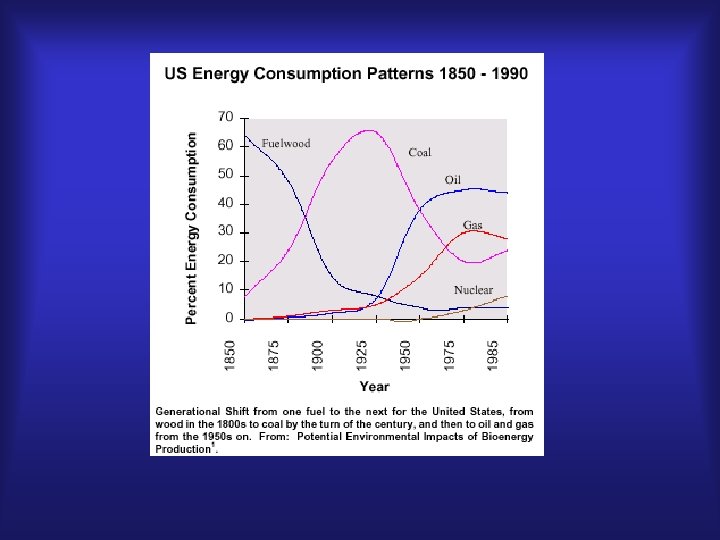
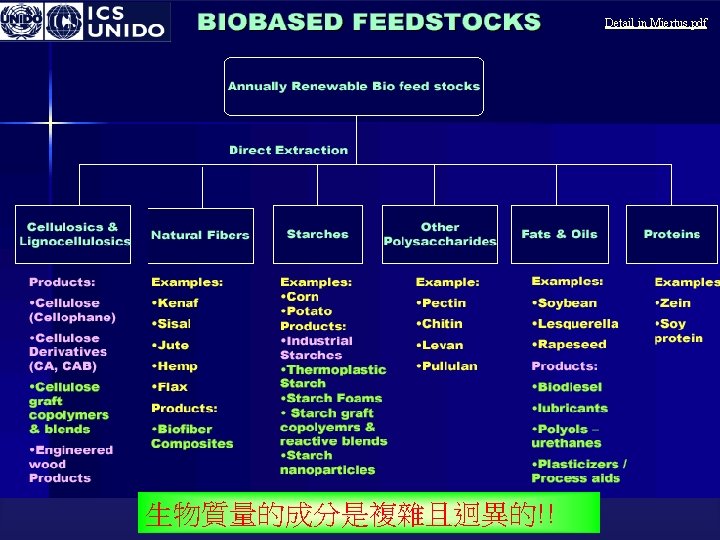
Biomass of green chemistry biomass energy Introduction and

Biomass of green chemistry (biomass energy) 交通大學應用化學系 鄭至玉, 李耀坤

Introduction and application of biomass 1. What is Biomass Energy? 2. Who Can Use Bioenergy? 3. Who Should be Developing or Using Biomass? 4. Why Biomass? 5. Socioeconomic Benefits of Biomass 6. Current Industrial Applications of Biomass 7. How Can We Use Biomass? 8. Sources & Barriers of Biomass 9. Costs of Biomass More information from http: //oahu. ol. mala. bc. ca/RE

Why do we need so badly?

