Biomarkers in Prostate Cancer Symposium The Prostate Net

Biomarkers in Prostate Cancer Symposium The Prostate Net September 17, 2011 Steven Lucas Wayne State University School of Medicine Karmanos Cancer Institute

Why are Biomarkers Important • Prostate Cancer diagnosed in 200, 000 men annually in the united states • 30, 000 cancer specific deaths per year • Yet, a substantial portion of cancers diagnosed through PSA screening do not progress to clinically significant disease.

Problem with PSA screening • It is not specific, resulting in a negative biopsy rate of up to 70% in some series • European randomized trial: 1410 men screened and 48 treated to prevent 1 death from prostate cancer • Sweedish Randomized trial of watchful waiting: – Relative risk for surgery: 0. 62 (0. 44 -0. 87) – NNT 15 overall and 7 for men younger than 65 y

How do we improve screening and treatment decisions? • Clinical nomograms – Include Gleason Score, positive cores, percent involvement of cores, and PSA – Other risk factors: family history, age, race • Biomarkers – Supplement known clinical information
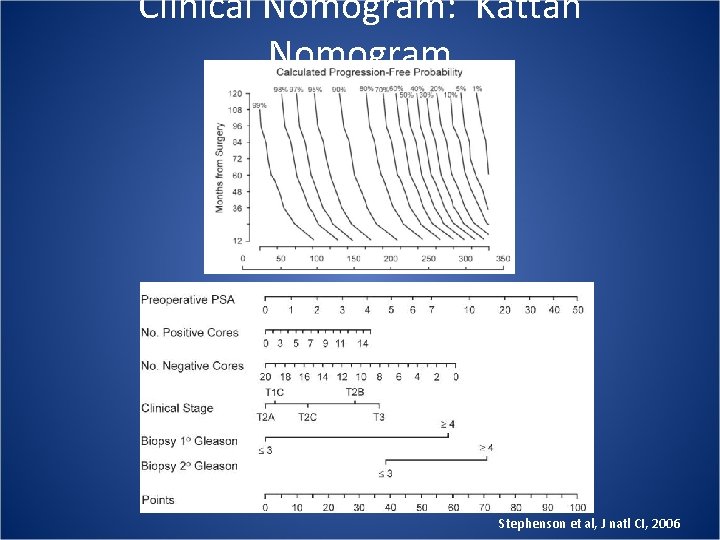
Clinical Nomogram: Kattan Nomogram Stephenson et al, J natl CI, 2006
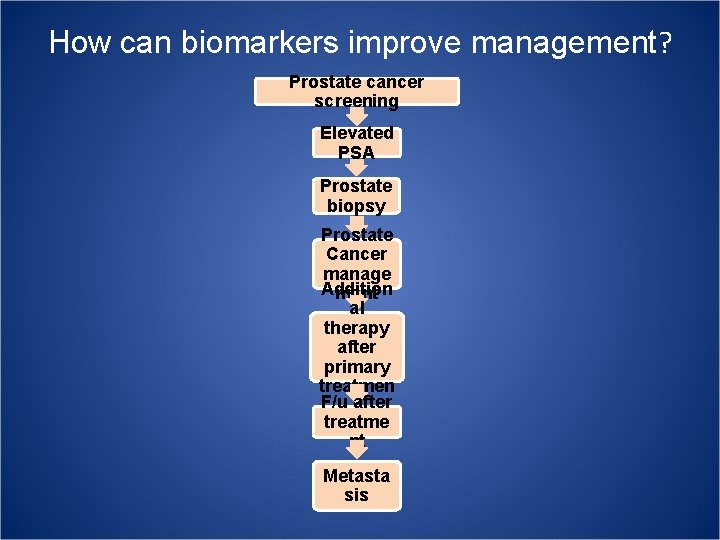
How can biomarkers improve management? Prostate cancer screening Elevated PSA Prostate biopsy Prostate Cancer manage Addition ment al therapy after primary treatmen F/u after t treatme nt Metasta sis

