Biomarkers and personalized Therapy for CRC T Thomaidis

Biomarkers and personalized Therapy for CRC T. Thomaidis
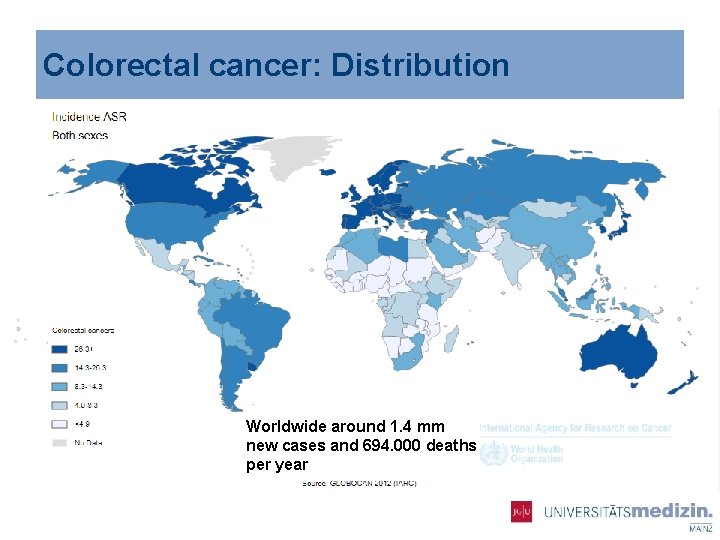
Colorectal cancer: Distribution Worldwide around 1. 4 mm new cases and 694. 000 deaths per year
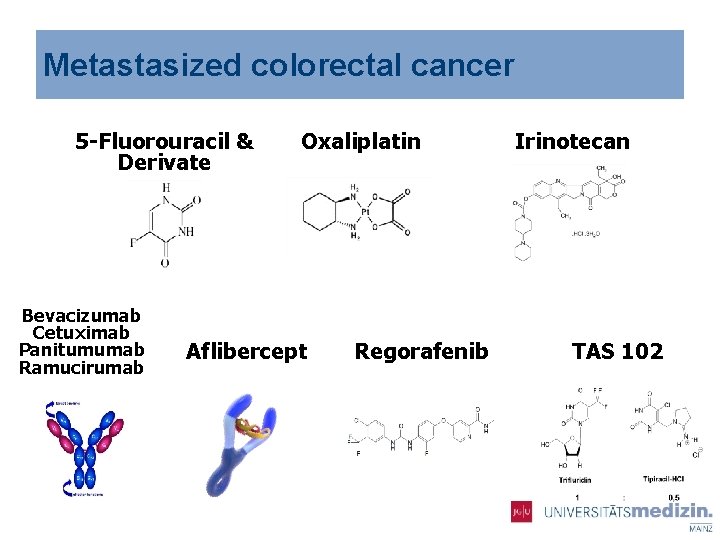
Metastasized colorectal cancer 5 -Fluorouracil & Derivate Bevacizumab Cetuximab Panitumumab Ramucirumab Oxaliplatin Aflibercept Regorafenib Irinotecan TAS 102
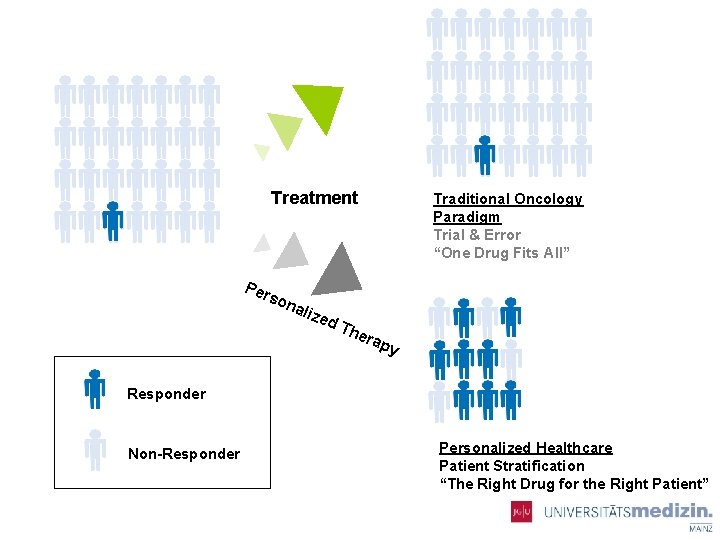
Treatment Per s ona lize Traditional Oncology Paradigm Trial & Error “One Drug Fits All” d. T her ap y Responder Non-Responder Personalized Healthcare Patient Stratification “The Right Drug for the Right Patient”

Biomarkers § Prognostic Biomarkers: Parameters that show the progression of the disease regardless of the type of treatment. § Predictive Biomarkers: Parameter that assess the most likely response to a particular treatment type
