BIOMEMS DEVICES TO MONITOR NEURAL ELECTRICAL CIRCUITRY Andres

BIO-MEMS DEVICES TO MONITOR NEURAL ELECTRICAL CIRCUITRY Andres Huertas, Michele Panico, Shuming Zhang ME 381 Final Project, Dec 4 th, 2003
OUTLINE • Planar monitoring system • Neurons cage array • Living chips with peptide amphiphiles gels

ORIGINAL TECHNIQUE • Simple • Rugged • Easiest solution “Future hybrid neuron-semiconductor chips will consist of complex neural networks that are directly interfaced to electronic integrated circuits. . . and may lead to novel computational facilities. ” Jenker, Müller, Fromherz, Biol. Cybern. 84, 239 -249 (2001)
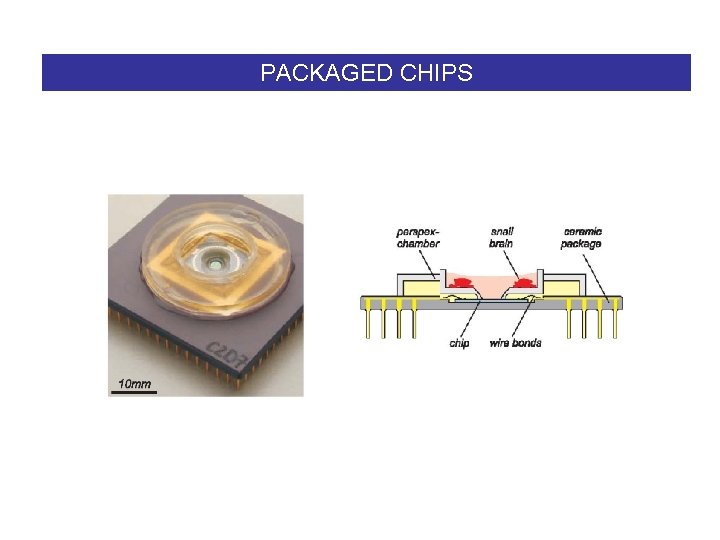
PACKAGED CHIPS
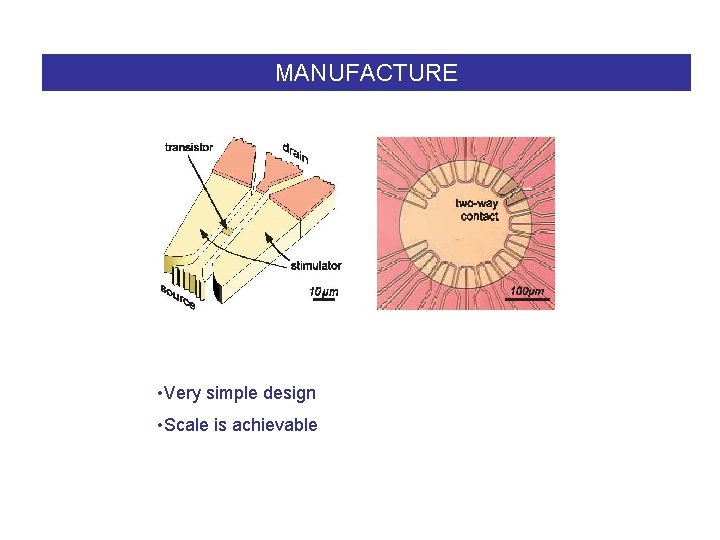
MANUFACTURE • Very simple design • Scale is achievable
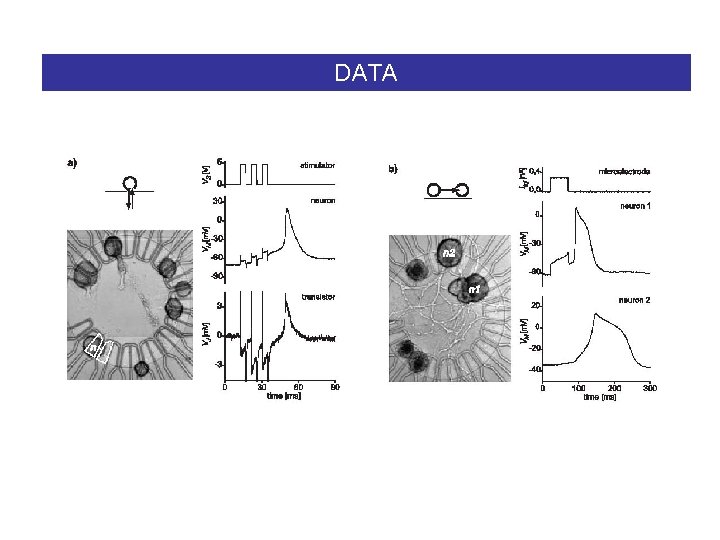
DATA
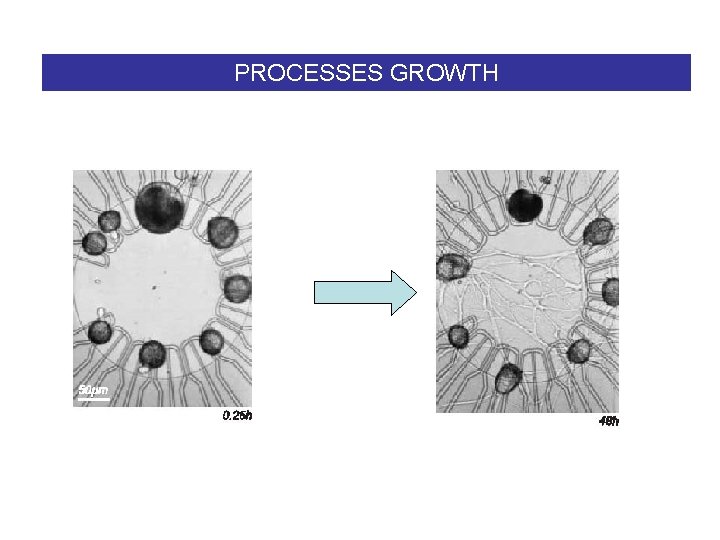
PROCESSES GROWTH
