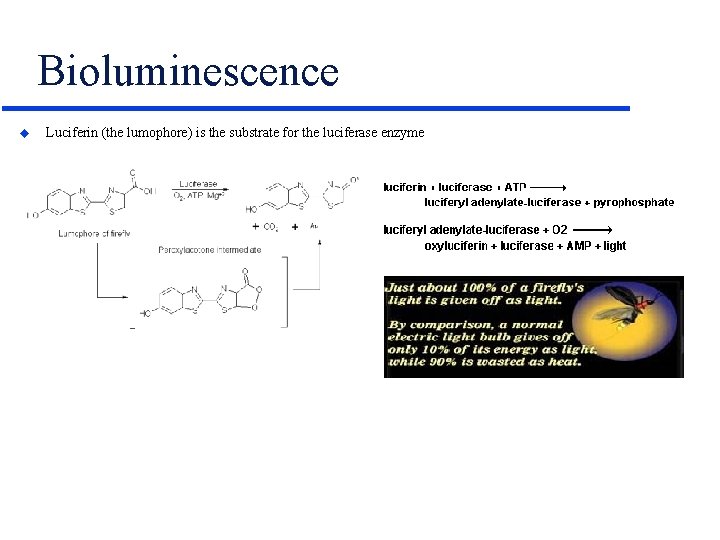
Bioluminescence u Luciferin (the lumophore) is the substrate for the luciferase enzyme
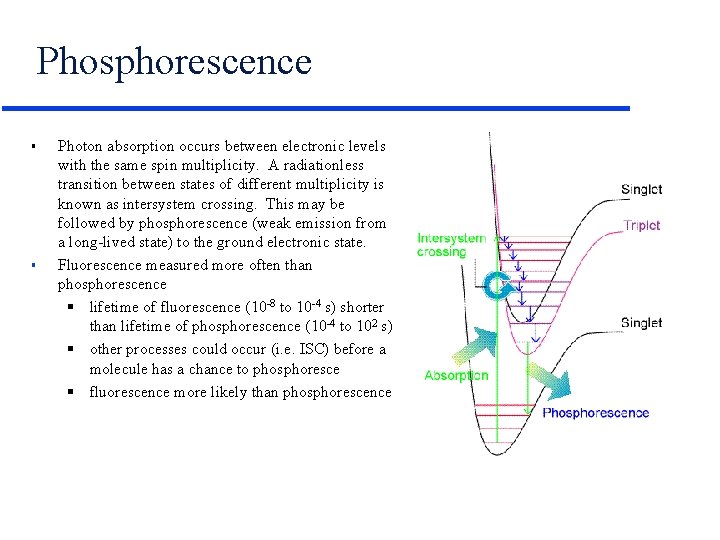
Phosphorescence § § Photon absorption occurs between electronic levels with the same spin multiplicity. A radiationless transition between states of different multiplicity is known as intersystem crossing. This may be followed by phosphorescence (weak emission from a long-lived state) to the ground electronic state. Fluorescence measured more often than phosphorescence § lifetime of fluorescence (10 -8 to 10 -4 s) shorter than lifetime of phosphorescence (10 -4 to 102 s) § other processes could occur (i. e. ISC) before a molecule has a chance to phosphoresce § fluorescence more likely than phosphorescence
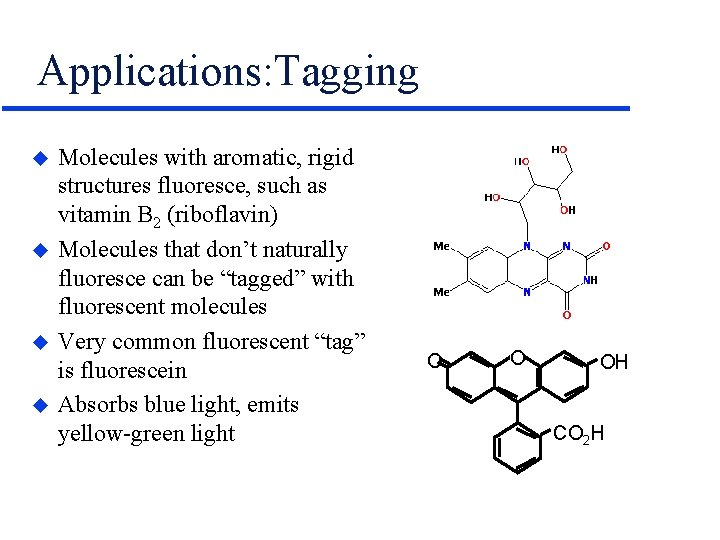
Applications: Tagging u u Molecules with aromatic, rigid structures fluoresce, such as vitamin B 2 (riboflavin) Molecules that don’t naturally fluoresce can be “tagged” with fluorescent molecules Very common fluorescent “tag” is fluorescein Absorbs blue light, emits yellow-green light O O OH CO 2 H