Global carbon cycling and the importance of renewable resources 我們在 10年之間, 用掉了佰萬年的積蓄!!
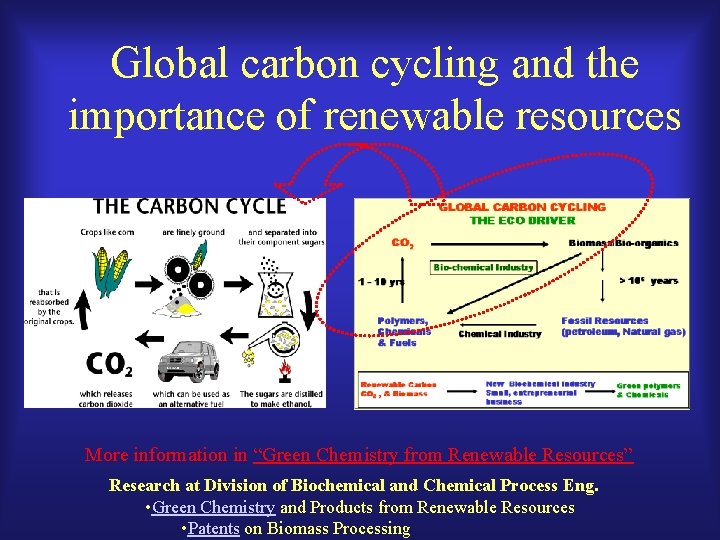
Global carbon cycling and the importance of renewable resources More information in “Green Chemistry from Renewable Resources” Research at Division of Biochemical and Chemical Process Eng. • Green Chemistry and Products from Renewable Resources • Patents on Biomass Processing

Any other energy can we get? Green and renewable
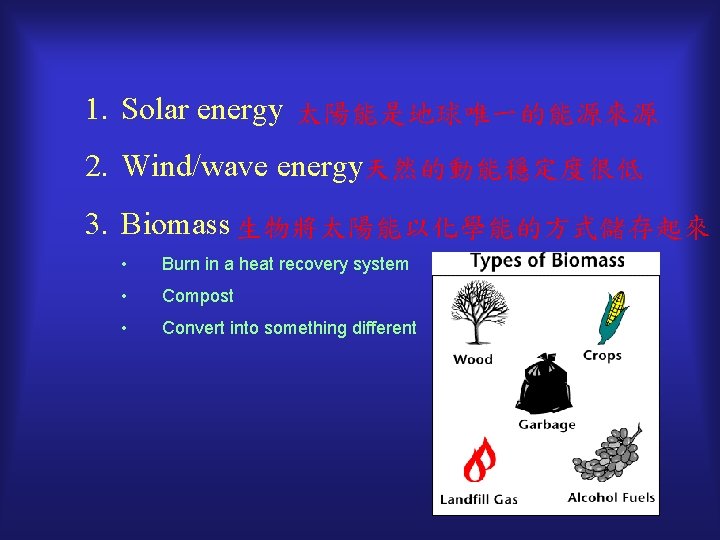
1. Solar energy 太陽能是地球唯一的能源來源 2. Wind/wave energy天然的動能穩定度很低 3. Biomass 生物將太陽能以化學能的方式儲存起來 • Burn in a heat recovery system • Compost • Convert into something different
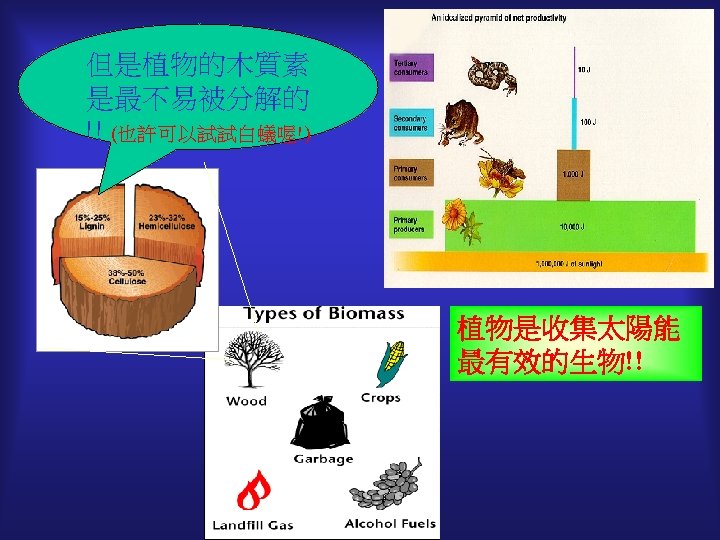