Biomarkers Normal Tumour cell
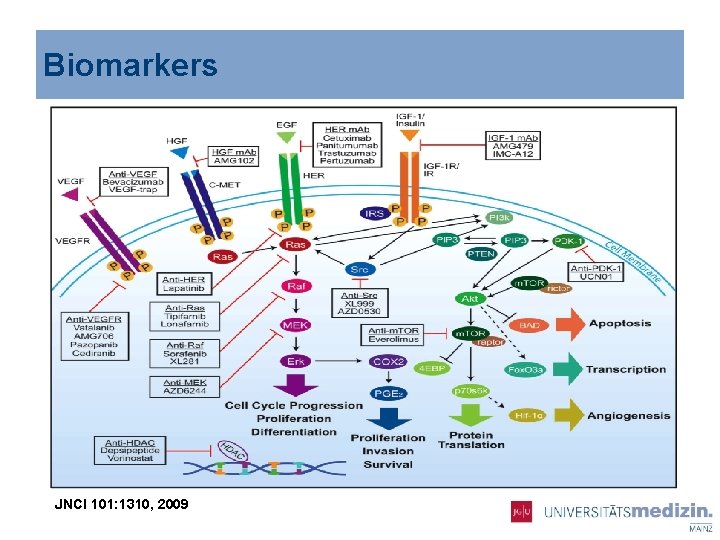
Biomarkers JNCI 101: 1310, 2009
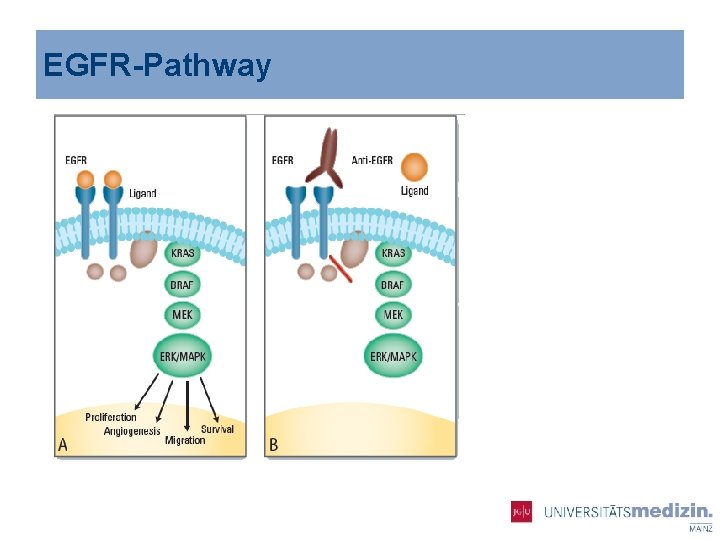
EGFR-Pathway
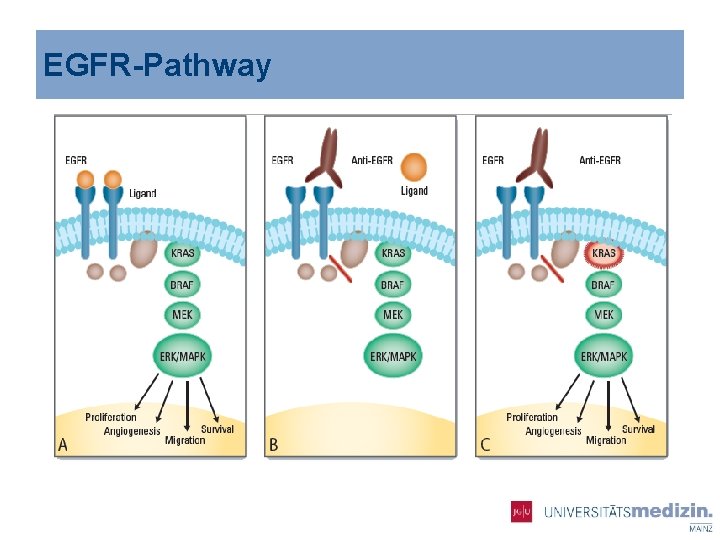
EGFR-Pathway
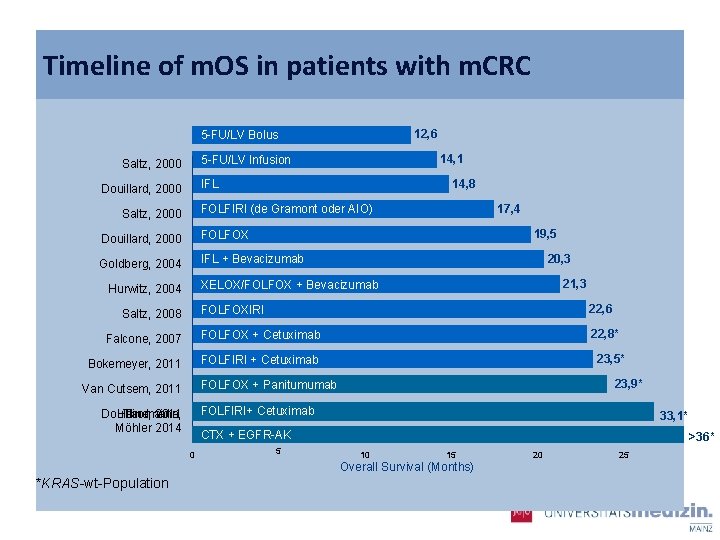
Timeline of m. OS in patients with m. CRC 12, 6 5 -FU/LV Bolus 14, 1 5 -FU/LV Infusion Saltz, 2000 14, 8 IFL Douillard, 2000 17, 4 FOLFIRI (de Gramont oder AIO) Saltz, 2000 19, 5 Douillard, 2000 FOLFOX Goldberg, 2004 IFL + Bevacizumab 20, 3 21, 3 XELOX/FOLFOX + Bevacizumab Hurwitz, 2004 FOLFOXIRI 22, 6 Falcone, 2007 FOLFOX + Cetuximab 22, 8* Bokemeyer, 2011 FOLFIRI + Cetuximab Saltz, 2008 23, 5* 23, 9* FOLFOX + Panitumumab Van Cutsem, 2011 FOLFIRI+ Cetuximab Douillard, Heinemann, Thomaidis, 2011 Möhler 2014 33, 1* CTX + EGFR-AK 0 5 >36* 10 15 Overall Survival (Months) *KRAS-wt-Population 20 25

Mutations in CRC § Why 40 -60 % K-ras wt do not respond to a treatment with EGFR-Ab?
