BIOMATERIALS ENT 219 Lecture 10 Polymeric Biomaterials 1

BIOMATERIALS ENT 219 Lecture 10 Polymeric Biomaterials

1. 0 Introduction n Application n n of synthetic polymers medical disposable supply prosthetic materials, dental materials implants dressings extracorporeal devices encapsulants polymeric drug delivery systems tissue engineered products orthodosis 2

1. 0 Introduction Main Advantages n n Ease of manufacturability to produce various shapes Ease of secondary processability Reasonable cost Availability with desired mechanical and physical properties. 3

1. 0 Introduction 4
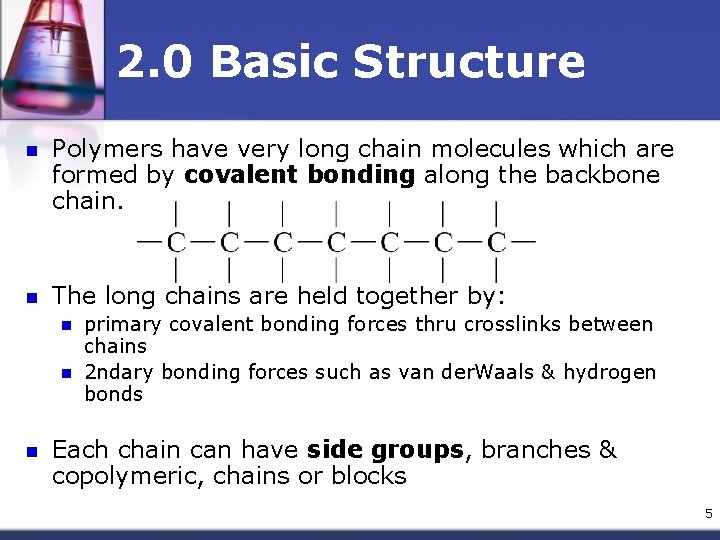
2. 0 Basic Structure n Polymers have very long chain molecules which are formed by covalent bonding along the backbone chain. n The long chains are held together by: n n n primary covalent bonding forces thru crosslinks between chains 2 ndary bonding forces such as van der. Waals & hydrogen bonds Each chain can have side groups, branches & copolymeric, chains or blocks 5

2. 0 Basic Structure n n n As the molecular chains become longer, their relative mobility decreases The higher the molecular weight, the less the mobility of chains which results in higher strength & greater thermal stability Polymer chains can be arranged in 4 ways: n n Linear Branched, Cross-linked Three-dimensional network 6
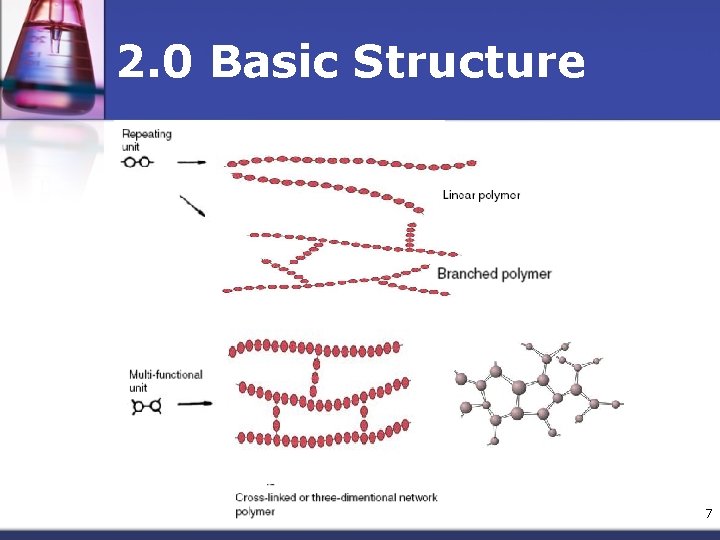
2. 0 Basic Structure 7

