Biomaterials Artificial Organs and Tissue Engineering Chapter 11

Biomaterials, Artificial Organs and Tissue Engineering Chapter 11 Biomedical Hydrogels Jason A. Burdick 1 and Molly M. Stevens 2 1 Department of Bioengineering, University of Pennsylvania, Philadelphia, USA 2 Department of Materials, Imperial College London, UK

Hydrogels as biomaterials • Hydrogels are insoluble water-swollen networks that are being widely investigated for biomedical applications such as drug delivery and tissue engineering. Table 11. 1 Advantages and disadvantages of using hydrogels in biomedical applications Advantages Wide range of chemistries available Typically biocompatible High water content Potentially injectable for minimally invasive implantation Disadvantages Poor mechanical properties Difficult to sterilise
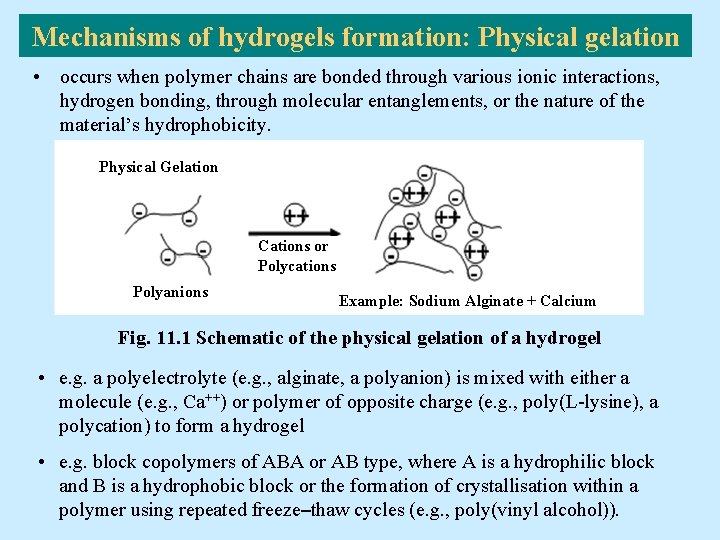
Mechanisms of hydrogels formation: Physical gelation • occurs when polymer chains are bonded through various ionic interactions, hydrogen bonding, through molecular entanglements, or the nature of the material’s hydrophobicity. Physical Gelation Cations or Polycations Polyanions Example: Sodium Alginate + Calcium Fig. 11. 1 Schematic of the physical gelation of a hydrogel • e. g. a polyelectrolyte (e. g. , alginate, a polyanion) is mixed with either a molecule (e. g. , Ca++) or polymer of opposite charge (e. g. , poly(L-lysine), a polycation) to form a hydrogel • e. g. block copolymers of ABA or AB type, where A is a hydrophilic block and B is a hydrophobic block or the formation of crystallisation within a polymer using repeated freeze–thaw cycles (e. g. , poly(vinyl alcohol)).
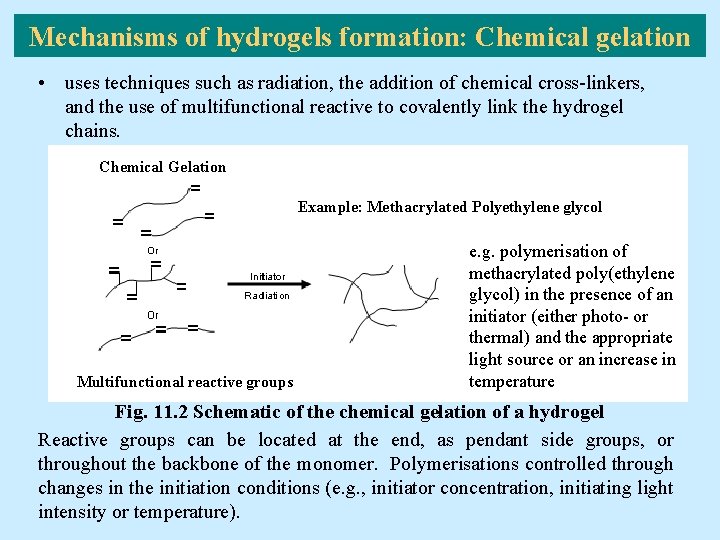
Mechanisms of hydrogels formation: Chemical gelation • uses techniques such as radiation, the addition of chemical cross-linkers, and the use of multifunctional reactive to covalently link the hydrogel chains. Chemical Gelation Example: Methacrylated Polyethylene glycol Multifunctional reactive groups e. g. polymerisation of methacrylated poly(ethylene glycol) in the presence of an initiator (either photo- or thermal) and the appropriate light source or an increase in temperature Fig. 11. 2 Schematic of the chemical gelation of a hydrogel Reactive groups can be located at the end, as pendant side groups, or throughout the backbone of the monomer. Polymerisations controlled through changes in the initiation conditions (e. g. , initiator concentration, initiating light intensity or temperature).