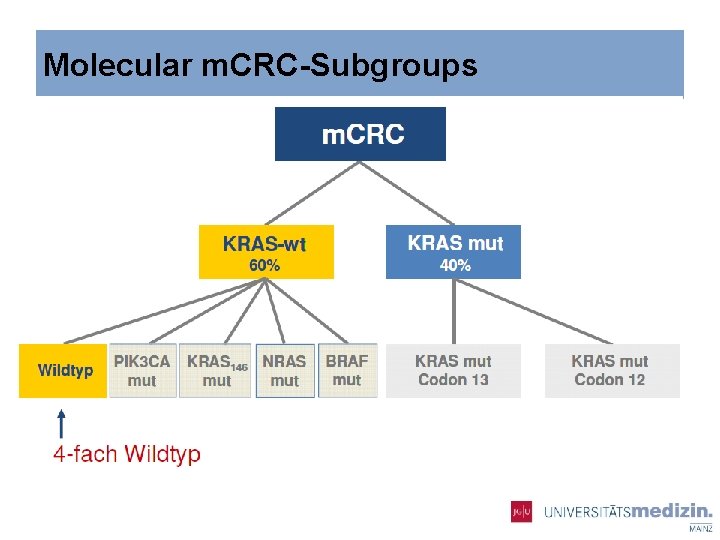
Molecular m. CRC-Subgroups
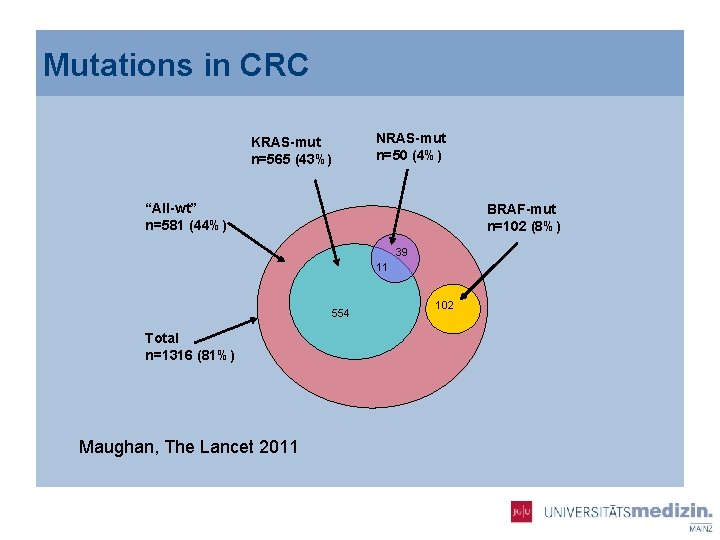
Mutations in CRC KRAS-mut n=565 (43%) NRAS-mut n=50 (4%) “All-wt” n=581 (44%) BRAF-mut n=102 (8%) 39 11 554 Total n=1316 (81%) Maughan, The Lancet 2011 102
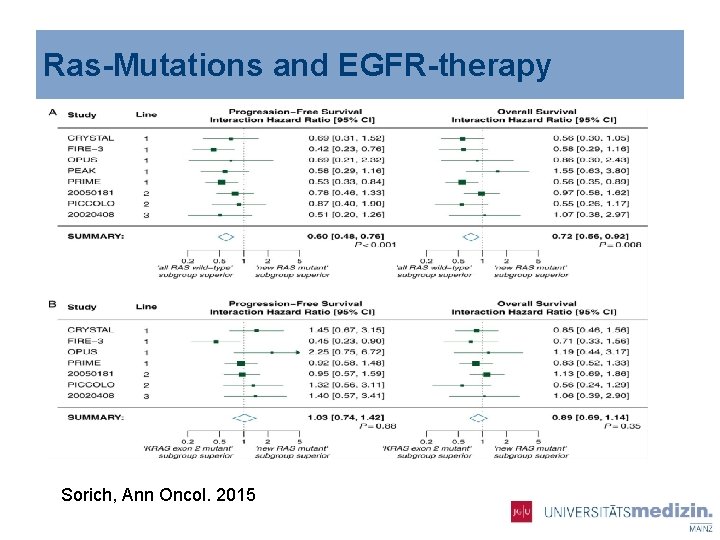
Ras-Mutations and EGFR-therapy Sorich, Ann Oncol. 2015
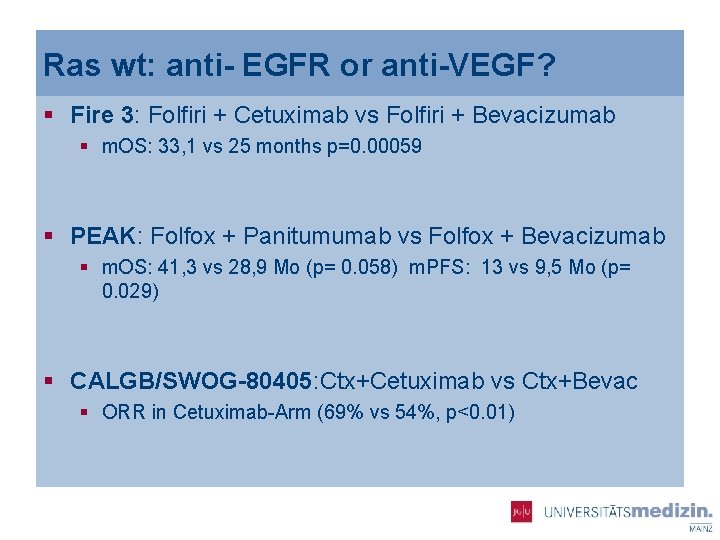
Ras wt: anti- EGFR or anti-VEGF? § Fire 3: Folfiri + Cetuximab vs Folfiri + Bevacizumab § m. OS: 33, 1 vs 25 months p=0. 00059 § PEAK: Folfox + Panitumumab vs Folfox + Bevacizumab § m. OS: 41, 3 vs 28, 9 Mo (p= 0. 058) m. PFS: 13 vs 9, 5 Mo (p= 0. 029) § CALGB/SWOG-80405: Ctx+Cetuximab vs Ctx+Bevac § ORR in Cetuximab-Arm (69% vs 54%, p<0. 01)