What else can we do with biomass?
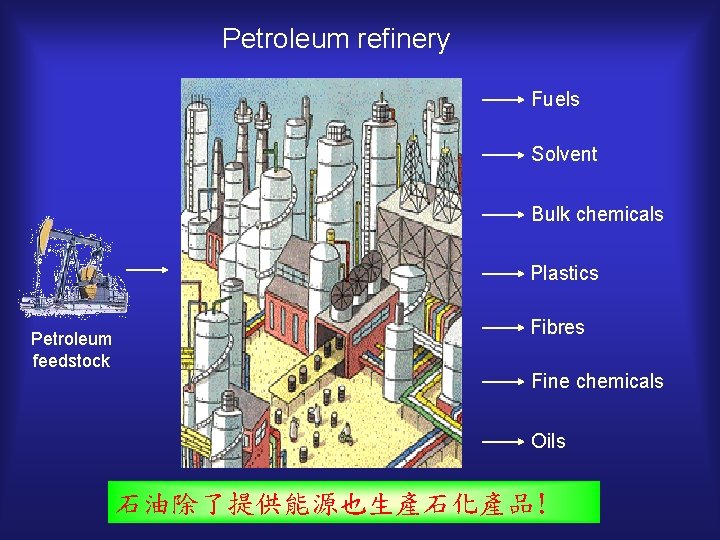
Petroleum refinery Fuels Solvent Bulk chemicals Plastics Petroleum feedstock Fibres Fine chemicals Oils 石油除了提供能源也生產石化產品!
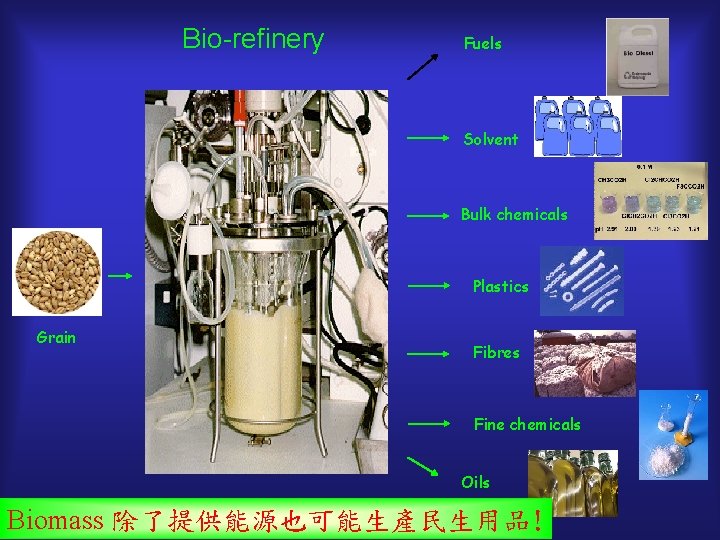
Bio-refinery Fuels Solvent Bulk chemicals Plastics Grain Fibres Fine chemicals Oils Biomass 除了提供能源也可能生產民生用品!