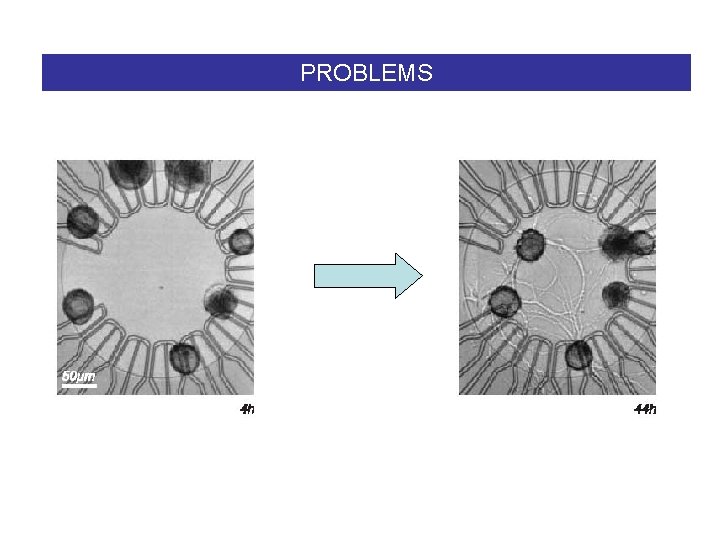
PROBLEMS

MANUFACTURE Polyimide Picket Fences. Günther Zeck and Peter, Fromherz, PNAS, 2001 vol. 98, no. 18, 10457 -10462
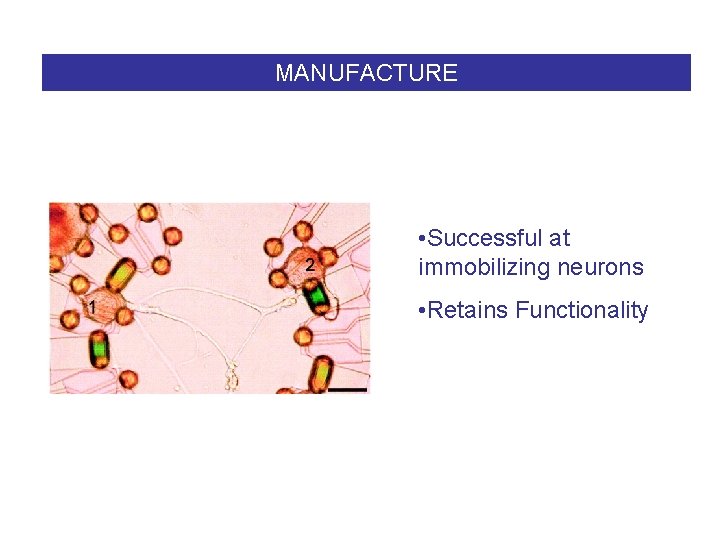
MANUFACTURE • Successful at immobilizing neurons • Retains Functionality
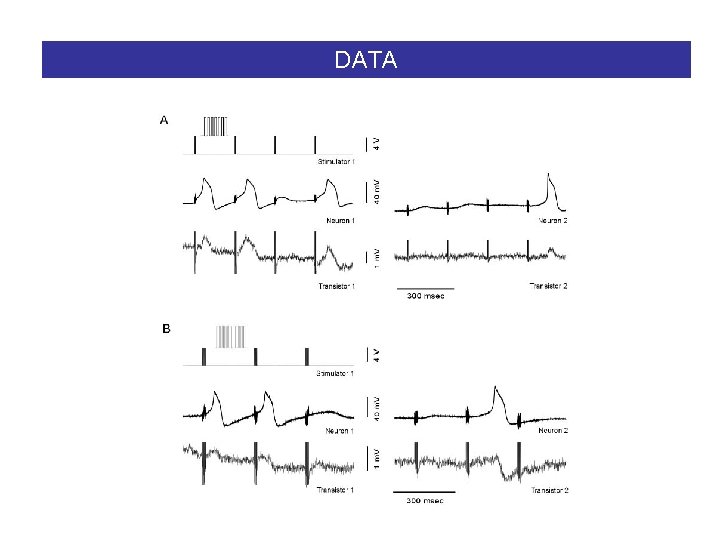
DATA

NEURO-CAGES FOR LIVE NEURAL NETWORKS STUDY To study neural networks of individual neurons is the aim of neuroscience Conventional technique: planar arrays of extracellular metal electrodes on which neural cultures are grown Limitations: • Small proportion of neurons can be accessed • Neurons are mobile, thus repeated measurements of a specific neuron are difficult Micro-cages: each neuron is trapped into one cage Features: • Arrays of neuro-cages to allow the formation of neural networks • One-to-one correspondence between neurons and