3. 0 Crystal & Amorphous Structure in Biopolymer n n n Crystallization is easier for polymer with shorter chain Branched polymer in which side chains are attached to the main backbone chain at positions will not crystallize easily Linear polymers are much easier to crystallize Partially crystallized structure (semicrystalline) is commonly occur in linear polymers The cross-linked or 3 -D network polymers cannot be crystallized at all & they become amorphous polymers. 8

3. 0 Crystal & Amorphous Structure in Biopolymer n n Polymer with small side group are easy to crystallize Isotactic & syndiotactic polymers usually crystallize even when the side groups are larger Copolymerization always disrupts the regularity of polymer chains thus it is more amorphous Plasticizers can prevent crystallization by keeping the chains separated from one another 9
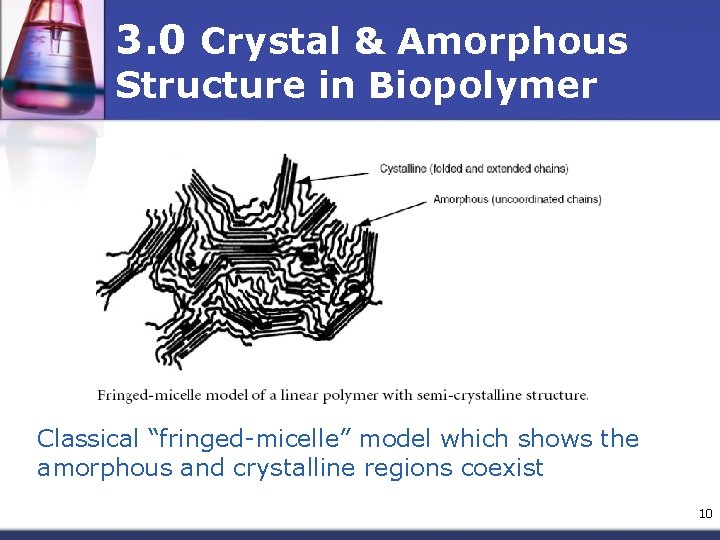
3. 0 Crystal & Amorphous Structure in Biopolymer Classical “fringed-micelle” model which shows the amorphous and crystalline regions coexist 10

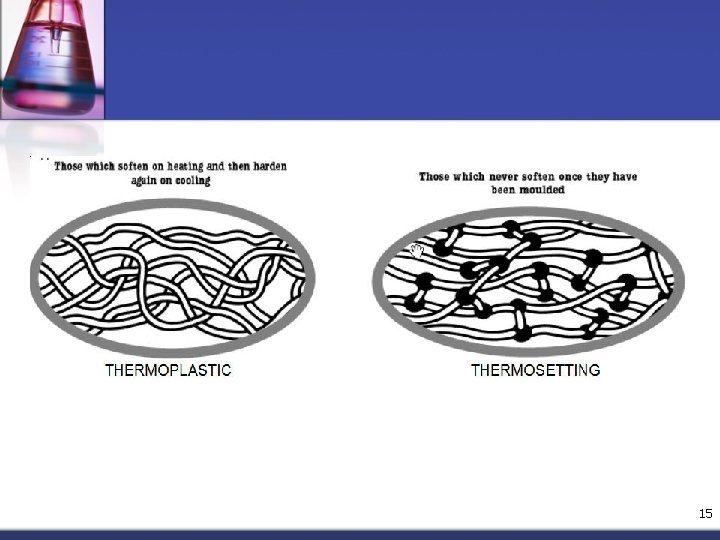
4. 0 Biopolymers Properties n Thermoplastic Polymers n Usually have linear & branched structures, they soften when heated & harden when cooled. n The process reversible & can be repeated n The reheating and reforming process did not have significant change on the polymer properties n Mostly consist of a very long main chain of carbon atoms covalently bonded together 11
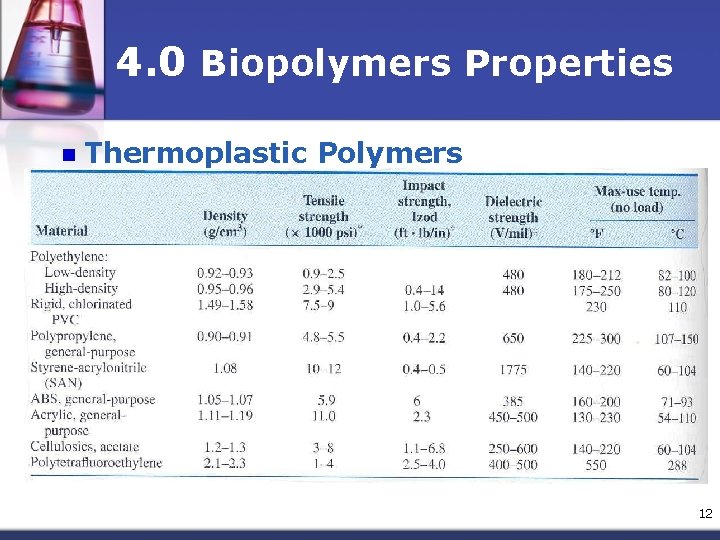
4. 0 Biopolymers Properties n Thermoplastic Polymers 12