• The problem Petrochemicals – Scarce resource – CO 2 pollution – Strategic and political weapon – Low security of supply – Cheap • The solution Biomass (ethanol and diesel) – Renewable – CO 2 neutral – Can be grown and exploited anywhere – High security of supply – Expensive (yet) – Processing to gasoline or diesel can be combined with use as solid fuel 石油的優點, biomass取代! 石油的缺點, biomass解決! Detail in Felby. ppt
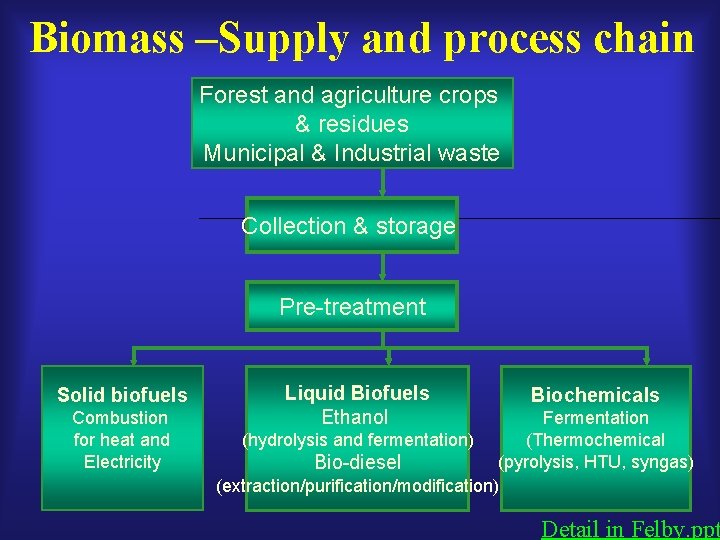
Biomass –Supply and process chain Forest and agriculture crops & residues Municipal & Industrial waste Collection & storage Pre-treatment Solid biofuels Combustion for heat and Electricity Liquid Biofuels Ethanol Biochemicals Fermentation (hydrolysis and fermentation) (Thermochemical (pyrolysis, HTU, syngas) Bio-diesel (extraction/purification/modification) Detail in Felby. ppt

Biomass based energy • Plant oils –Biodiesel • Solid biomass –Heat and electricity • Fermentable sugars –Starch and cellulose for ethanol production Detail in Felby. ppt

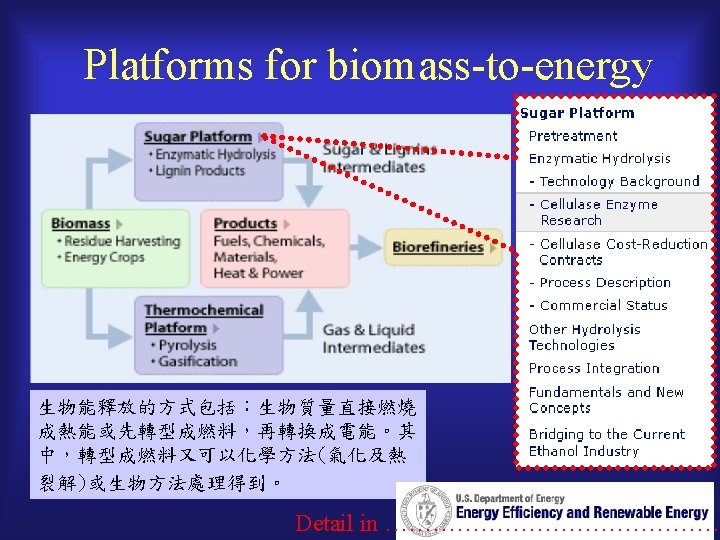
Platform of biomass energy
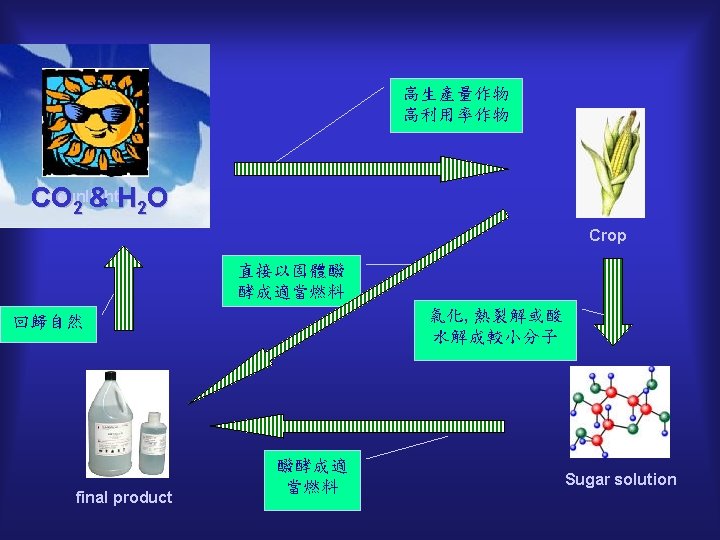