Mechanical Properties • Hydrogels are typically weak compared to other polymers due to the high water content. • Mechanical properties altered by cross-linking density of the polymer, polymerisation conditions during network formation and hydrogel swelling. • Tensile testing and dynamic mechanical analysis (in liquid) measure the rubber elastic and viscoelastic behaviors, respectively.

Mass transport properties • Dependent on swelling, pores within gel, and molecular weight, Stokes radii and charge of diffusing molecules. • Swelling is also a dynamic process that changes with time if the hydrogel was not formed at the equilibrium swelling ratio or if the structure changes with hydrogel degradation.

Degradation properties • Mechanisms include hydrolysis, enzymatic cleavage and dissolution. • Degradation controls properties such as hydrogel mesh size which is important in the release of entrapped molecules and the diffusion of extracellular matrix components produced by encapsulated cells.
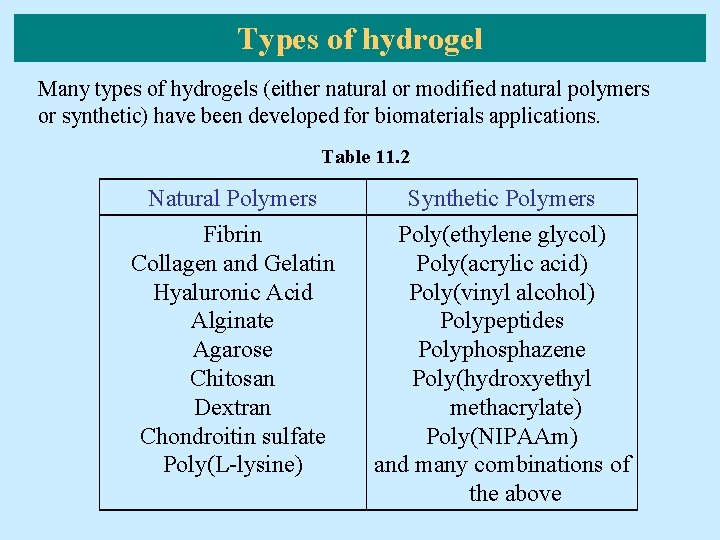
Types of hydrogel Many types of hydrogels (either natural or modified natural polymers or synthetic) have been developed for biomaterials applications. Table 11. 2 Natural Polymers Fibrin Collagen and Gelatin Hyaluronic Acid Alginate Agarose Chitosan Dextran Chondroitin sulfate Poly(L-lysine) Synthetic Polymers Poly(ethylene glycol) Poly(acrylic acid) Poly(vinyl alcohol) Polypeptides Polyphosphazene Poly(hydroxyethyl methacrylate) Poly(NIPAAm) and many combinations of the above

Collagen • Natural protein abundant in the body (e. g. tendons and ligaments). • Cross-linked with various techniques (e. g. , glutaraldehyde, carbodiimide, photooxidation) to improve the physical properties (mechanics and degradation). • Controllable resorbability in vivo. • Sterilisation by gamma irradiation, ethylene oxide treatment, or electron beam irradiation.

Hyaluronic acid • A non-sulfated glycosaminoglycan found in vivo which consists of repeating units of N-acetyl-D-glucosamine and Dglucuronic acid. • Degradation by hyaluronidases. • Cross-linked with various techniques (e. g. , photocross-linking, aldehyde cross-linking, carbodiimide cross-linking) to improve the physical properties (mechanics and degradation).

