Biology study of life Bio life Make observations
Biology – study of life Bio =life

Make observations and perform studies to better understand the world and make useful predictions about the future.
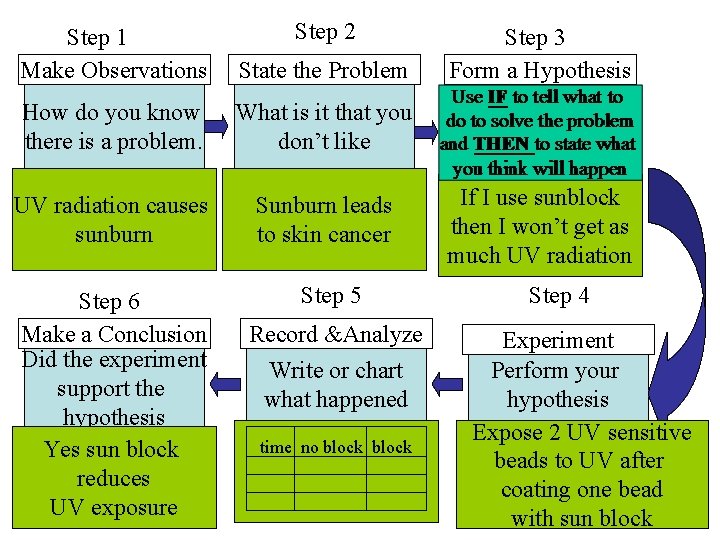
Step 2 Step 1 Make Observations State the Problem How do you know there is a problem. What is it that you don’t like UV radiation causes sunburn Sunburn leads to skin cancer Step 6 Make a Conclusion Did the experiment support the hypothesis Yes sun block reduces UV exposure Step 5 Record &Analyze Write or chart what happened time no block Step 3 Form a Hypothesis If I use sunblock then I won’t get as much UV radiation Step 4 Experiment Perform your hypothesis Expose 2 UV sensitive beads to UV after coating one bead with sun block

Scientific theory vs. common theory Hypothesis vs. Scientific Theory Scientific Law -

When experiments are repeatable by others and additional experiments are used to challenge the finding, but they continue to support the hypothesis, the scientific community elevates the hypothesis to the level of Scientific Theory. After numerous challenges and rigorous experiments testing the Scientific Theory continue to support theory it is elevated to the level of Scientific Law ex. All living things are composed of cells.
Safety Process Precautions tools
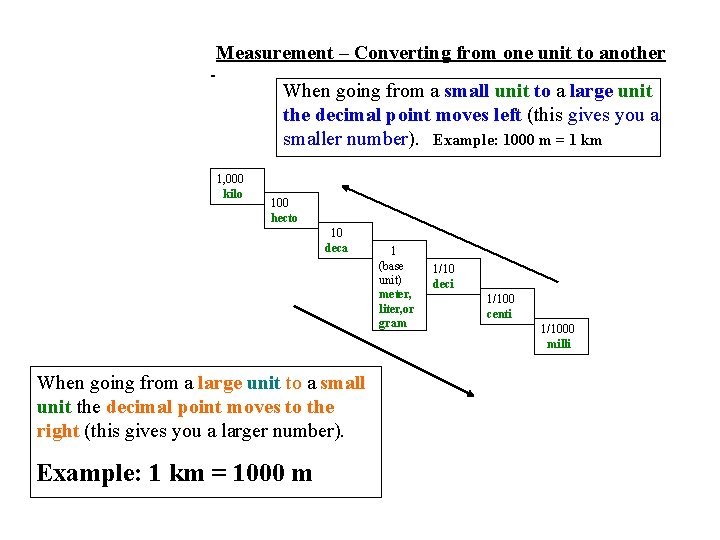
Measurement – Converting from one unit to another When going from a small unit to a large unit the decimal point moves left (this gives you a smaller number). Example: 1000 m = 1 km 1, 000 kilo 100 hecto 10 deca When going from a large unit to a small unit the decimal point moves to the right (this gives you a larger number). Example: 1 km = 1000 m 1 (base unit) meter, liter, or gram 1/10 deci 1/100 centi 1/1000 milli
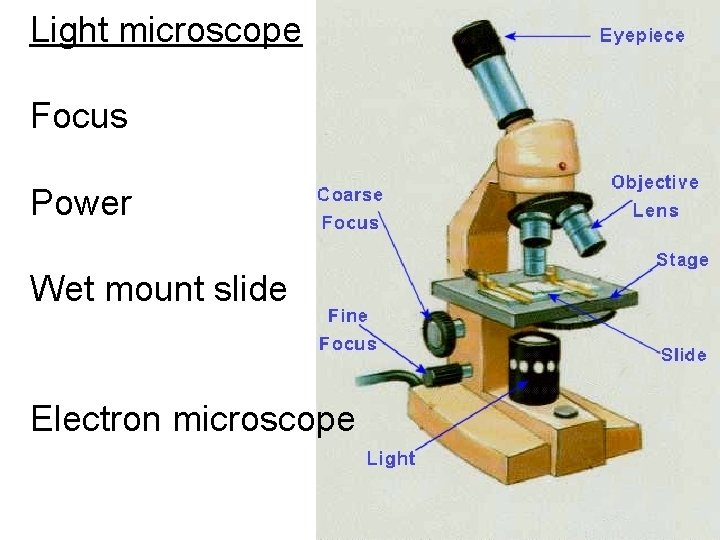
Light microscope Focus Power Wet mount slide Electron microscope