Categories of biomarkers Urine Based Blood Based Tissu Based
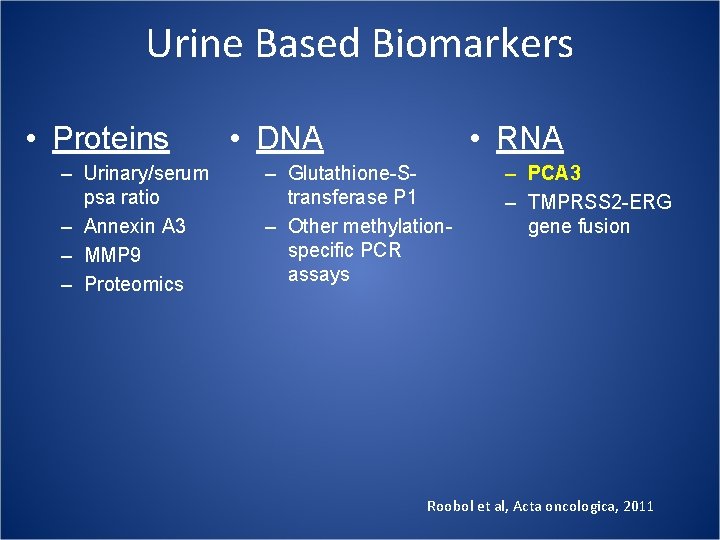
Urine Based Biomarkers • Proteins – Urinary/serum psa ratio – Annexin A 3 – MMP 9 – Proteomics • DNA • RNA – Glutathione-Stransferase P 1 – Other methylationspecific PCR assays – PCA 3 – TMPRSS 2 -ERG gene fusion Roobol et al, Acta oncologica, 2011

Urinary PCA 3 • Developed from differential expression of noncoding RNA’s in prostate cancer versus other prostate conditions • Commercially available, approved diagnostic test • Collected from urine sample following a firm DRE • Could function as a first line screen or prognostic indicator

Urinary PCA 3: First line screen • Several studies show superior overall specificity to PSA: 80 -90%, but include only patients with elevated PSA – In the REDUCE trial the placebo test characteristics for PSA were: Se = 0. 518 Sp = 0. 629 • PCA 3 in patients with PSA 4 -10 ng/ml: – Specificity: 71 -93% Sensitivity: 53 -84% Is PCA 3 Better than PSA for Ca. P diagnosis? • ERSPC: prostate biopsy trigger: PSA ≥ 3 or PCA 3 ≥ 10 • In 721 biopsied, PCA 3 performed only marginally better: AUC: PCA 3=0. 64 PSA=0. 58 Roobol et al, Eur Urol, 2010

PCA 3 and TMPRSS 2 -ERG fusion • Fusion of a strong androgen promoter (transmembrane serine protease) and an oncogene • Further improved diagnostic accuracy (AUC) – PCA 3: 0. 65 PCA 3 + Fusion: 0. 77 – PCA 3 + gene-fusion + PSA: 0. 80 Aubin et al, J Urol, 2008

PCA 3: prognostic indicator • Conflicting studies show a positive relationship 1 with cancer aggressiveness or no relationship 2 • Reduce Trial- chemoprevention of Ca. P with Dutasteride 3: – Weak association of PCA 3 with Gl 7 or higher cancer – OR: 1. 017 (CI 95%: 1. 01 -1. 03) – Though low numbers of high grade prostate cancer weakens the analysis 1. Hessels et al, Prostate, 2010 2. Whitman et al, J Urol, 2008 3. Aubin et al, Urology, 2011