BRAF § V 600 E mutations 8 -12 % in patients with m. CRC § Female, older patients § 2/3 right Colon § Mucinous differentiation § High rate on lymphnodes and peritoneal carcinomatosis § Worse prognosis, m. PFS < 6 Mo m. Os< 1 year
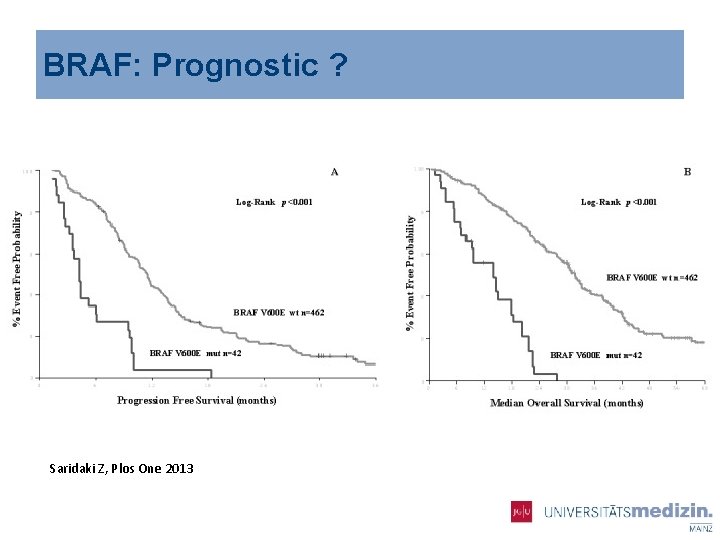
BRAF: Prognostic ? Saridaki Z, Plos One 2013
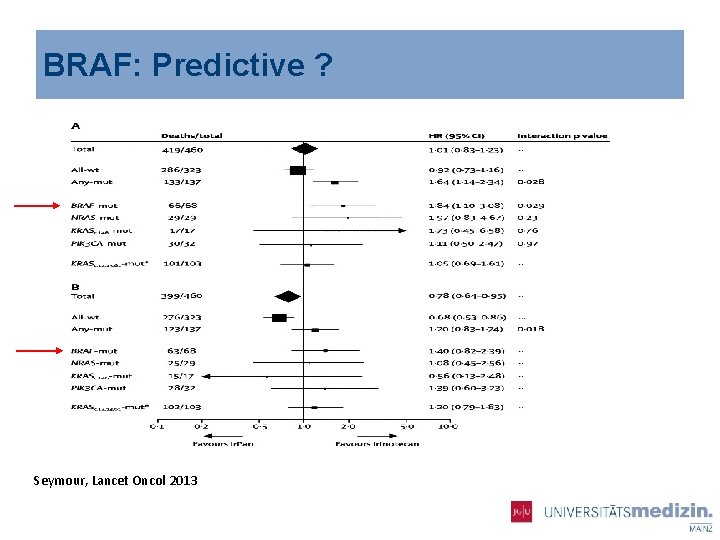
BRAF: Predictive ? Seymour, Lancet Oncol 2013
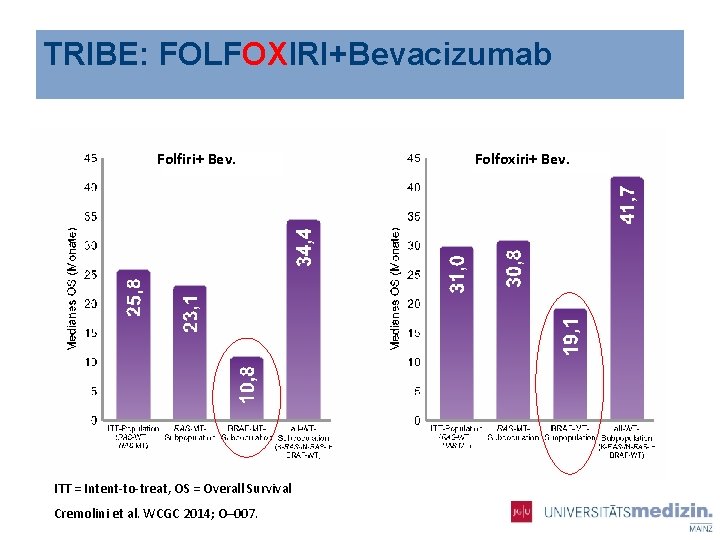
TRIBE: FOLFOXIRI+Bevacizumab Folfiri+ Bev. ITT = Intent-to-treat, OS = Overall Survival Cremolini et al. WCGC 2014; O– 007. Folfoxiri+ Bev.