The steps of ethanol production by fermentation The production of ethanol by fermentation involves four major steps: (a) the growth, harvest and delivery of raw material to an alcohol plant; (b) the pre-treatment or conversion of the raw material to a substrate suitable for fermentation to ethanol; (c) fermentation of the substrate to alcohol, and purification by distillation; and (d) treatment of the fermentation residue to reduce pollution and to recover by-products.

Three types of raw materials for bioethanol The many and varied raw materials for bioethanol production can be conveniently classified into three types: (a) sugar from sugarcane, sugar beet and fruit, which may be converted to ethanol directly; (b) starches from grain and root crops, which must first be hydrolysed to fermentable sugars by the action of enzymes; and (c) cellulose from wood, agricultural wastes etc. , which must be converted to sugars using either acid or enzymatic hydrolysis. http: //www. viscostar. dk/application/food. htm
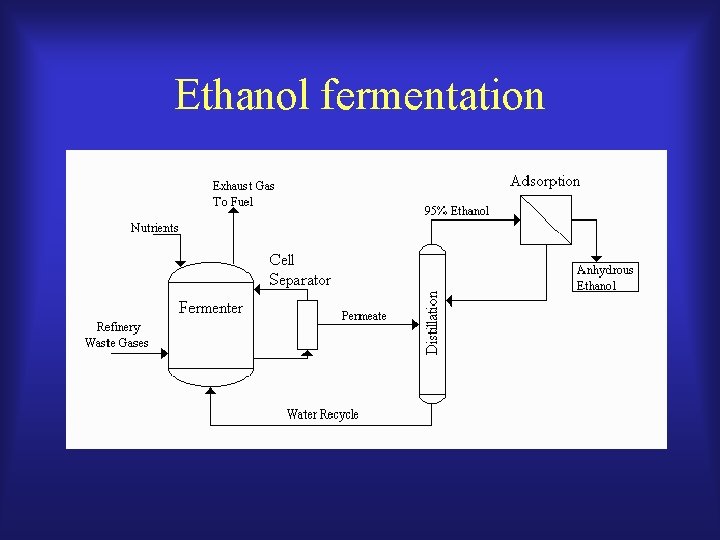
Ethanol fermentation
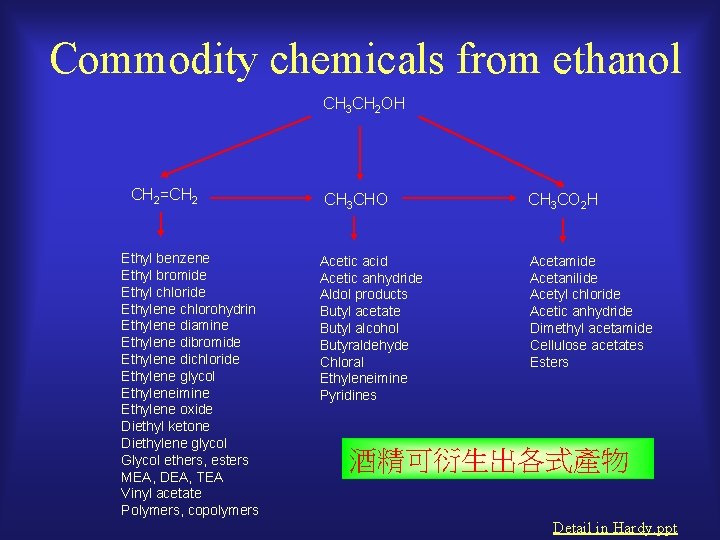
Commodity chemicals from ethanol CH 3 CH 2 OH CH 2=CH 2 Ethyl benzene Ethyl bromide Ethyl chloride Ethylene chlorohydrin Ethylene diamine Ethylene dibromide Ethylene dichloride Ethylene glycol Ethyleneimine Ethylene oxide Diethyl ketone Diethylene glycol Glycol ethers, esters MEA, DEA, TEA Vinyl acetate Polymers, copolymers CH 3 CHO Acetic acid Acetic anhydride Aldol products Butyl acetate Butyl alcohol Butyraldehyde Chloral Ethyleneimine Pyridines CH 3 CO 2 H Acetamide Acetanilide Acetyl chloride Acetic anhydride Dimethyl acetamide Cellulose acetates Esters 酒精可衍生出各式產物 Detail in Hardy. ppt

