Biomaterials and Protein Adsorption Examples of Biomaterials Medical

Biomaterials and Protein Adsorption
Examples of Biomaterials • • Medical implants Contact lenses Drug delivery systems Scaffolding for tissue regeneration

Proteins are amphiphilic molecules in an aqueous milieu • Polypeptides are amphiphilic molecules • BUT -- The human body is 90% water! • SO : hydrophobic regions of proteins seek refuge in supramolecular configurations that minimize their exposure to water
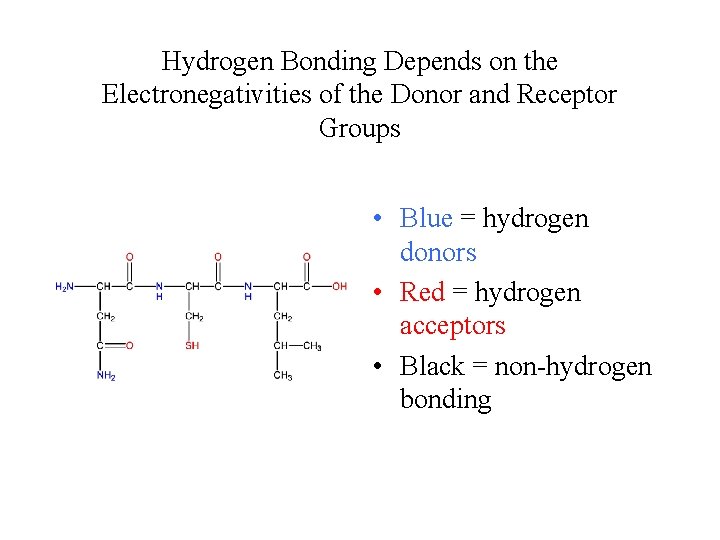
Hydrogen Bonding Depends on the Electronegativities of the Donor and Receptor Groups • Blue = hydrogen donors • Red = hydrogen acceptors • Black = non-hydrogen bonding
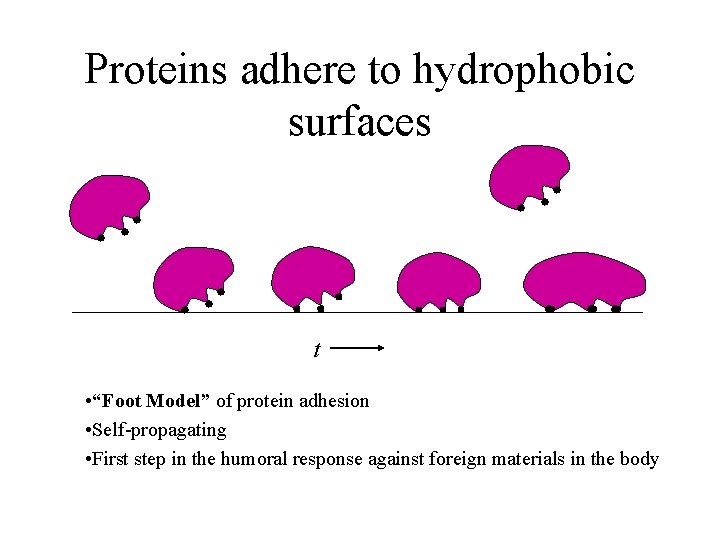
Proteins adhere to hydrophobic surfaces t • “Foot Model” of protein adhesion • Self-propagating • First step in the humoral response against foreign materials in the body
Design of Biomaterials Surfaces • Hydrophilicity inhibits protein adsorption, however: • Some cell adhesion may be desirable • Compliance is a key consideration • Solution? Polymers, of course! s
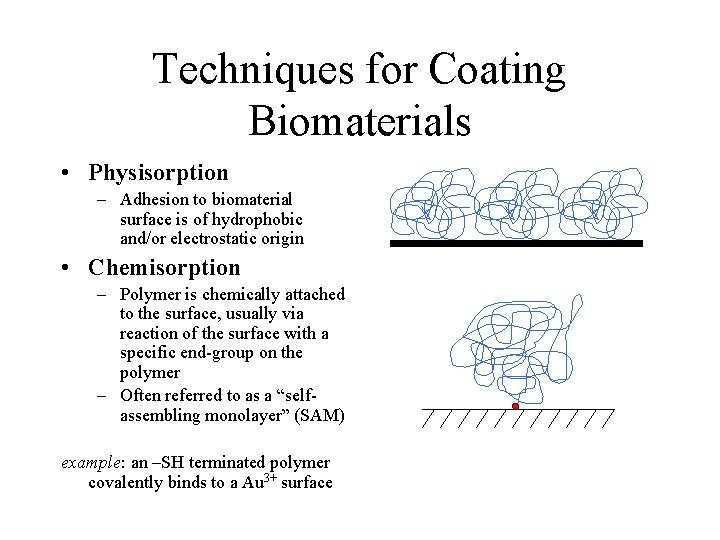
Techniques for Coating Biomaterials • Physisorption – Adhesion to biomaterial surface is of hydrophobic and/or electrostatic origin • Chemisorption – Polymer is chemically attached to the surface, usually via reaction of the surface with a specific end-group on the polymer – Often referred to as a “selfassembling monolayer” (SAM) example: an –SH terminated polymer covalently binds to a Au 3+ surface