4. 0 Biopolymers Properties n Thermosetting Polymers n The term implies that heat is required to permanently set the plastic n Thermosets polymer, once having hardened, will not soften upon heating, their structures are cross-linked & network. n They could be degrade or decompose if heated at very high temperature n Thermoset polymers are harder & stronger than thermoplastics 13
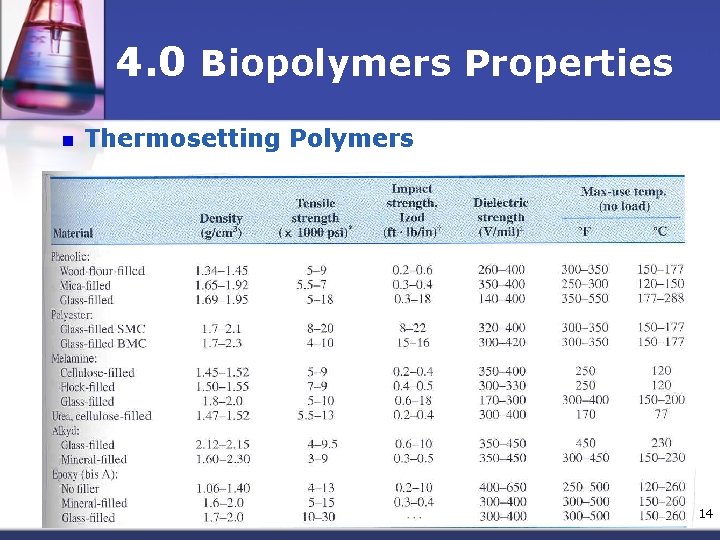
4. 0 Biopolymers Properties n Thermosetting Polymers 14
15
5. 0 n Polymeric Biomaterials Only ten to twenty polymers are mainly used in medical device fabrications from disposable to long-term implants 16