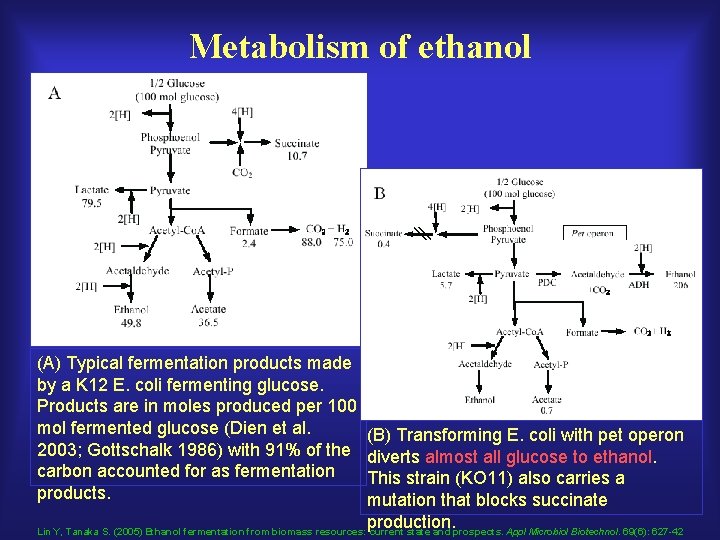
Metabolism of ethanol (A) Typical fermentation products made by a K 12 E. coli fermenting glucose. Products are in moles produced per 100 mol fermented glucose (Dien et al. 2003; Gottschalk 1986) with 91% of the carbon accounted for as fermentation products. (B) Transforming E. coli with pet operon diverts almost all glucose to ethanol. This strain (KO 11) also carries a mutation that blocks succinate production. Lin Y, Tanaka S. (2005) Ethanol fermentation from biomass resources: current state and prospects. Appl Microbiol Biotechnol. 69(6): 627 -42

Metabolism of xylose to ethanol Dien BS, Cotta MA, Jeffries TW. (2003) Bacteria engineered for fuel ethanol production: current status. Appl Microbiol Biotechnol. 63(3): 258 -66.
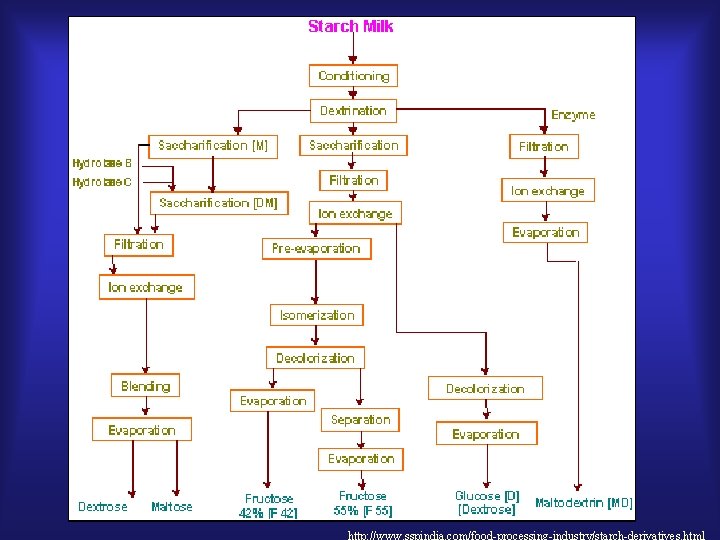
http: //www. sspindia. com/food-processing-industry/starch-derivatives. html