Blood Based biomarkers • Diagnosis – – – PSA velocity Free PSA Pro-PSA BPH-associated PSA • Prognosis – Human Kallikrein 2 – Urokinase plasminogen activator – Transforming Growth factor β 1 – Interleukin-6 – Endoglin
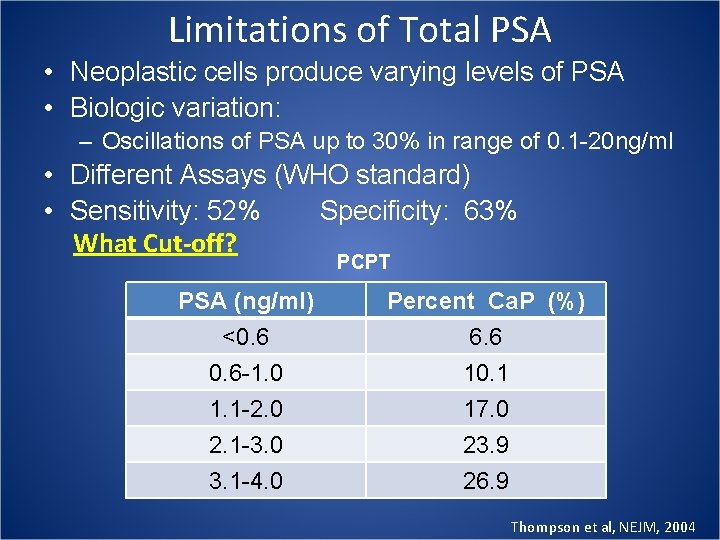
Limitations of Total PSA • Neoplastic cells produce varying levels of PSA • Biologic variation: – Oscillations of PSA up to 30% in range of 0. 1 -20 ng/ml • Different Assays (WHO standard) • Sensitivity: 52% Specificity: 63% What Cut-off? PCPT PSA (ng/ml) <0. 6 -1. 0 1. 1 -2. 0 Percent Ca. P (%) 6. 6 10. 1 17. 0 2. 1 -3. 0 3. 1 -4. 0 23. 9 26. 9 Thompson et al, NEJM, 2004
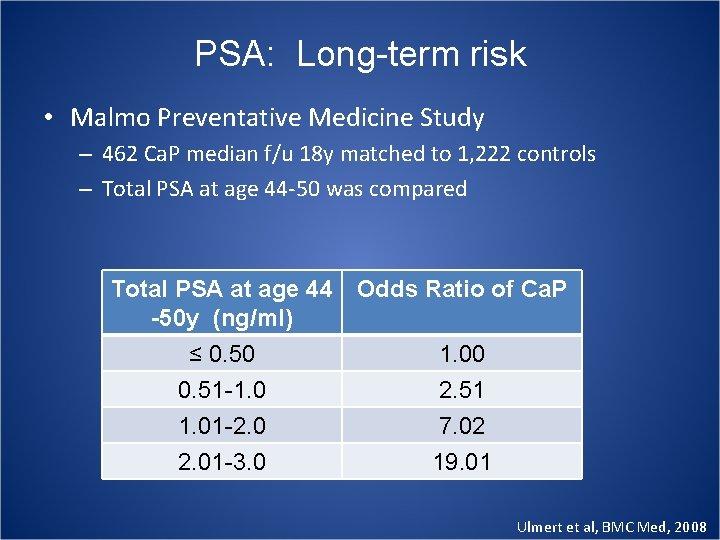
PSA: Long-term risk • Malmo Preventative Medicine Study – 462 Ca. P median f/u 18 y matched to 1, 222 controls – Total PSA at age 44 -50 was compared Total PSA at age 44 Odds Ratio of Ca. P -50 y (ng/ml) ≤ 0. 50 0. 51 -1. 01 -2. 01 -3. 0 1. 00 2. 51 7. 02 19. 01 Ulmert et al, BMC Med, 2008

PSA Velocity Diagnosis • Measurement of change in total PSA over time • Two large prospective trials found no independent predictive value beyond total PSA and other standard variables (PCPT and ERSPC) Prognosis • Increase risk of death determined at PSAv levels greater than 0. 35 – 2. 0 ng/ml per year • May not predict early progression but an indication of aggressive disease beyond treatment window 1. Shariat et al, Acta Onc, 2011 2. D’amico et al, NEJM, 2004 3. Carter et al, J NCI, 2006

Percent Free PSA • Isoform of PSA that remains unbound in plasma • Percent free PSA relative to the total PSA is FDA approved as an adjunct to total PSA between 410 ng/ml – f. PSA < 25% used as a trigger for biopsy • Multicenter, prospective trial – Specificity: 95%, Sensitivity: 20% over PSA – AUC: %f. PSA = 0. 72 PSA: 0. 53 – When use 10 -12 core biopsy: efficiency decreases 1. Catalona et al, JAMA, 1998 2. Canto et al, J Urol, 2004
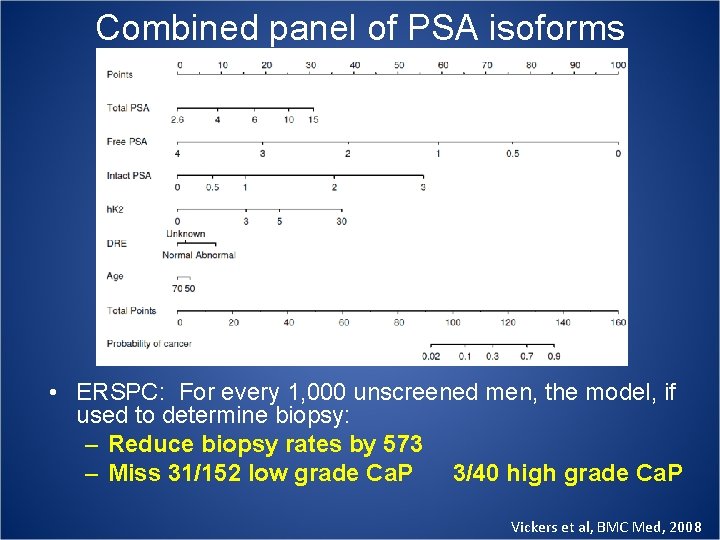
Combined panel of PSA isoforms • ERSPC: For every 1, 000 unscreened men, the model, if used to determine biopsy: – Reduce biopsy rates by 573 – Miss 31/152 low grade Ca. P 3/40 high grade Ca. P Vickers et al, BMC Med, 2008
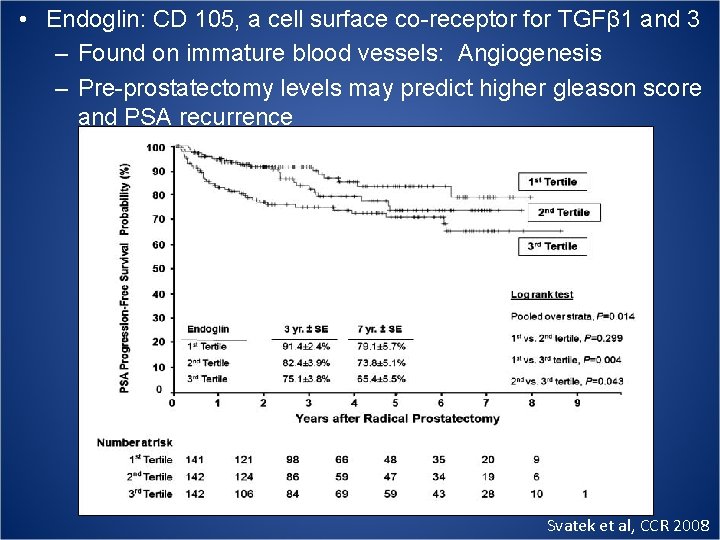
• Endoglin: CD 105, a cell surface co-receptor for TGFβ 1 and 3 – Found on immature blood vessels: Angiogenesis – Pre-prostatectomy levels may predict higher gleason score and PSA recurrence Svatek et al, CCR 2008