5. 0 Polymeric Biomaterials Polyethylene (PE) n Available commercially as n n n high density (HDPE) low density (LDPE) linear low density (LLDPE) very low density (VLDPE) ultra high molecular weight (UHMWPE) Clear to whitish translucent thermoplastic 17
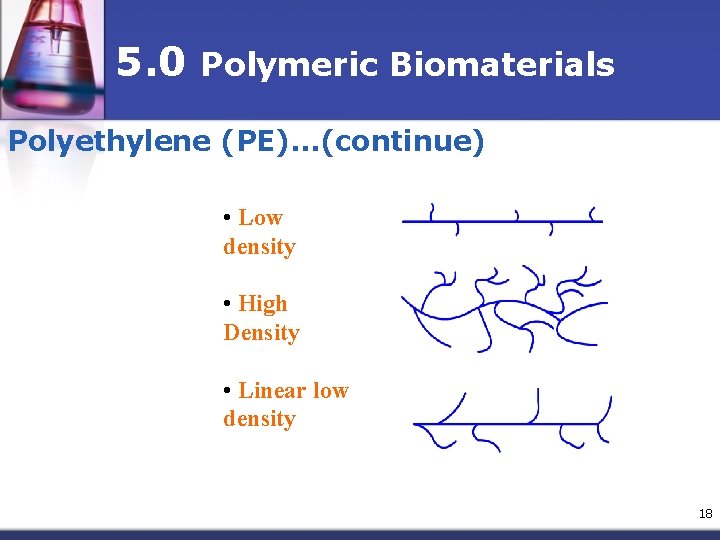
5. 0 Polymeric Biomaterials Polyethylene (PE)…(continue) • Low density • High Density • Linear low density 18
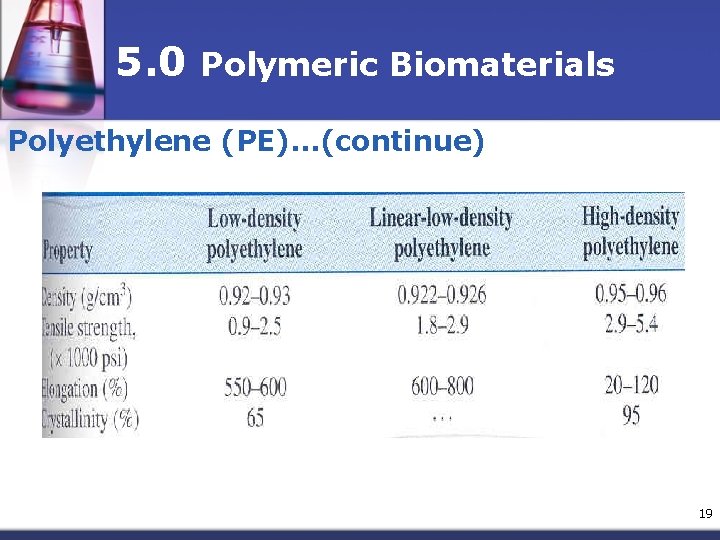
5. 0 Polymeric Biomaterials Polyethylene (PE)…(continue) 19

5. 0 Polymeric Biomaterials Polyethylene (PE)…(continue) n n HDPE -pharmaceutical bottles, nonwoven fabrics, & caps LDPE - flexible container applications, nonwovendisposable & laminated (or coextruded with paper) foil & polymers for packaging. LLDPE - pouches & bags due to its excellent puncture resistance VLDPE - extruded tubes. 20

5. 0 Polymeric Biomaterials Polyethylene (PE)…(continue) n n UHMWPE (MW >2× 106 g/mol) has been used for orthopedic implant fabrications. This orthopedic implant fabrications include load-bearing applications: n n Acetabular cup of total hip Tibial plateau & Patellar surfaces of knee joints. Specific Properties: Low cost, easy to process, excellent electrical insulator, excellent chemical resistance, tough & flexible even at low temperature 21

5. 0 Polymeric Biomaterials Polyvinylchloride (PVC) n n PVC is amorphous, does not recrystallize due to the large side group (Cl, chloride) It has a high melt viscosity hence it is difficult to process. PVC homopolymer has high strength (7. 5 to 9 psi) & brittle PVC sheets & films – blood, solution storage bags & surgical packaging 22