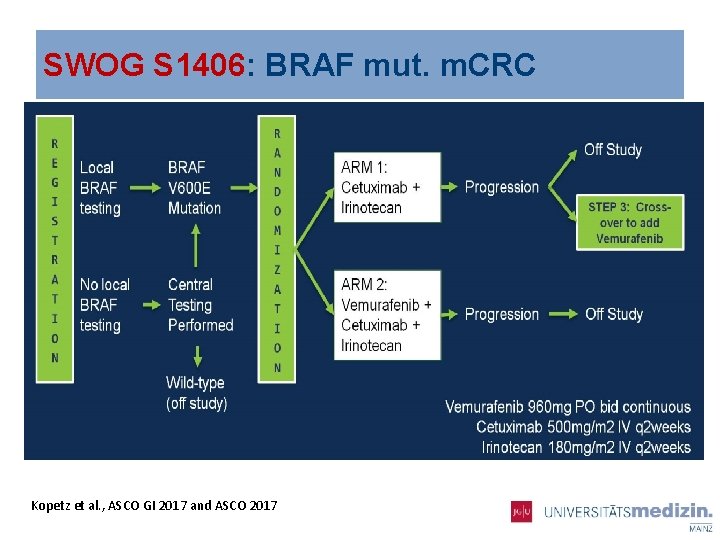
SWOG S 1406: BRAF mut. m. CRC Kopetz et al. , ASCO GI 2017 and ASCO 2017
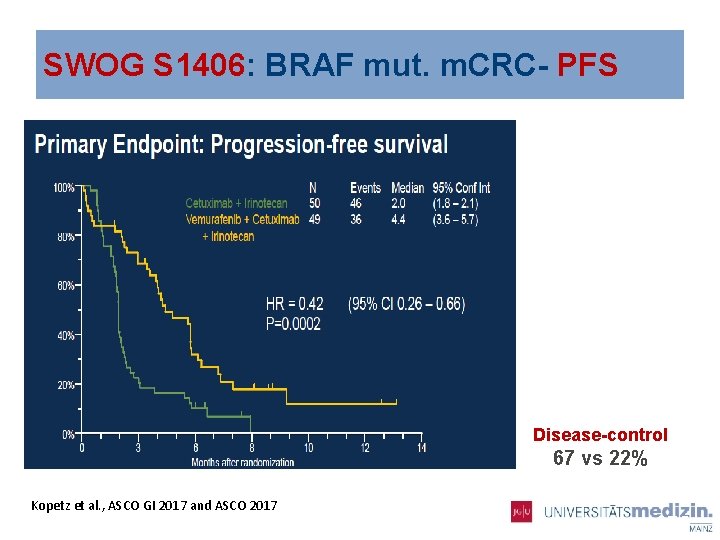
SWOG S 1406: BRAF mut. m. CRC- PFS Disease-control 67 vs 22% Kopetz et al. , ASCO GI 2017 and ASCO 2017 075
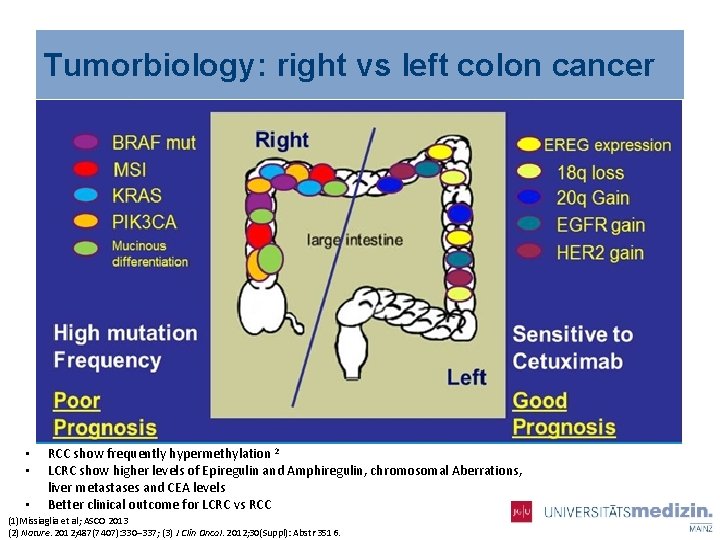
Tumorbiology: right vs left colon cancer • • • RCC show frequently hypermethylation 2 LCRC show higher levels of Epiregulin and Amphiregulin, chromosomal Aberrations, liver metastases and CEA levels Better clinical outcome for LCRC vs RCC (1)Missiaglia et al; ASCO 2013 (2) Nature. 2012; 487(7407): 330– 337; (3) J Clin Oncol. 2012; 30(Suppl): Abstr 3516.