Combining Panel of Markers and Nomogram • Biomarkers used to supplement not replace clinical data to improve accuracy of prognosis • Kattan nomogram + biomarker panel: – TGF-β 1, IL-6 R, IL-6, endoglin, VEGF, VCAM-1 • Predictive accuracy of the Kattan nomogram improved by 15% – 71. 6% versus 86. 6% Shariat et al, Acta Onc, 2011

Tissue Based Biomarkers • Diagnostic – High molecular weight cytokeratin – p 63 – AMACR • Prognostic – Human kallikrein type 2 – Prostate specific membrane antigen – Ki-67 – Androgen receptor – Gene fusions – PTEN – P 53 – SPINK 1/TATI – MSMB – EZH 2 – Heat shock proteins – DNA methylation – HER 2
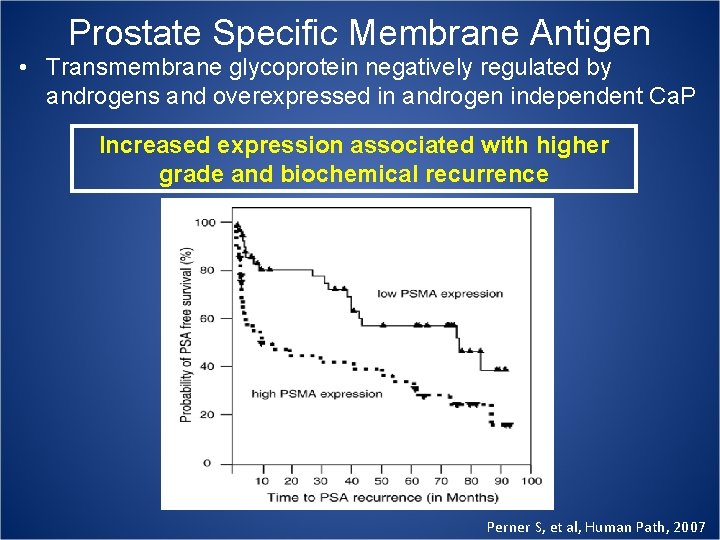
Prostate Specific Membrane Antigen • Transmembrane glycoprotein negatively regulated by androgens and overexpressed in androgen independent Ca. P Increased expression associated with higher grade and biochemical recurrence Perner S, et al, Human Path, 2007

Translating biomarkers into therapeutic Targets • PSMA – PSMA- antibody drug conjugate currently in phase 1 trial – Castration-resistent metastatic Ca. P • Endoglin – TRC 105 is a human/murine chimeric monoclonal antibody that binds to endoglin, thus inhibiting angiogenesis – Phase 1 / phase 2 trial for CRPC www. nih. gov, 2011

Summary • Biomarkers serve as a powerful adjunct to the diagnosis and management of prostate cancer • Biomarkers are testable in the urine, blood, and prostate cancer tissue • Further validation of these biomarkers and research into potential therapeutic targets is needed
- Slides: 24