5. 0 Polymeric Biomaterials Polyvinylchloride (PVC)…continue PVC tubing-commonly used in intravenous (IV) administration, dialysis devices, catheters, & cannulae n Specific Properties: Excellent resistance to abrasion, good dimensional stability, high chemical resistance Note: n To prevent thermal degradation of the polymer (HCl could be released), thermal stabilizers -metallic soaps/salts are incorporated n Di-2 -ethylhexylphthalate (DEHP or DOP) is used in medical PVC formulation. n 23
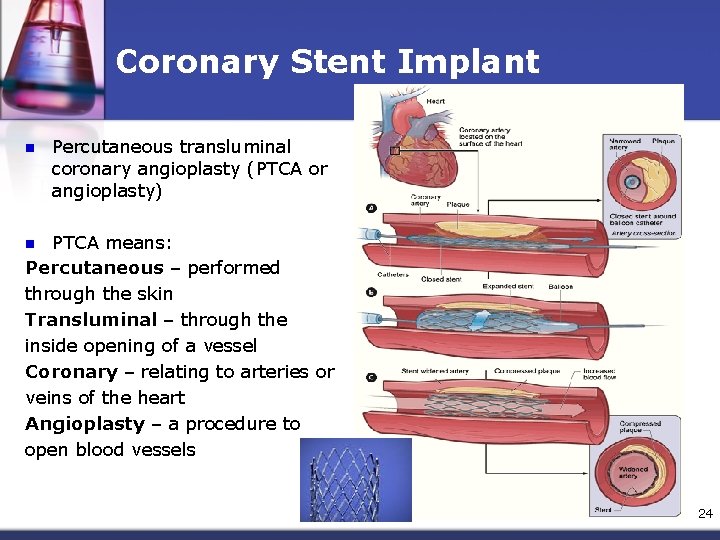
Coronary Stent Implant n Percutaneous transluminal coronary angioplasty (PTCA or angioplasty) PTCA means: Percutaneous – performed through the skin Transluminal – through the inside opening of a vessel Coronary – relating to arteries or veins of the heart Angioplasty – a procedure to open blood vessels n 24

5. 0 Polymeric Biomaterials Polypropylene (PP) n High melting (165 -1770 C) & heat deflection temperature n Additives for PP such as antioxidants, light stabilizer, nucleating agents, lubricants, mold release agents, antiblock, & slip agents are formulated to improve the physical properties & processability n PP has an exceptionally high flex life & excellent environment stress-cracking resistance, hence it had been tried for finger joint prostheses with an integrally molded hinge design [Park, 1984] 25

5. 0 Polymeric Biomaterials Polypropylene (PP)…continue n PP is used to make disposable hypothermic syringes, blood oxygenator membrane, packaging for devices, solutions, and drugs, suture, artificial vascular grafts, nonwoven fabrics, etc. n Specific Properties: Low density, good chemical resistance, moisture resistance & heat resistance n Good surface hardness & dimensional stability 26

5. 0 Polymeric Biomaterials Polymethylmetacrylate (PMMA) n n n Commercial PMMA-amorphous material with good resistance to dilute alkalis & other inorganic solutions Best known for exceptional light transparency (92% transmission), high refractive index (1. 49), good weathering properties & as one of the most biocompatible polymers Used broadly in medical applications: n n blood pump & reservoir, IV system, membranes for blood dialyzer in vitro diagnostics. 27
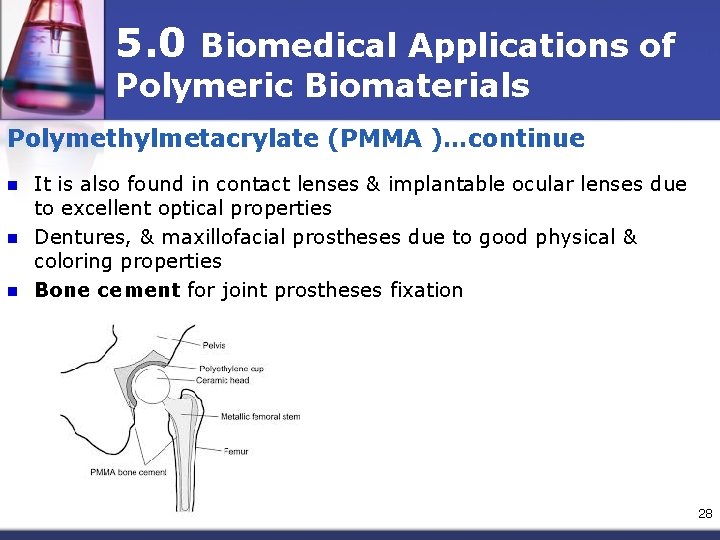
5. 0 Biomedical Applications of Polymeric Biomaterials Polymethylmetacrylate (PMMA )…continue n n n It is also found in contact lenses & implantable ocular lenses due to excellent optical properties Dentures, & maxillofacial prostheses due to good physical & coloring properties Bone cement for joint prostheses fixation 28