Characteristics of Life • How do we know if something is alive or not? • If the following characteristics are present then the thing is alive.
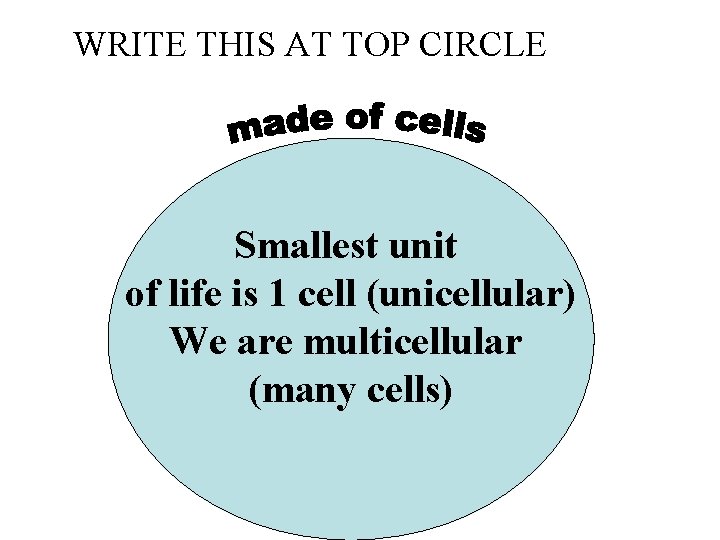
WRITE THIS AT TOP CIRCLE Smallest unit of life is 1 cell (unicellular) We are multicellular (many cells)
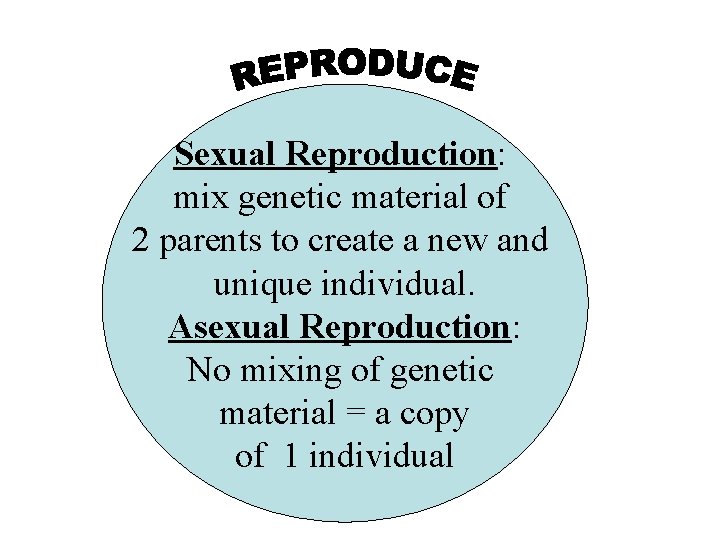
Sexual Reproduction: mix genetic material of 2 parents to create a new and unique individual. Asexual Reproduction: No mixing of genetic material = a copy of 1 individual
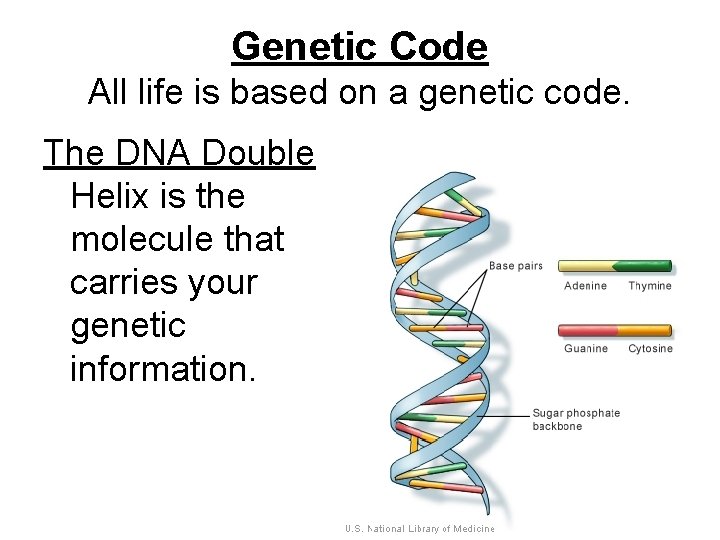
Genetic Code All life is based on a genetic code. The DNA Double Helix is the molecule that carries your genetic information.

