Biomass and Fossil Fuels What is Biomass Biomass

Biomass and Fossil Fuels • What is Biomass? • Biomass Fossil Fuels • Burning Fossil Fuels and the Greenhouse affect • Fuel from Biomass • Acid Precipitation and p. H

Biomass • Is biological material from living, or recently living organisms – E. g. wood, waste, plants and animals • This material is usually recycled (broken down and released) allowing the material to be reused

Biomass Fossil Fuels • However some of the biomass was trapped in places without oxygen – Oxygen is needed to break down organic (living) matter • Over time with pressure and heat, this trapped biomass is converted into fossil fuels (oil, gas and coal)

Burning Fossil Fuels • The burning of fossil fuels is the world’s leading method of producing energy • In the process, Oxygen is used to produce energy and Carbon Dioxide (CO 2)

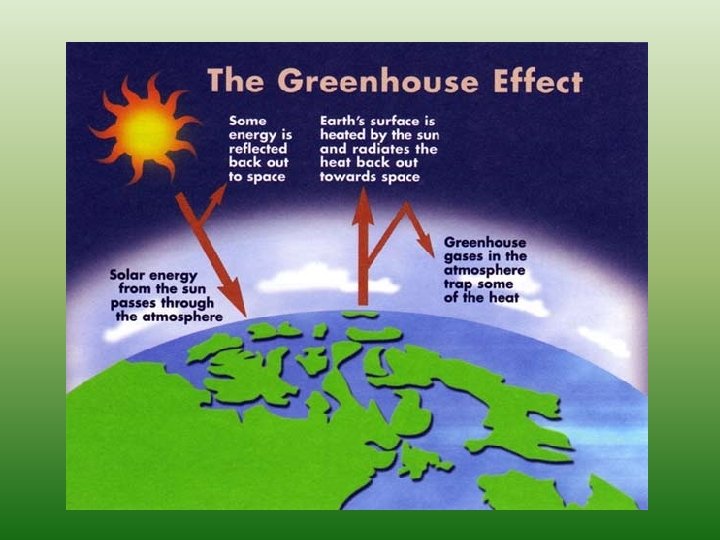
Burning Fossil Fuels • Fossil fuels have been accumulating for Billions of years however, significant portions of the Earths reserves have been burned by humans over the last 100 years • It’s widely believed that the release of CO 2 is increasing the “Greenhouse Effect” which is responsible for Global Warming.

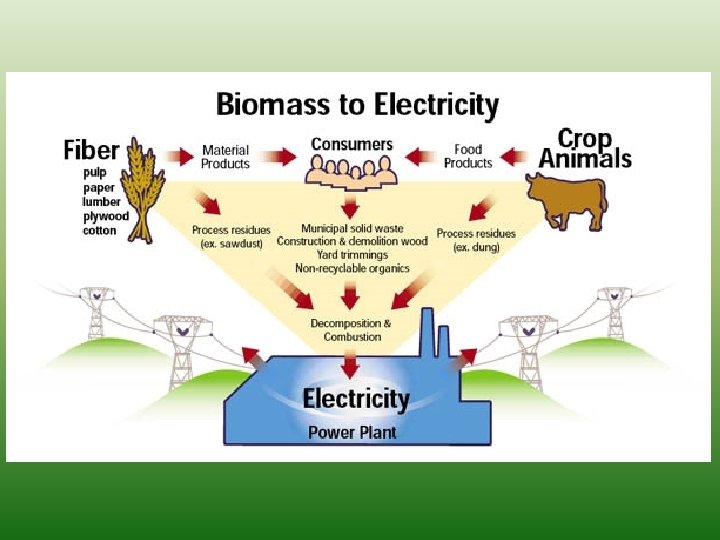
Fuel from biomass waste • When bacteria break down the sugar found in biomass waste (decomposition) in a process called fermentation, they release a gas called methane (CH 4). • This gas can then be collected and transported to a site where it can be burned to generate electricity Biomass Energy Production

Acid Precipitation • Since fossil fuels come from organisms, they contain the same compounds needed for life. • However when these compounds are released from the burning of fossil fuels, they can form undesirable substances • Nitrogen Oxides (NOx) and Sulfur Dioxide (SO 2) are released and combine with Water (H 2 O) in the atmosphere form acids (Nitric Acid - HNO 3 & Sulfuric Acid – H 2 SO 4) • These acids then fall to earth as acid precipitation in the form of rain, sleet or snow
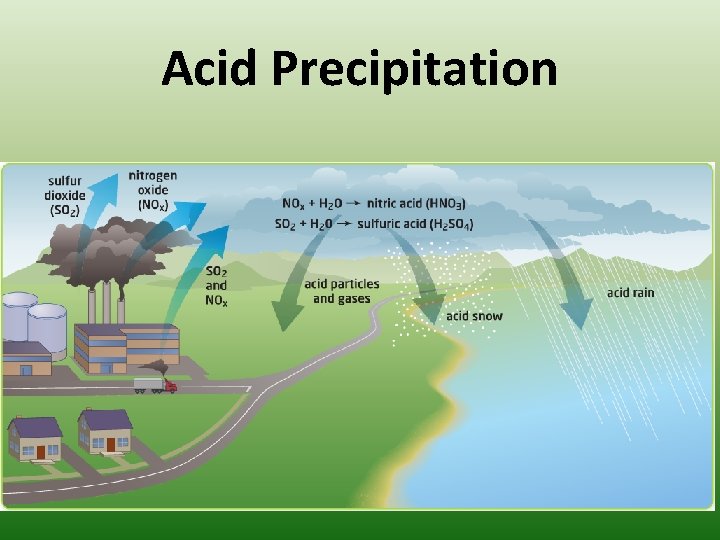
Acid Precipitation

Acid Precipitation

p. H • Is a measure of how acidic a substance is – Acid examples Sulfuric Acid, Citric Acid • It is measured on a scale (p. H scale) from 0 -14 7 (Neutral) = Water and Blood 3 (Acidic) = Vinegar 12 (Basic) = Ammonia (household cleaner)

Effects of Acid Precipitation • Forest soils lose valuable nutrients killing plant life • Can cause numerous problems in aquatic ecosystems - killing fish, amphibians and other organisms due to a decrease in p. H • Destroy stone work

Reducing Acid Precipitation We can reduce acid rain by: • Reducing the amount of fossil fuels used • Improving technologies to prevent harmful gases from escaping • Improving standards for factories and motor-vehicles emissions
- Slides: 14