Hexoses Hydrolysis (Chemical) Pentoses Lignin Fermentation Alcohols (ethyl-, butyl, isopropyl-) Polyols (glyceral, ethylene glycol) Ketones (acetone) Acids (acetic, lactic, butyric) Yeast Dehydrogenation Hydrolysis Hydrogenation Crystallisation Hydroxymethylfurfural Levulinic acid Polyols Glucose Fermentation Dehydration Hydrogenation Crystallisation Yeast Furfural Polyols (Xylitol) Xylose Hydrogenation Hydrolysis Oxidation Phenol derivatives, hydrocarbons Phenol derivatives, catechols Vanillin 善用不同生物質量的成分, 調整 製程方法可製備出多樣產物!! Detail in Hardy. ppt

Microbial Conversion Dear God: I pray on bended knee’s, That all my syntheses, Will never be inferior, To those conducted by bacteria Organic Chemists Prayer (unknown origin)

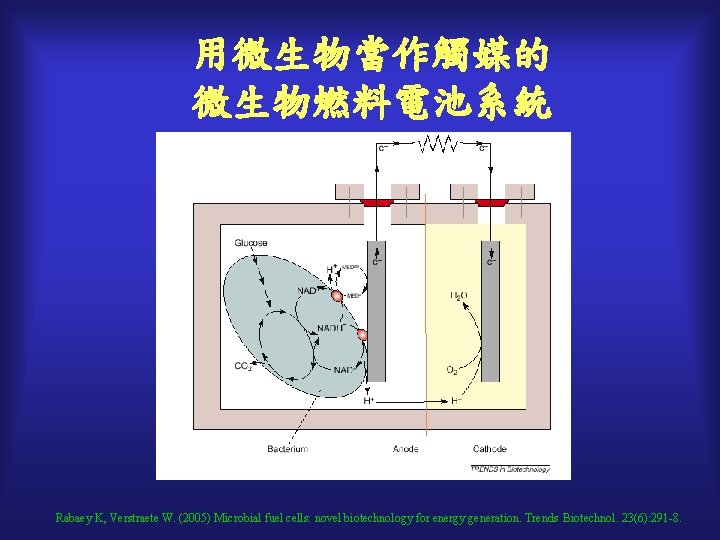
用微生物當作觸媒的 微生物燃料電池系統 Rabaey K, Verstraete W. (2005) Microbial fuel cells: novel biotechnology for energy generation. Trends Biotechnol. 23(6): 291 -8.
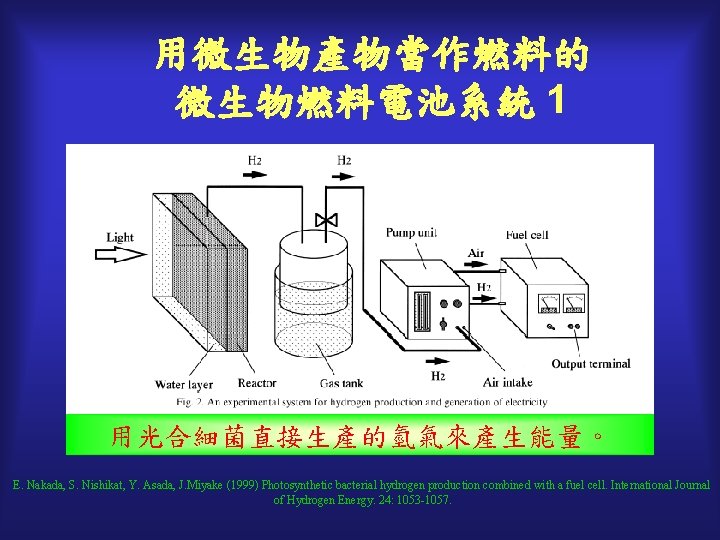
用微生物產物當作燃料的 微生物燃料電池系統 1 用光合細菌直接生產的氫氣來產生能量。 E. Nakada, S. Nishikat, Y. Asada, J. Miyake (1999) Photosynthetic bacterial hydrogen production combined with a fuel cell. International Journal of Hydrogen Energy. 24: 1053 -1057.