Grow = increase in size and Number of cells. Develop = Cells or body’s characteristics change with Maturity.

Nutrients and energy are used To grow and provide energy for movement and cell activities. Metabolism is how fast your body breaks down food and releases the energy to be used. Materials can be used for shelter or attracting a mate

Responds to the Environment What changes in the environment do organisms respond to? Temperature Amount of sunlight Humidity Soil Moisture Predators Potential Mates

Maintain Internal Conditions (homeostasis) During activities we use up energy, increase our temperature, as well as lose water and salts. Our enzymes only work in a limited range. sweating Breathing Drink Eat

Change over time (Evolution) Over time species change. All dogs that are alive today had an ancestor who was a wolf.

As the environment changes those that survive and have successful offspring will shape what the future generations look like. (Offspring are similar to parents)

Biochemistry The study of the organic molecules of life.
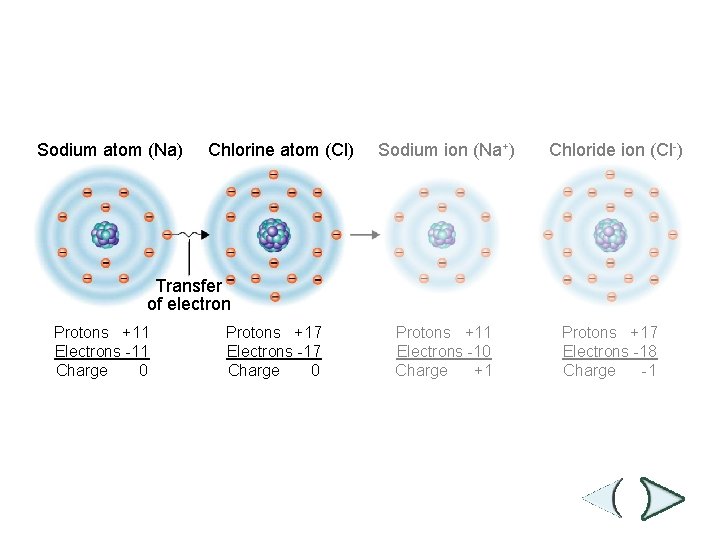
Figure 2 -3 Ionic Bonding Section 2 -1 Sodium atom (Na) Chlorine atom (Cl) Sodium ion (Na+) Chloride ion (Cl-) Transfer of electron Protons +11 Electrons -11 Charge 0 Protons +17 Electrons -17 Charge 0 Protons +11 Electrons -10 Charge +1 Protons +17 Electrons -18 Charge -1