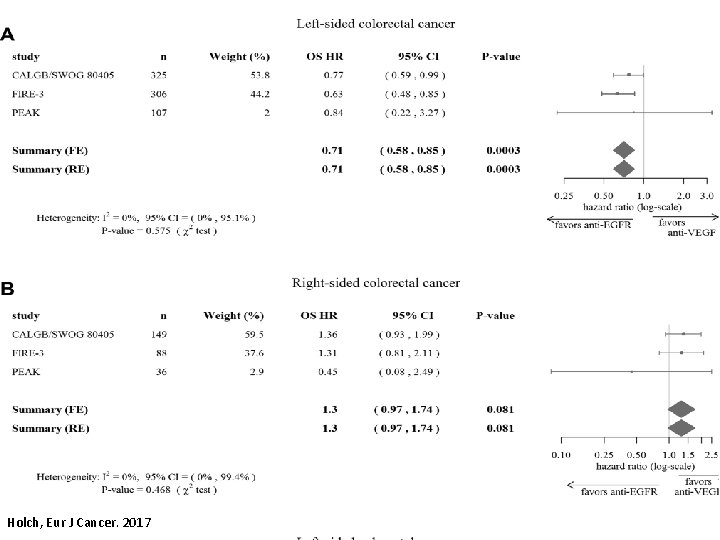
Holch, Eur J Cancer. 2017
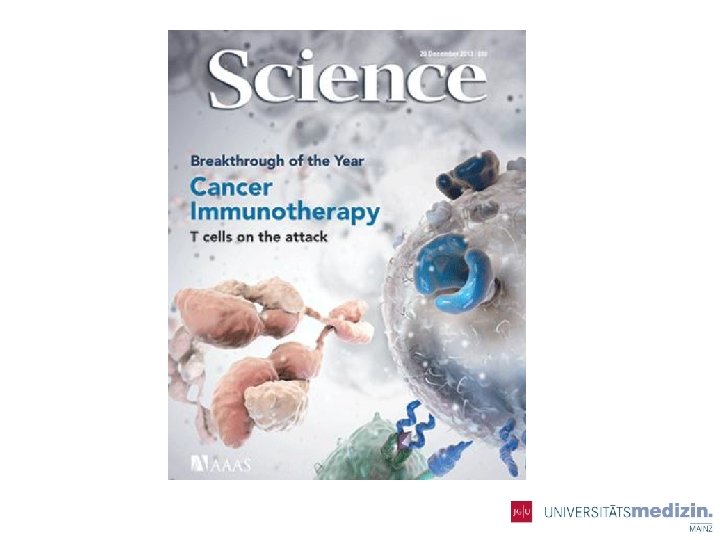
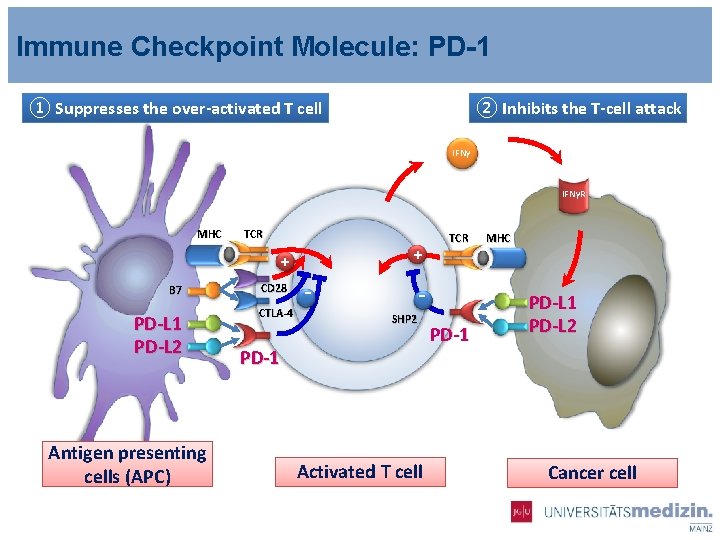
Immune Checkpoint Molecule: PD-1 ① Suppresses the over-activated T cell ② Inhibits the T-cell attack IFNγR MHC TCR + + B 7 PD-L 1 PD-L 2 Antigen presenting cells (APC) CD 28 CTLA-4 - TCR SHP 2 PD-1 Activated T cell PD-1 MHC PD-L 1 PD-L 2 Cancer cell
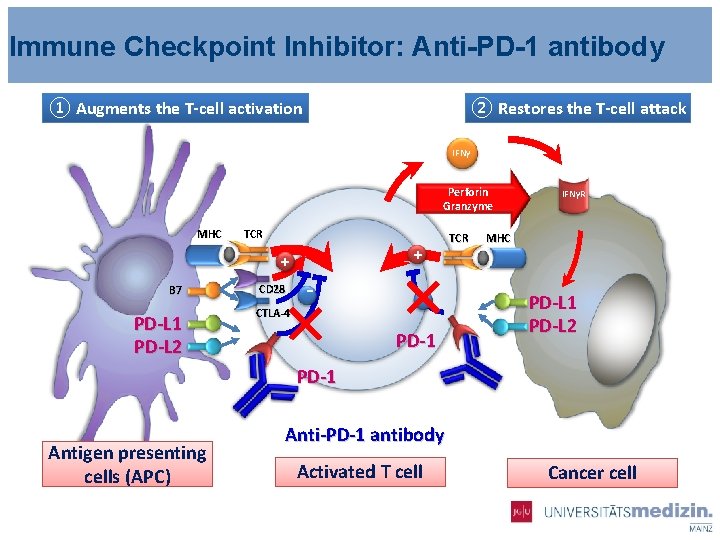
Immune Checkpoint Inhibitor: Anti-PD-1 antibody ① Augments the T-cell activation ② Restores the T-cell attack IFNγ Perforin Granzyme MHC TCR + + B 7 PD-L 1 PD-L 2 CD 28 - CTLA-4 PD-1 TCR IFNγR MHC PD-L 1 PD-L 2 PD-1 Antigen presenting cells (APC) Anti-PD-1 antibody Activated T cell Cancer cell
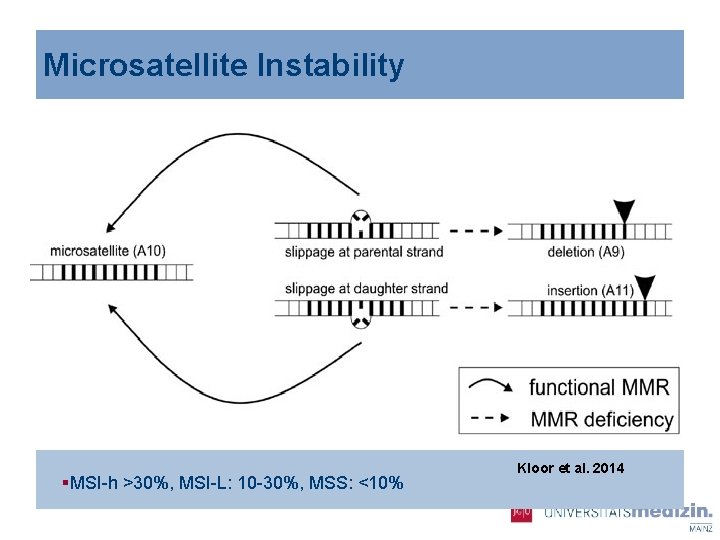
Microsatellite Instability §MSI-h >30%, MSI-L: 10 -30%, MSS: <10% Kloor et al. 2014