electrodes • Neurite growth is not affected by the physical confinement Qing He. , Ellis Meng, Yu-Chong Tai, Christopher M. Jerome Pine, etc, The 12 th International Conference on Solid-State Sensors, Boston, MA, Jun 8 -12, 2003

SILICON MICROMACHINED NEUROCHIPS Neurochip: 1 cm square, 500 µm thick silicon wafer, with a 4 x 4 array of wells spaced on 100 µm centers

MICROFABRICATION PROCESS 1) Composite layer of 180 nm LPCVD silicon nitride 2) on top of 50 nm thermal oxide is formed 2) Pattern nitride-oxide layer to define the openings 3) for the metal electrodes 3) 1 µm oxide step around the electrode openings 4) Nitride is stripped 5) Metallization is done using lift-off process 6) Metallization is covered by a composite insulation 7) Layer of 0. 5 µm LTO and 1 µm PECVD nitride 7) Opening of bonding pads and alignment marks 8) EDP etching on the back using the boron-doped 9) layer as an etch stop 9) RIE is used to form grillwork 10) Neuron wells are formed by EDP etching to the 11) electrodes on the front side of the wafer 11) Removal of pad oxide at the bottom of the wells
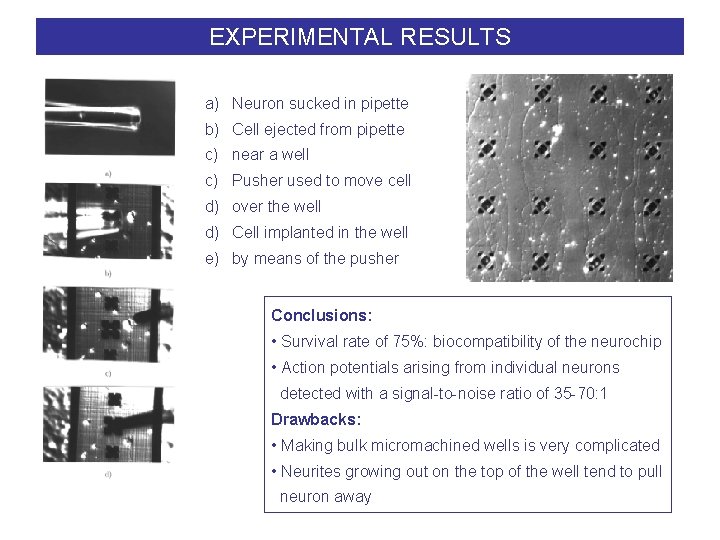
EXPERIMENTAL RESULTS a) Neuron sucked in pipette b) Cell ejected from pipette c) near a well c) Pusher used to move cell d) over the well d) Cell implanted in the well e) by means of the pusher Conclusions: • Survival rate of 75%: biocompatibility of the neurochip • Action potentials arising from individual neurons detected with a signal-to-noise ratio of 35 -70: 1 Drawbacks: • Making bulk micromachined wells is very complicated • Neurites growing out on the top of the well tend to pull neuron away
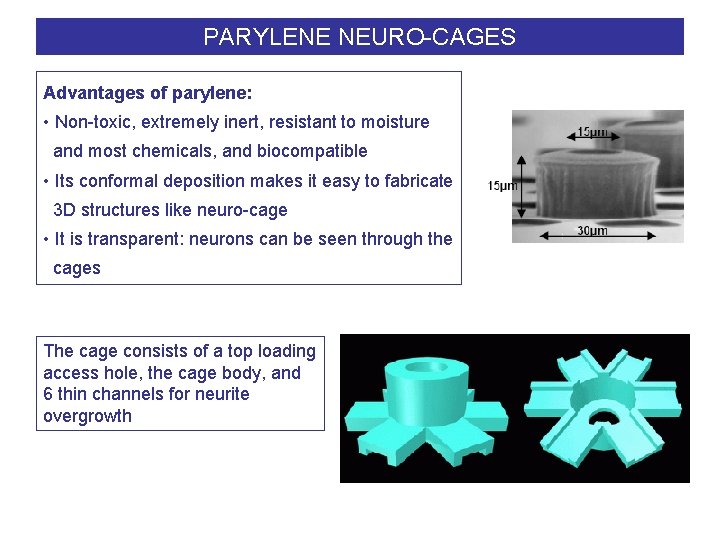