Carbon is made up of Carbon atoms. Atoms of carbon make up the element Carbon. (pure) Compounds are made When 2 or more elements Are combined in a specific ratio. (mixed)

2 Types of bonds between elements
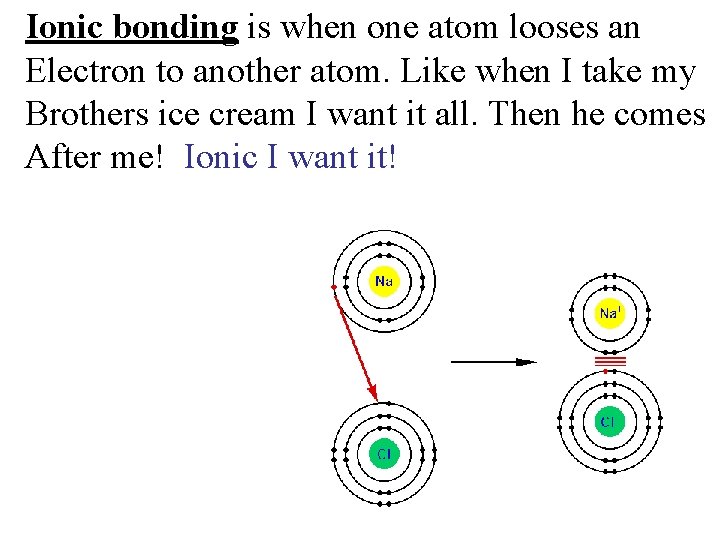
Ionic bonding is when one atom looses an Electron to another atom. Like when I take my Brothers ice cream I want it all. Then he comes After me! Ionic I want it!

Covalent bonds share electrons. This is a single covalent bond sharing 2 electrons. Double covalent bonds share 4 electrons. Covalent bonds like Co-workers share items.
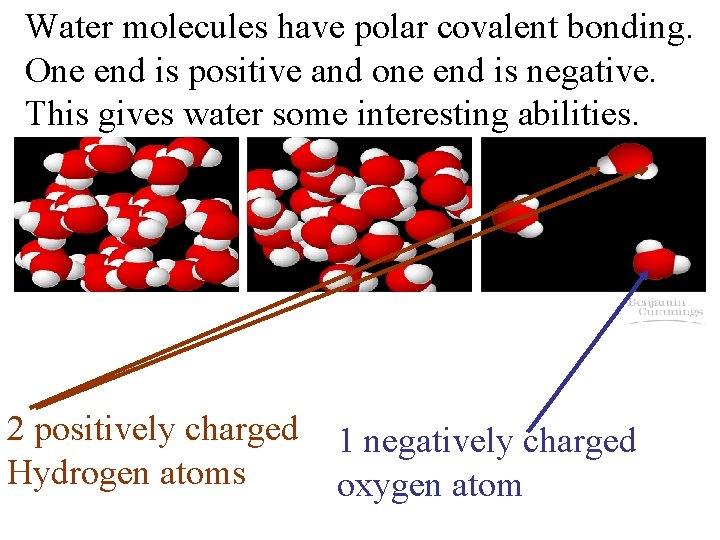
Water molecules have polar covalent bonding. One end is positive and one end is negative. This gives water some interesting abilities. 2 positively charged 1 negatively charged Hydrogen atoms oxygen atom
4 macromolecules or nutrient groups