Polymer Brushes • A “brush” is formed when the spacing d between end-grafted polymers is less than twice the Flory radius, RF, where RF ~ a. N 3/5 and a is the monomer size
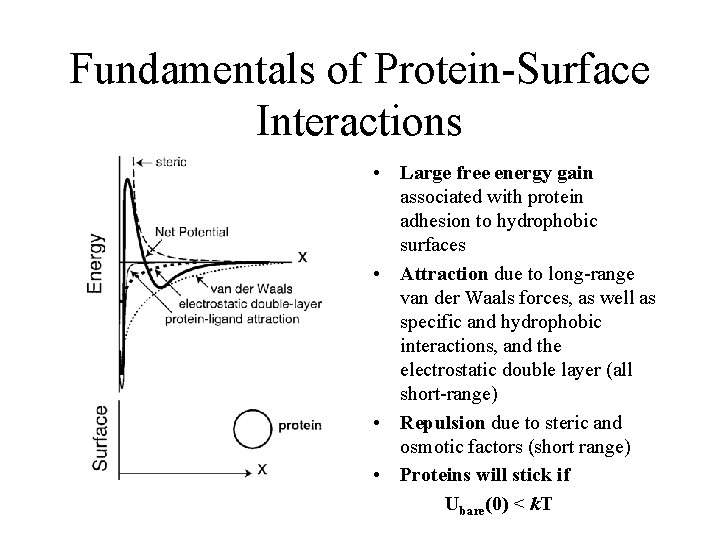
Fundamentals of Protein-Surface Interactions • Large free energy gain associated with protein adhesion to hydrophobic surfaces • Attraction due to long-range van der Waals forces, as well as specific and hydrophobic interactions, and the electrostatic double layer (all short-range) • Repulsion due to steric and osmotic factors (short range) • Proteins will stick if Ubare(0) < k. T

Steric and Osmotic Factors • Atoms and molecules take up a finite amount of space which cannot be occupied by other elements – i. e. they introduce an excluded volume – Dense packing, rotations, and/or rearrangements may therefore not be energetically allowed: i. e. steric hindrance – Crowding leads to an increase in the internal energy and thus the osmotic pressure