PARYLENE NEURO-CAGES Advantages of parylene: • Non-toxic, extremely inert, resistant to moisture and most chemicals, and biocompatible • Its conformal deposition makes it easy to fabricate 3 D structures like neuro-cage • It is transparent: neurons can be seen through the cages The cage consists of a top loading access hole, the cage body, and 6 thin channels for neurite overgrowth
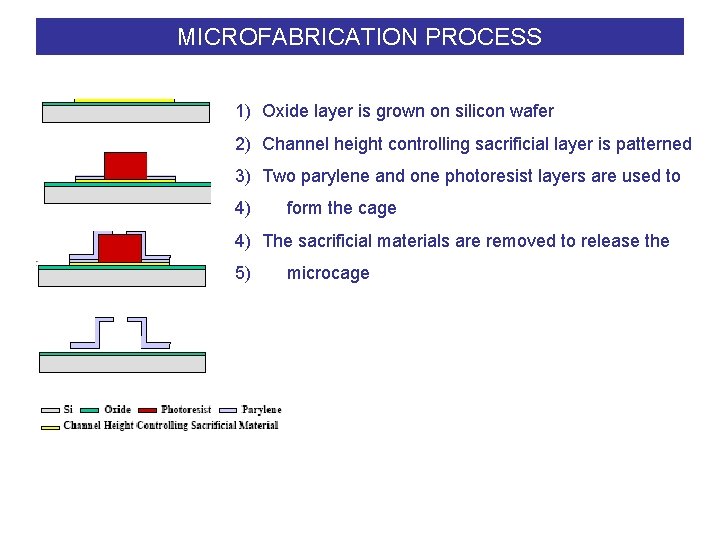
MICROFABRICATION PROCESS 1) Oxide layer is grown on silicon wafer 2) Channel height controlling sacrificial layer is patterned 3) Two parylene and one photoresist layers are used to 4) form the cage 4) The sacrificial materials are removed to release the 5) microcage

PARYLENE-TO-OXIDE ADHESION To improve the adhesion of the cages to the substrate: • Mechanically anchoring parylene to substrate using DRIE • To roughen the anchoring area with short time etching in Br. F 3 or Xe. F 2
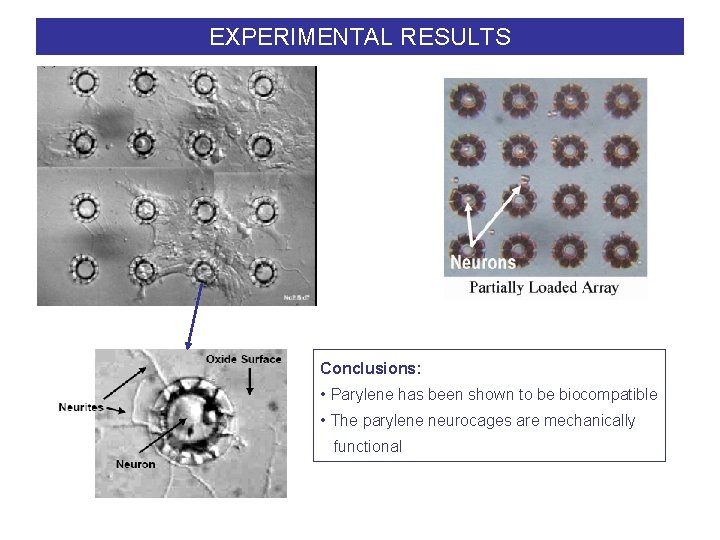