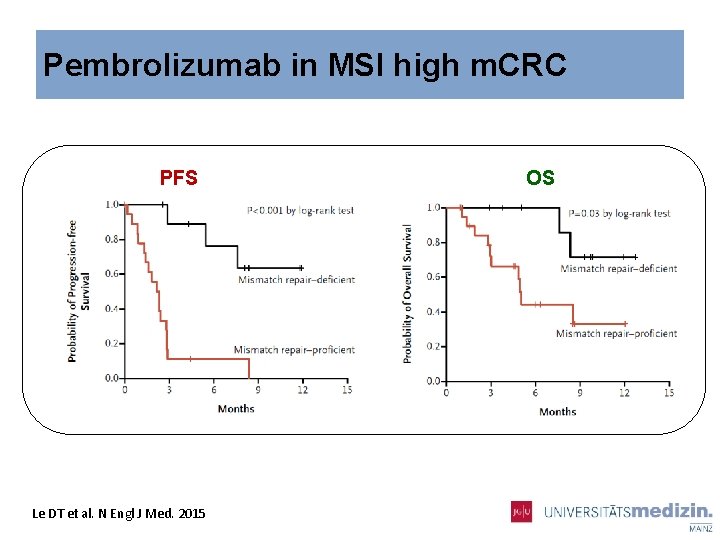
Pembrolizumab in MSI high m. CRC PFS Le DT et al. N Engl J Med. 2015 OS
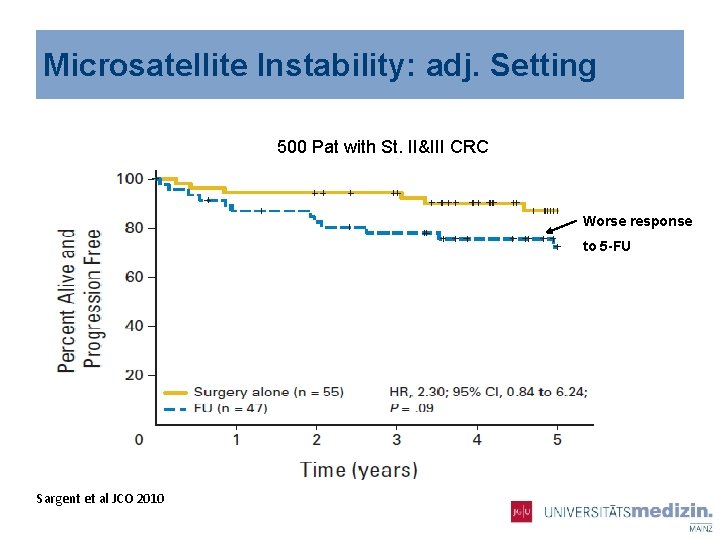
Microsatellite Instability: adj. Setting 500 Pat with St. II&III CRC Worse response to 5 -FU Sargent et al JCO 2010
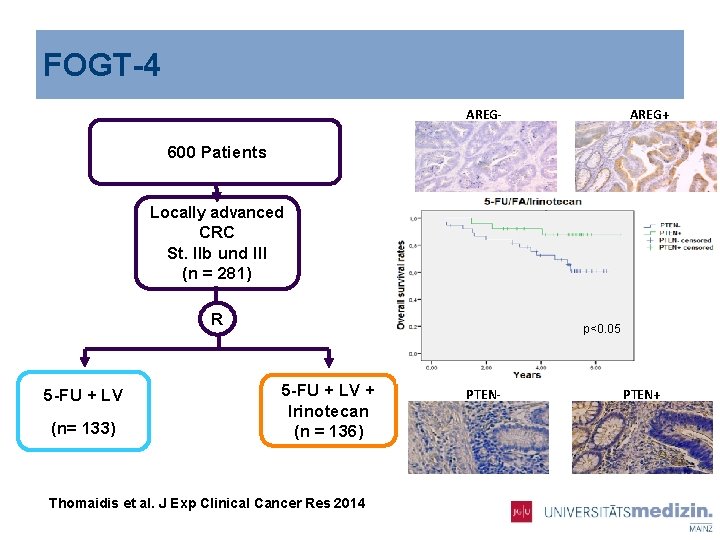
FOGT-4 AREG- AREG+ Hif-1α- Hif-1α+ 600 Patients Locally advanced CRC St. IIb und III (n = 281) VEGFR-3 - R 5 -FU + LV (n= 133) VEGFR-3+ p<0. 05 5 -FU + LV + Irinotecan (n = 136) Thomaidis et al. J Exp Clinical Cancer Res 2014 PTEN- PTEN+

Future molecules § HER 2 neu Trastuzumab+Lapatinib ? § Mutations in PIK 3 CA, exon 20 § Evaluation of PTEN loss by IHC § Evaluation of the levels of the EGFR ligands amphiregulin, epiregulin and TGF-α § EGFR protein expression § EGFR amplification and copy number § EGFR ectodomain mutations § Evaluation of HER 3, and MET receptor overexpression