Alginate • A natural polysaccharide (from seaweed) composed of a-Dmannuronic acid and b-L-guluronic acid. • Ionic cross-linking occurs in the presence of various divalent cations (e. g. , Ca++, Mg++) by cross-linking the carboxylate groups of the guluronate groups on the polymer backbone.

Poly(ethylene glycol) • Widely used due to the extreme hydrophilicity and biocompatibility of the polymer. • Modification possible with acrylate or methacrylate groups that allow for hydrogel formation in the presence of initiators by a thermally or photoinitiated polymerisation. • Hydrolytically degradable hydrogels can be formed with introduction of a-hydroxy acid groups (e. g. , lactic acid) between the polymer and functional group.

Poly(N-isopropylacrylamide) • Exhibits a reversible phase transition around 32 C where the chains go from water soluble to water insoluble. The phase change is altered by the addition of hydrophobic and hydrophilic groups. • Increase in gelation occurs after in vivo injection (37 C).
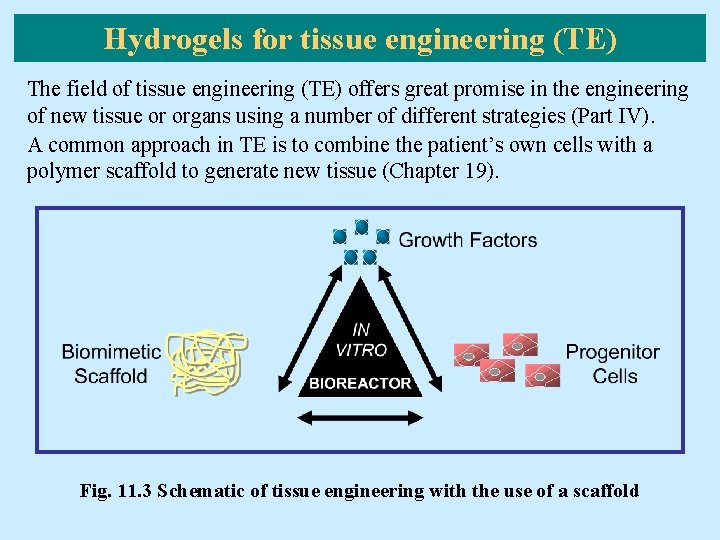
Hydrogels for tissue engineering (TE) The field of tissue engineering (TE) offers great promise in the engineering of new tissue or organs using a number of different strategies (Part IV). A common approach in TE is to combine the patient’s own cells with a polymer scaffold to generate new tissue (Chapter 19). Fig. 11. 3 Schematic of tissue engineering with the use of a scaffold

Design criteria for hydrogel scaffolds for TE Physical Properties • Gel formation mechanisms and dynamics (to allow minimally invasive delivery) • Mechanical properties (appropriate integrity and strength) • Degradation rates (appropriate to application) Mass Transport Properties • Diffusion requirements (appropriate diffusion of nutrients and metabolites) Biological Properties • Biocompatibility • Promotion of cell adhesion, proliferation and differentiation

References • K. S. Anseth, C. N. Bowman, L. Brannon-Peppas, Mechanical Properties of Hydrogels and Their Experimental Determination, Biomaterials, 17, 1996, 1647– 1657. • A. Atala and R. P. Lanza (editors), Methods of Tissue Engineering, 2002, Elsevier Science. • T. Canal, N. A. Peppas, Correlation Between Mesh Size and Equilibrium Degree of Swelling of Polymeric Networks, Journal of Biomedical Materials Research, 23, 1989, 1183– 1193. • J. L. Drury, D. J. Mooney, Hydrogels for tissue engineering: scaffold design variables and applications, Biomaterials, 24, 2003, 4337– 4351. • A. S. Hoffman, Hydrogels for Biomedical Applications, Advanced Drug Delivery Reviews, 43, 2002, 3– 12. • R. P. Lanza, R. Langer and W. L. Chick (editors), Principles of Tissue Engineering, 1997, R G Landes Company and Academic Press, Inc. • K. Y. Lee, D. J. Mooney, Hydrogels for Tissue Engineering, Chemical Reviews, 101, 2001, 1869– 1879. • B. D. Ratner, A. S. Hoffman, F. J. Schoen, J. E. Lemons (editors), Biomaterials Science, 1996, Elsevier Science.
- Slides: 16