The Free Energy Profile of the Brush has Two Minima a) brush potential, Ubrush(z) b) attractive [primarily] van der Waals potential Uvd. W(z) c) net interaction potential
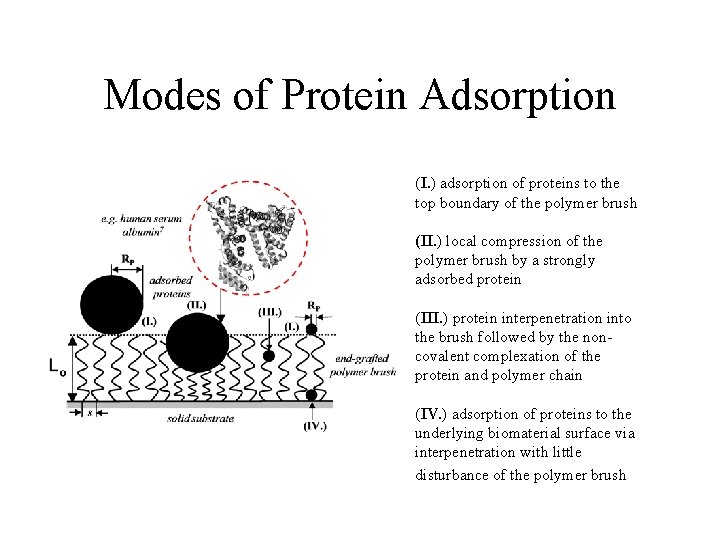
Modes of Protein Adsorption (I. ) adsorption of proteins to the top boundary of the polymer brush (II. ) local compression of the polymer brush by a strongly adsorbed protein (III. ) protein interpenetration into the brush followed by the noncovalent complexation of the protein and polymer chain (IV. ) adsorption of proteins to the underlying biomaterial surface via interpenetration with little disturbance of the polymer brush
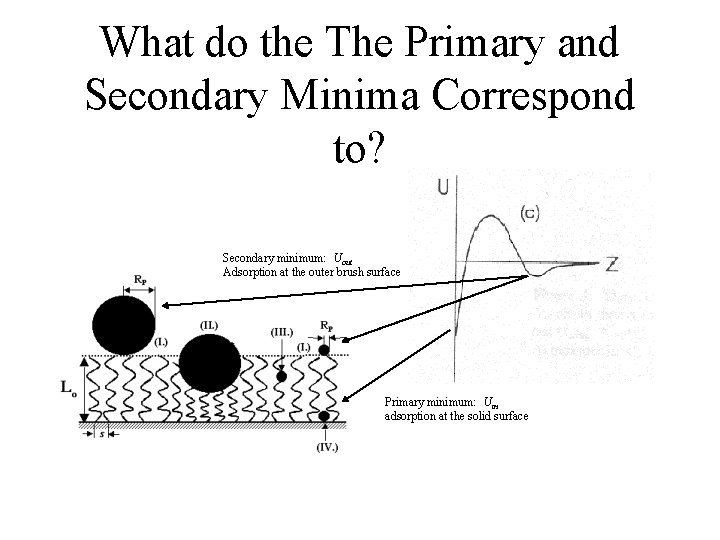
What do the The Primary and Secondary Minima Correspond to? Secondary minimum: Uout Adsorption at the outer brush surface Primary minimum: Uin adsorption at the solid surface

Osmotic vs Entropic Forces The brush thickness, L depends on a balance of forces: Osmotic Force Elastic Force At Equilibrium or Brush thickness: where Monomer volume fraction: So the corresponding force and free energy per chain: Variables: And the corresponding osmotic pressure:

Secondary Adsorption Occurs when Uout < -k. T • Since there is no energy barrier, it is only possible to control Uout thermodynamically • Uout Uvd. W(L) • Because penetration of the brush requires chain compression, large proteins will preferentially undergo secondary adsorption so long as Uvd. W(L) < -k. T • For a rod-like protein (fibrinogen, e. g. ) of radius R and length H, suppression of secondary adsorption may only be achieved if: Where A is the Hamaker constant, A ~ 10 -21 J for proteins interacting with organic materials

Primary Adsorption Occurs when Uin < -k. T ** The presence of an energy barrier enables both thermodynamic and kinetic control When Rp << L : There is negligible effect on When Rp >> LApproach : to the surface results in compression of the brush and an increase in osmotic pressure where The rate constant for adsorption: and Where is the width of the energy barrier and D is the diffusion coefficient And the free energy barrier, U * for primary adsorption: Finally: Where Uads is the interaction potential of the adsorbed protein at the bare surface

Methods for Counteracting Protein-Surface Interaction with Polymer Coatings Uout may be manipulated by varying N or s Uin is primarily controlled by varying s • Dense polymer coatings (low s) • Long polymer chains (large N) R N d s
Poly(ethylene oxide) (PEO) in Biomaterials • The most extensively used polymer for biomaterial surface coatings, because: – Completely water-soluble – Creates an extensive Hbonding network – Helical conformation – Proven to be extremely protein resistant – Capable of being functionalized for ligandreceptor specificity • However: – Poor mechanical stability – Protein adhesioin reported under certain conditions
- Slides: 18