Carbohydrates Good or Bad ? ? ? ? You get to answer this question at the end!
PROTEINS
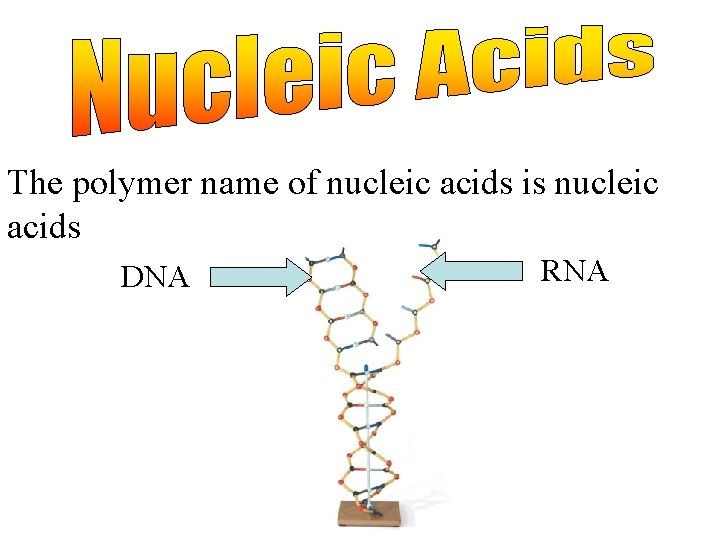
The polymer name of nucleic acids is nucleic acids DNA RNA
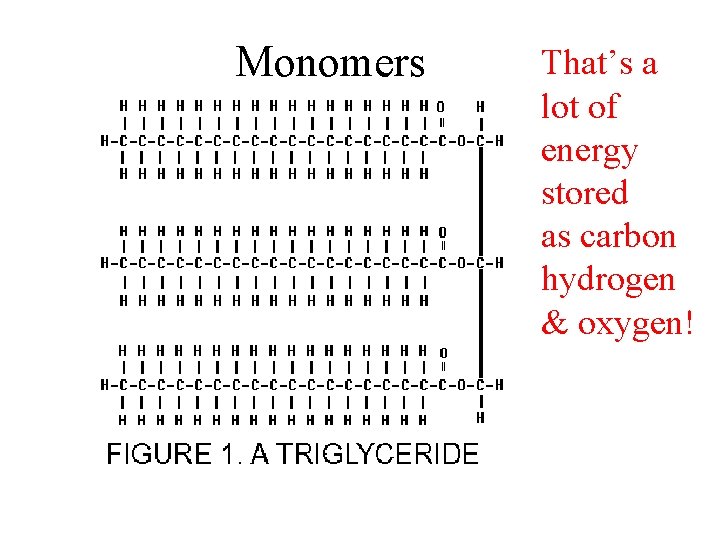
Monomers • That’s a lot of energy stored as carbon hydrogen & oxygen!