EXPERIMENTAL RESULTS Conclusions: • Parylene has been shown to be biocompatible • The parylene neurocages are mechanically functional
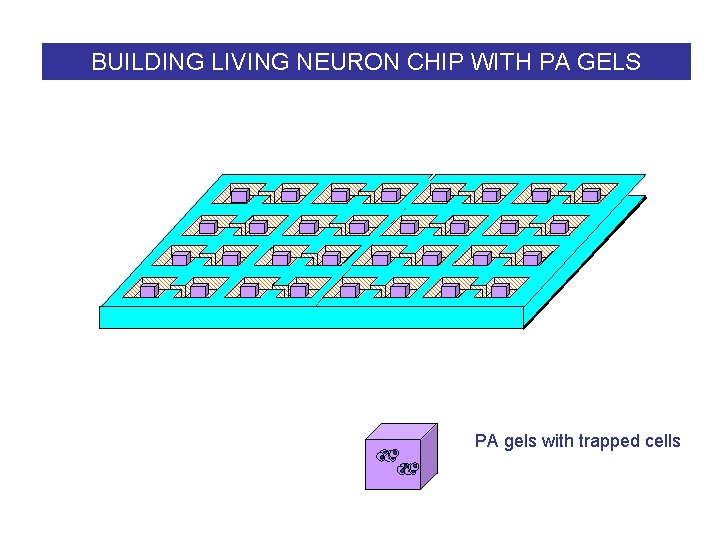
BUILDING LIVING NEURON CHIP WITH PA GELS PA gels with trapped cells
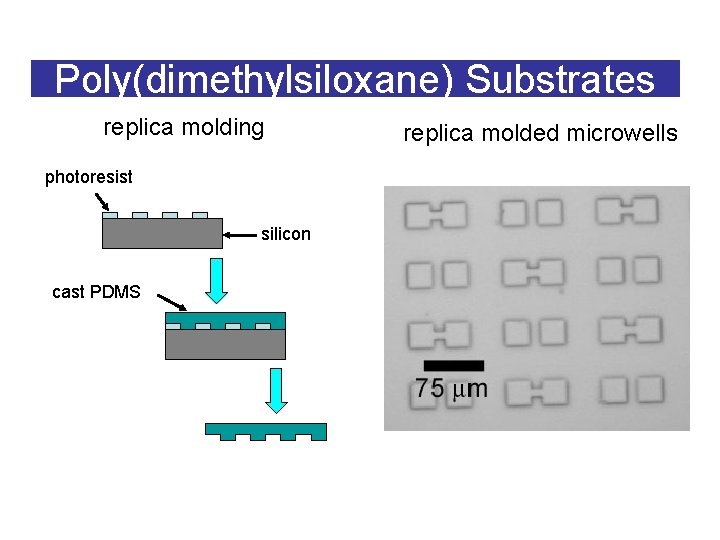
Poly(dimethylsiloxane) Substrates replica molding photoresist silicon cast PDMS replica molded microwells
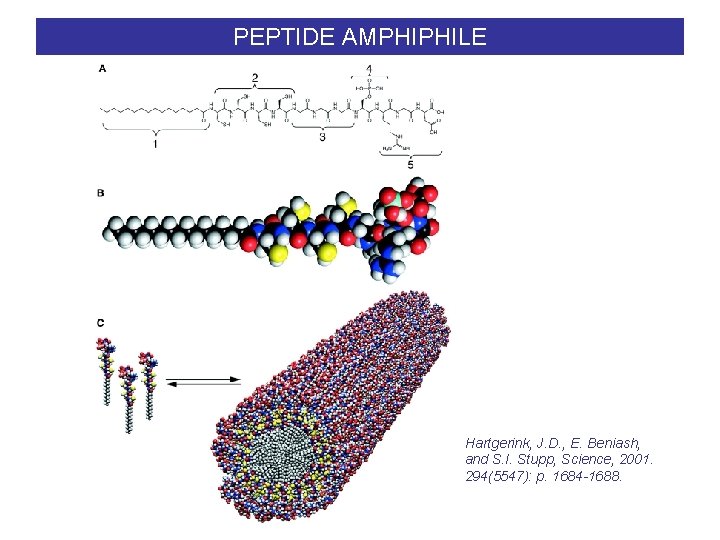
PEPTIDE AMPHIPHILE Hartgerink, J. D. , E. Beniash, and S. I. Stupp, Science, 2001. 294(5547): p. 1684 -1688.
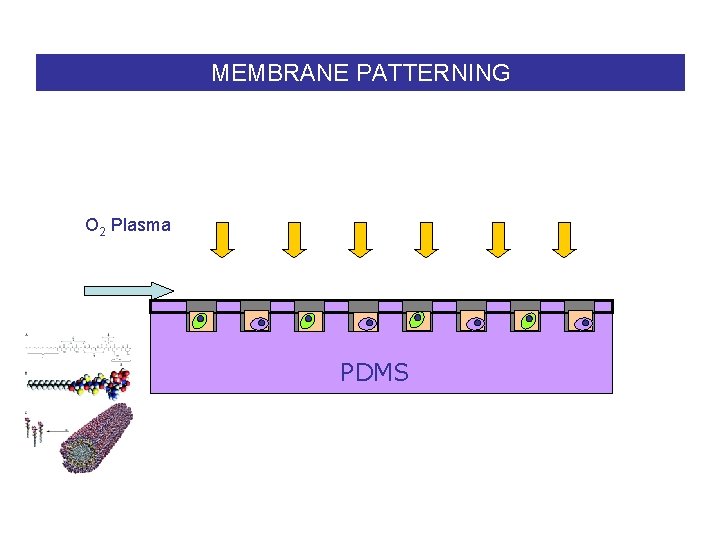
MEMBRANE PATTERNING O 2 Plasma PDMS
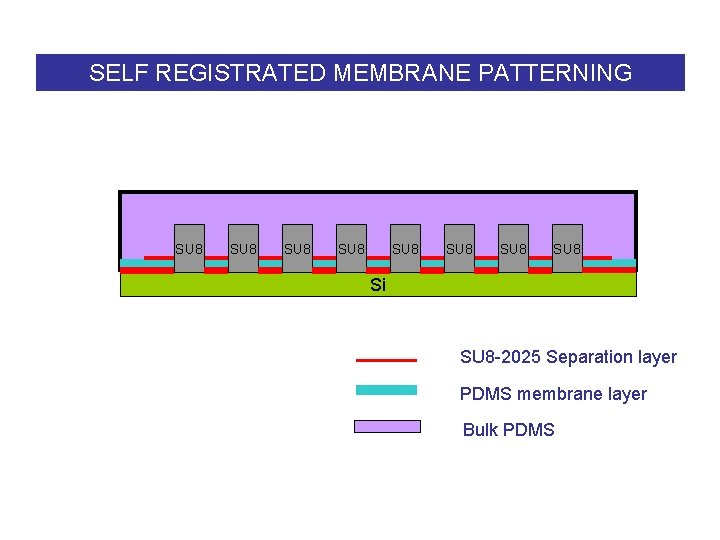
SELF REGISTRATED MEMBRANE PATTERNING SU 8 SU 8 Si SU 8 -2025 Separation layer PDMS membrane layer Bulk PDMS

ACKNOWLEDGEMENT • Professor Horacio D. Espinosa • TA: Yong Zhu • Team members
- Slides: 25