5. 0 Biomedical Applications of Polymeric Biomaterials Polystyrene (PS) and Its Copolymers n n n PS has good transparency, lack of color, ease of fabrication, thermal stability, low specific gravity & relatively high modulus Commonly used in tissue culture flasks, roller bottles, vacuum canisters & filterware Acrylonitrile–butadiene–styrene (ABS) copolymers are produced by 3 monomers: acrylonitrile, butadiene & styrene Resistant to common inorganic solutions, have good surface properties, and dimensional stability For IV sets, clamps, blood dialyzers, diagnostic test kits 29

5. 0 Polymeric Biomaterials Polyesters n n n Frequently found in medical applications due to their unique chemical & physical properties PET (polyethyleneterephthalate) is so far the most important Biomedical applications-as artificial vascular graft, sutures & meshes. It is highly crystalline with high melting temperature, hydrophobic & resistant to hydrolysis in dilute acids Polycaprolactone is crystalline & has a low melting temperature. Soft matrix or coating for conventional polyester fibers. Tissue engineering 30

Polyamides (Nylon) n n n Flexibility of carbon chain contributes to molecular flexibility, low melt viscosity and high lubricity Nylons are hygroscopic and lose their strength in vivo when implanted Poly (p-phenylene terephthalate) commonly known as Kevlar® Very good mechanical properties, good thermal properties, good chemical resistance, permeable to gases Tubes for intracardiac catheters, surgical sutures, dialysis devices components, heart mitral valves, sutures 31

Polytetrafluoroethylene (PTFE) n n n Commonly known as Teflon® The polymer is highly crystalline, high density, low modulus of elasticity & tensile strength It also has a very low surface tension & friction coefficient (0. 1) Specific Properties: Chemical inertness, exceptional weathering & heat resistance, nonadhesive, very low coefficient of friction Application: Vascular & auditory prostheses, catheters, tubes 32

5. 0 Polymeric Biomaterials Rubbers n n n Rubbers have been used for the fabrication of implants Natural rubber is compatible with blood in pure form Crosslinking by x-ray & organic peroxides produces rubber with superior blood compatibility Silicone rubber developed for medical use Good thermal stability, resistance to atmospheric & oxidative agents, physiological inertness Burn treatment, shunt, mammary prostheses, maxillofacial implants 33

5. 0 Polymeric Biomaterials Polyurethanes n n Polyurethanes are usually thermosetting polymers: they are widely used to coat implants polyurethane rubber is quite strong and has good resistance to oil and chemicals Exceptional resistance to abrasion, resistance to breaking, very high elasticity modulus at compression traction & sheering remarkable Adhesives, dental materials, blood pumps, artificial heart & skin 34

5. 0 Polymeric Biomaterials Polyacetal, Polysulfone & Polycarbonate n n n Polyacetals & polysulfones are being tested as implant materials Polycarbonates have found their applications in the heart/lung assist devices & food packaging Polyacetals have reasonably high molecular weight & excellent mechanical properties Excellent resistance to most chemicals & to water over wide temperature ranges Hard tissue replacement 35

5. 0 Polymeric Biomaterials Polyacetal, Polysulfone & Polycarbonate n n n Polysulfones have a high thermal stability due to the bulky side groups (therefore, they are amorphous) & rigid main backbone chains Polycarbonates are tough, amorphous, & transparent polymers Excellent mechanical & thermal properties, hydrophobicity & antioxidative properties 36

5. 0 Polymeric Biomaterials Biodegradable Polymers n n n Hydrolysis of PLA yields lactic acid which is a normal byproduct of anaerobic metabolism in the human body & is incorporated in the tricarboxylic acid (TCA) cycle to be finally excreted by the body as CO 2 & water PGA biodegrades by a combination of hydrolytic scission & enzymatic (esterase) action producing glycolic acid either enter the TCA cycle or is excreted in urine and can be eliminated as CO 2 & water PLGA can be controlled from weeks to over a year by varying the ratio of monomers & the processing conditions 37