What is meant by the following; It’s the same for computers and bodies… Garbage in garbage out.
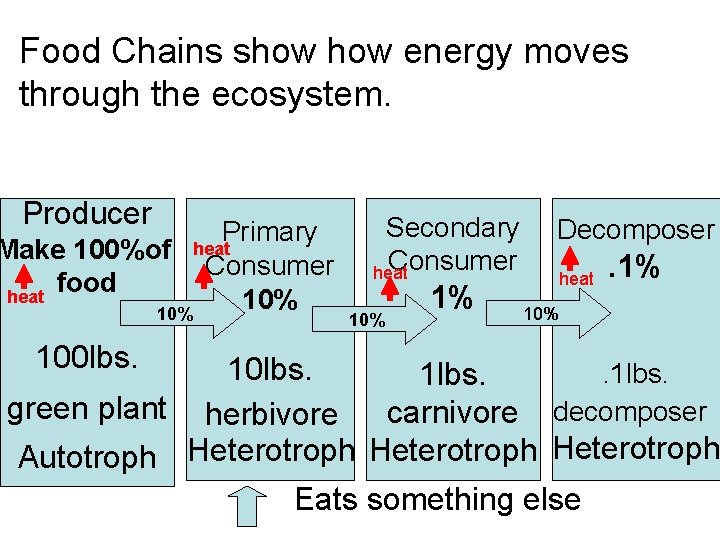
Food Chains show energy moves through the ecosystem. Producer Primary heat Make 100%of Consumer food heat 10% 100 lbs. Secondary Consumer heat 10% 1% Decomposer heat . 1% 10 lbs. . 1 lbs. green plant herbivore carnivore decomposer Autotroph Heterotroph Eats something else
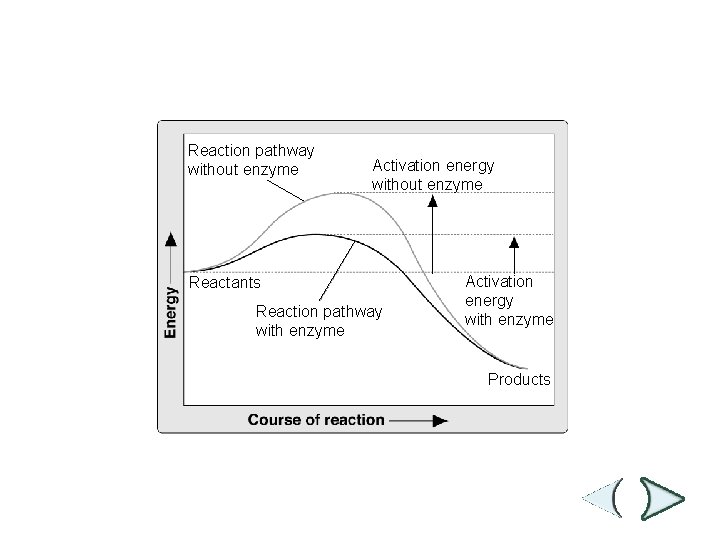
Effect of Enzymes Section 2 -4 Reaction pathway without enzyme Activation energy without enzyme Reactants Reaction pathway with enzyme Activation energy with enzyme Products
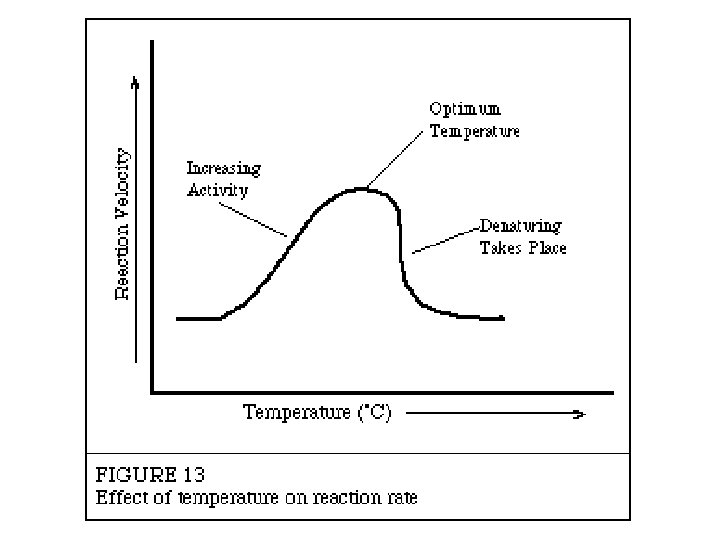
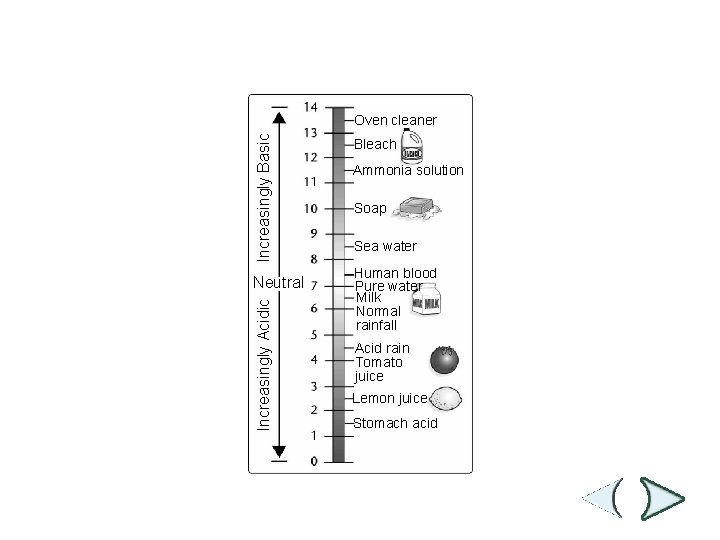
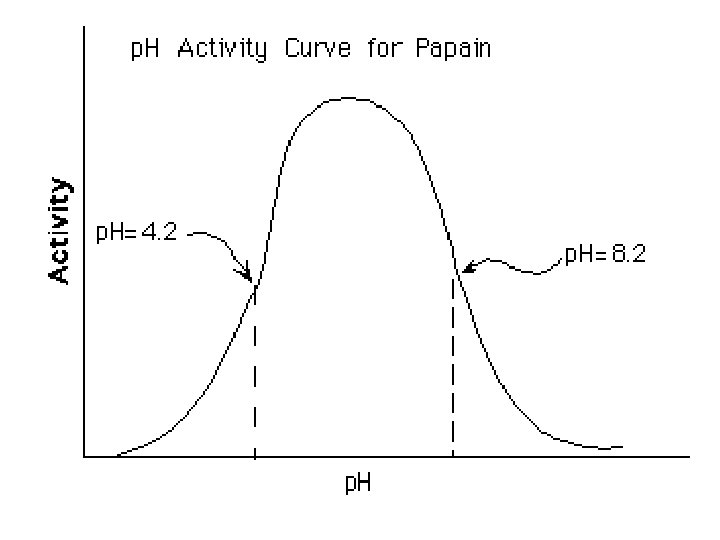