Personalized Factors Expansion of the tumor Local expansion? Lymph nodes? Metastases? General Condition Comorbidity Patient wish
Interdisciplinary approach
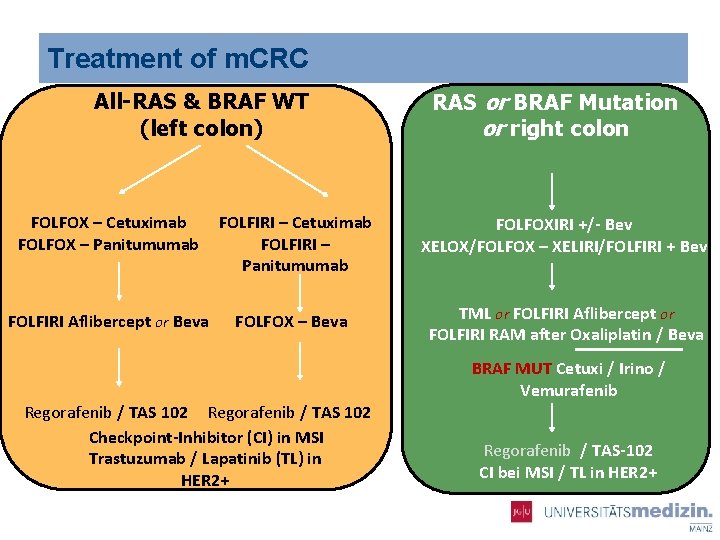
Treatment of m. CRC All-RAS & BRAF WT (left colon) RAS or BRAF Mutation or right colon FOLFOX – Cetuximab FOLFOX – Panitumumab FOLFIRI – Cetuximab FOLFIRI – Panitumumab FOLFOXIRI +/- Bev XELOX/FOLFOX – XELIRI/FOLFIRI + Bev FOLFIRI Aflibercept or Beva FOLFOX – Beva TML or FOLFIRI Aflibercept or FOLFIRI RAM after Oxaliplatin / Beva Regorafenib / TAS 102 Checkpoint-Inhibitor (CI) in MSI Trastuzumab / Lapatinib (TL) in HER 2+ BRAF MUT Cetuxi / Irino / Vemurafenib Regorafenib / TAS-102 CI bei MSI / TL in HER 2+

Thank you for your attention
- Slides: 35