5. 0 Polymeric Biomaterials Biodegradable Polymers…(continue) n n PLA-high tensile strength & low elongation resulting in a high modulus. Application: bone fracture fixation PLGA-tissue engineered repair systems where cells are implanted within PLGA films or scaffolds PLGA-drug delivery systems in which drugs are loaded within PLGA microspheres Other-Poly-p-dioxanon: bioabsorbable polymer which can be fabricated into flexible monofilament surgical sutures 38

5. 0 Polymeric Biomaterials Summary 39

Biopolymers n Polymers are used in biomedical applications Ø Ø • Cardiovascular, Opthalmic and Orthopaedic implants Dental implants, dental cements and denture bases Low density, easily formed and can be made biocompatible. Recent development – biodegradable polymers. 40
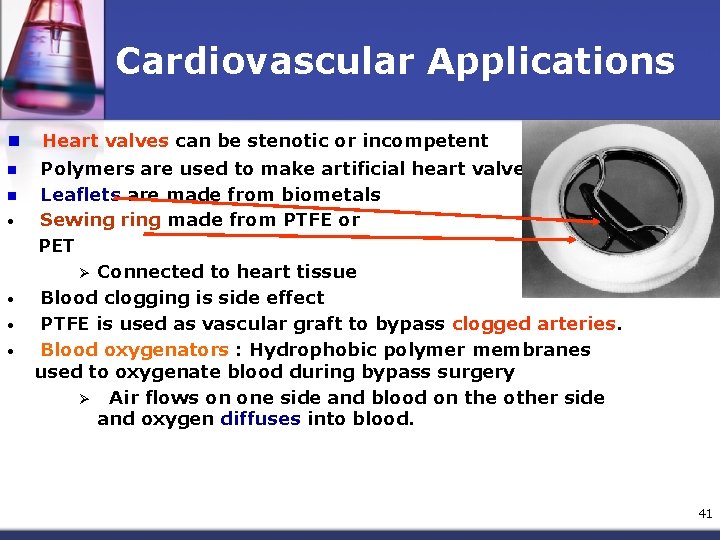
Cardiovascular Applications n n n • • Heart valves can be stenotic or incompetent Polymers are used to make artificial heart valves Leaflets are made from biometals Sewing ring made from PTFE or PET Ø Connected to heart tissue Blood clogging is side effect PTFE is used as vascular graft to bypass clogged arteries. Blood oxygenators : Hydrophobic polymer membranes used to oxygenate blood during bypass surgery Ø Air flows on one side and blood on the other side and oxygen diffuses into blood. 41

Extra: Opthalmic Applications n Eye glasses, contact lenses and Intraocular implants are • made of polymers. Hydrogel is used to make soft contact lenses Ø Absorbs water and allows snug fit Ø Oxygen permeable Ø Made of poly-HEMA Hard lenses made from PMMA Ø Not oxygen permeable Ø Mixed with Siloxanylalkyl Metacrylate and metacrylic acid to make permeable and hydrophilic. Intraocular implants are made of PMMA • Poly-HEMA-Poly(hydroxyethyl methacrylic) acid n • 42

Orthopedic Applications n Bone cement: Fills space between implant and • • bone – PMMA Ø Centrifuging and vacuum techniques minimize porosity Used in joint prosthesis (Knee and Hip replacements) Other applications: Ø Drug delivery systems: Polymer matrix with drug implanted inside the body Ø Struture materials: High tensile and knot pull strength. Ø Non-absorbable: Polypropylene, Nylon Ø Absorbable : Polyglycolic acid. 43
Tissue Engineering n n n Polymers can be synthesized and blend to suite the applications Biodegradable polymers are used as scaffolding for generation of new tissues In future, tissues can be generated in vivo or in vitro for repair or replacement. 44

Sterilization of healthcare products n n The objective of sterilization is to prevent the introduction into the body of pathogenic organisms not normally present. Sterilization can be defined as ‘the removal or destruction of all living organisms, including resistant forms such as bacterial or fungal spores’. 45