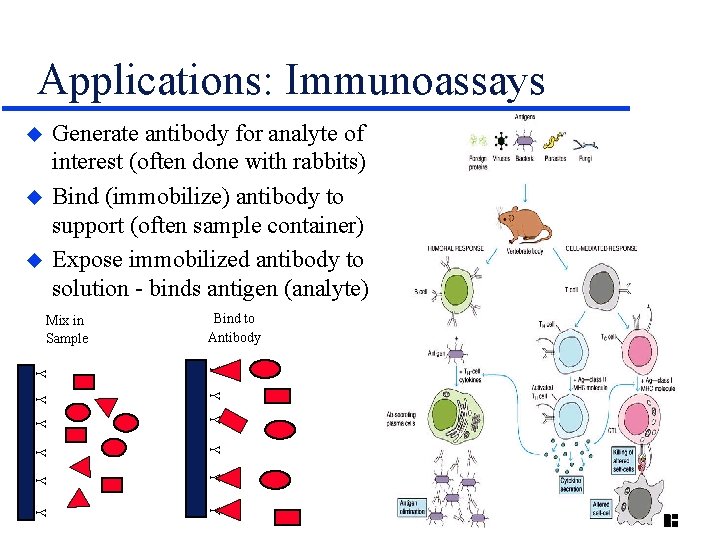
Applications: Immunoassays Generate antibody for analyte of interest (often done with rabbits) Bind (immobilize) antibody to support (often sample container) Expose immobilized antibody to solution - binds antigen (analyte) u u u Mix in Sample Bind to Antibody Y Y Y
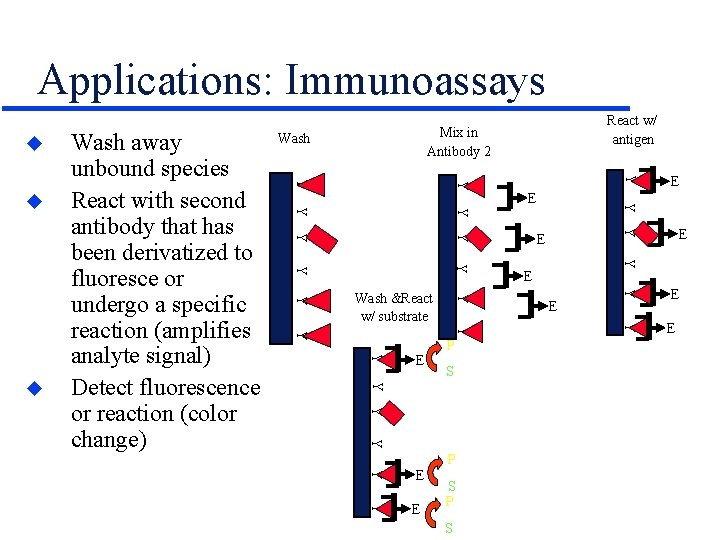
Applications: Immunoassays u Y Y Y Y Y E P S E E Y Y Y S E Y Y Y E P E Y Y Wash &React w/ substrate E Y Y u Wash away unbound species React with second antibody that has been derivatized to fluoresce or undergo a specific reaction (amplifies analyte signal) Detect fluorescence or reaction (color change) React w/ antigen Mix in Antibody 2 E
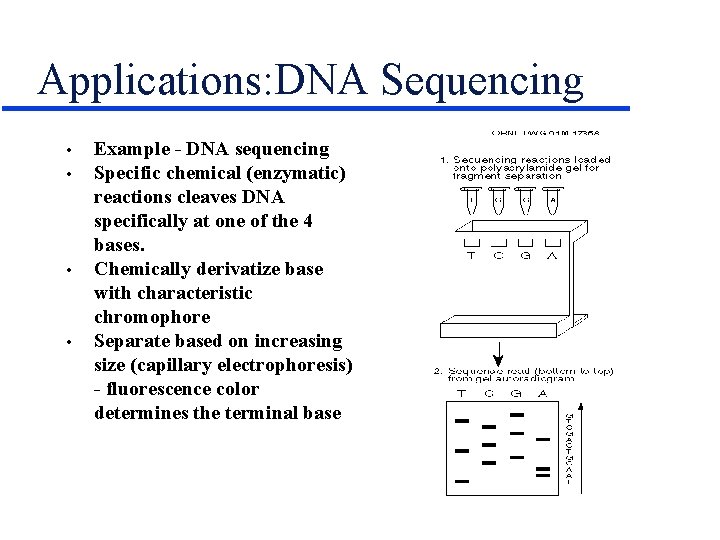
Applications: DNA Sequencing • • Example - DNA sequencing Specific chemical (enzymatic) reactions cleaves DNA specifically at one of the 4 bases. Chemically derivatize base with characteristic chromophore Separate based on increasing size (capillary electrophoresis) - fluorescence color determines the terminal base

Spectroscopy Overview
- Slides: 33