p. H Scale Section 2 -2 Increasingly Basic Oven cleaner Increasingly Acidic Neutral Bleach Ammonia solution Soap Sea water Human blood Pure water Milk Normal rainfall Acid rain Tomato juice Lemon juice Stomach acid
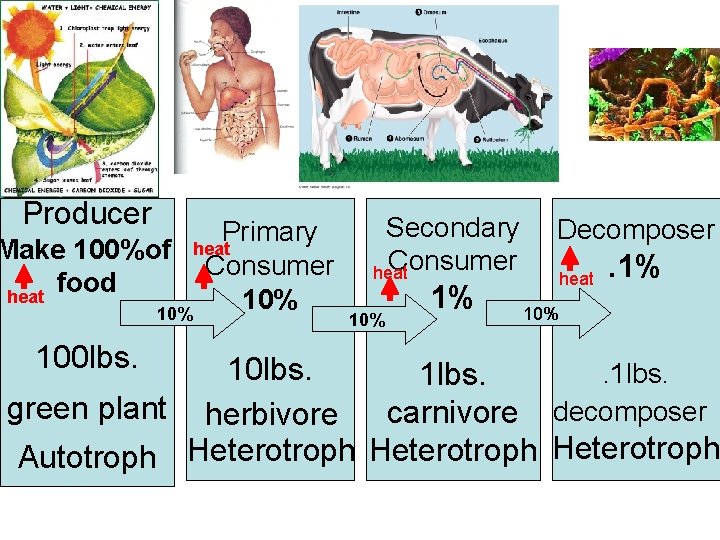
Producer Primary heat Make 100%of Consumer food heat 10% 100 lbs. Secondary Consumer heat 10% 1% Decomposer heat . 1% 10 lbs. . 1 lbs. green plant herbivore carnivore decomposer Autotroph Heterotroph
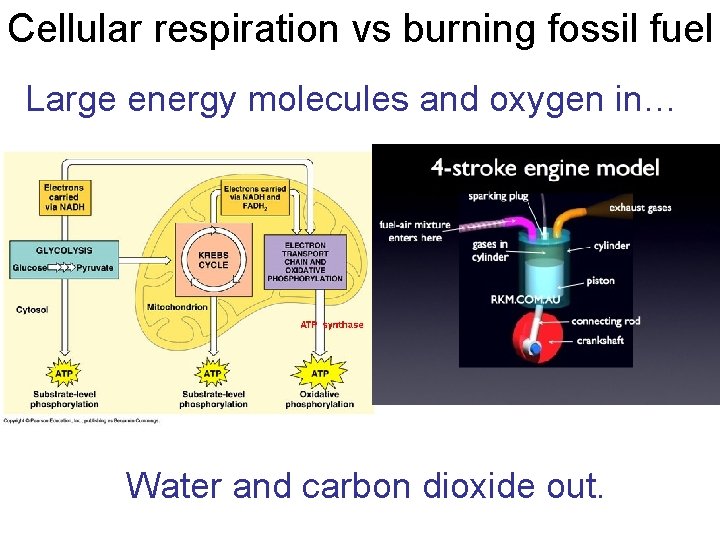
Cellular respiration vs burning fossil fuel Large energy molecules and oxygen in… Water and carbon dioxide out.

Defend the following statement. If plants don’t get enough nitrogen they will have difficulty making new cells.

CHO CHONP & sometimes S
- Slides: 40