Sterilization method 46

Dry Heat n n Dry heat is not generally regarded as being suitable for plastics due to the low thermal transmission properties of plastics and the difficulty of insuring that all parts of the product have been exposed to the required temperature for an adequate time. Most plastics will degrade during prolonged dry heat sterilization. 47

Autoclaving n n Autoclaving uses saturated steam to allow lower temperatures and shorter times than in the dry heat process. Steam will penetrate well into a product, as water vapor is lost due to condensation. Allow steam to reach all surfaces and for items to reach the required temperature for sterilization. The temperatures and times: lower temperatures must be held for longer times), but it is common for the temperature to be around 121 o. C 48

Autoclaving n n used significantly in hospitals for the sterilization of repeated use articles. not the predominant method in the commercial sterilization of medical devices because of the difficulties involved with autoclaving packaged products. 49

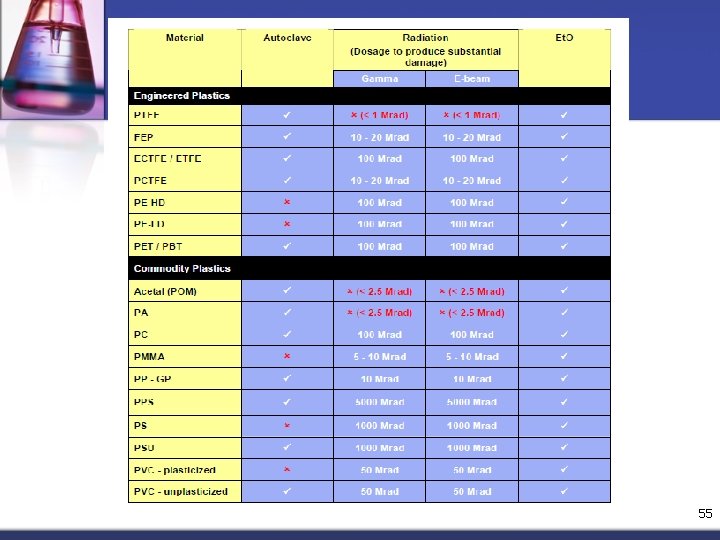
Irradiation n n Generated by either gamma rays from a Cobalt (Co 60) source or an electron beam (E -beam). The cost of capital equipment is great, but high throughputs will improve the return on investment. 50

n n n Dosage for either process is measured in Megarad (Mrad) and as a general rule a radiation dose of around 2. 5 Mrad will sterilize clean articles in air. very effective for fully packaged and sealed singleuse items (most plastic films are effectively transparent to radiation) where only one radiation dose is required. 51

n n lead to changes in the tensile strength, elongation at break and impact strength (depend both on the basic polymer and any additives used). Irradiated devices are completely safe to handle and can be released and used immediately after sterilization. 52

Ethylene oxide n n n Ethylene oxide is a powerful alkylating agent and is regarded by the EPA as a toxic and possibly carcinogenic gas (exposure to Et. O is regulated by the EPA and OSHA). When mixed with air, Et. O is not only flammable but can also be explosive. The effectiveness of Et. O sterilization depends on many variables such as time, gas concentration, temperature and relative humidity (necessary to moisten bacteria to insure effective destruction). monitoring Et. O sterilization difficult and time consuming 53

n n Some plastics are relatively permeable to Et. O and the process can then be used to sterilize fully packaged articles by using thin packaging films, such as PE. Et. O gas to enter the package and sterilize the contents. The packaging film must also be permeable to water vapor and air to be effective. 54
55

Standards AAMI and ISO have produced a range of standards for sterilization such as: • ISO 11135 - Medical devices - Validation and routine control of ethylene oxide sterilization. • ISO 11137 - Medical devices - Validation and routine control of radiation sterilization. • ISO 11737 - Sterilization of medical devices - Microbiological methods. o Part 1: Estimation of population of microorganisms on products. o Part 2: Tests of sterility performed in the validation of a sterilization process. n 56
- Slides: 56