用微生物產物當作燃料的 微生物燃料電池系統 2 Microbial Fuel Cell: High Yield Hydrogen Source And Wastewater Cleaner http: //www. sciencedaily. com/releases/2005/04/050422165917. htm

A case in Japan

Burden on commercial power grid by introduction of variable natural power source
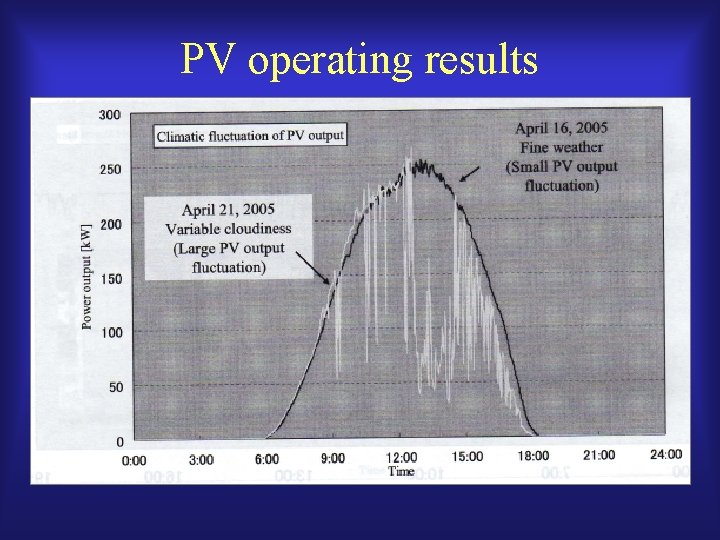
PV operating results

Expo site and new energy consortium Expo Site Members of the New Energy Consortium and Their Poles
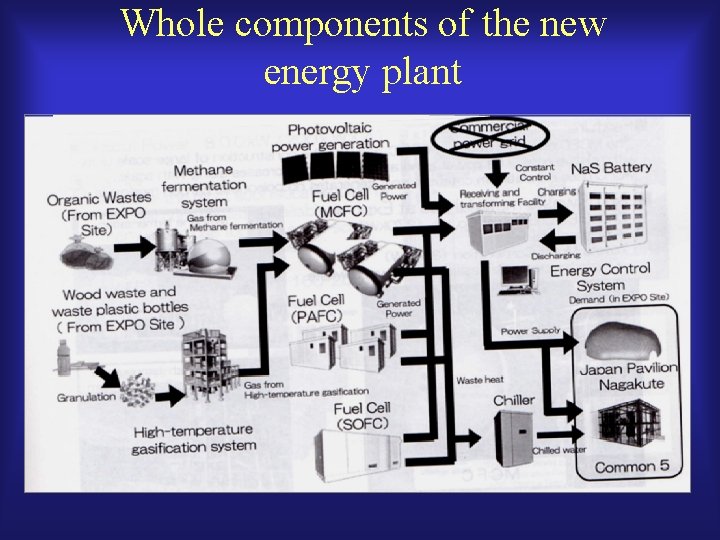
Whole components of the new energy plant

State of demonstrative project in nagakute Expo site Na. S battery 500 KW MCFC 270 KW PAFC 200 KW x 4 SOFC 25 KW Bifacial single-crystaline Photovoltaic power generation Multi-crystaline Photovoltaic Amorphous silicon Photovoltaic power generation PV: 30 + 100 + 200 KW
- Slides: 42