Biology Review L 18 1 Macromolecules Enzymes Describe

Biology Review L. 18. 1 Macromolecules, Enzymes • Describe the basic molecular structures and primary functions of the four major categories of biological macromolecules • Identify and/or describe their basic molecular structure and function in organisms.
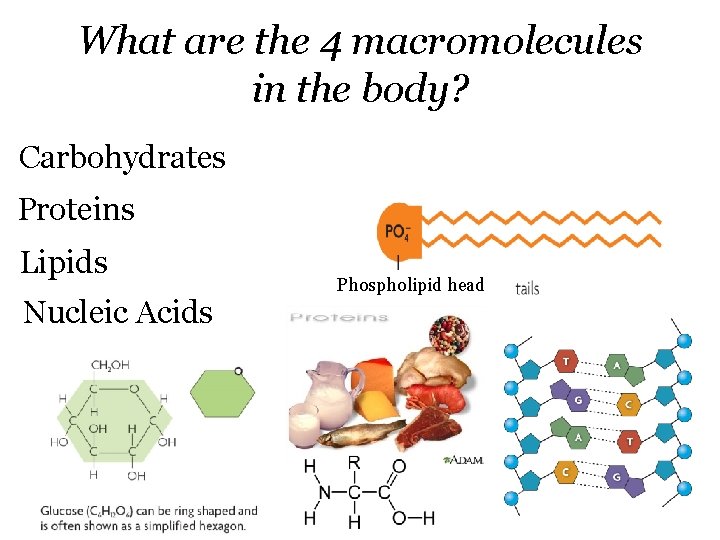
What are the 4 macromolecules in the body? Carbohydrates Proteins Lipids Nucleic Acids Phospholipid head

How do each of the four major categories of macromolecules support the body? • Carbohydrates- ▫ Main source of short term energy • Proteins ▫ ▫ Control chemical reactions (enzymes) Bone/muscle formation Move substances in/out of cells Antibodies • Lipids▫ ▫ Long term energy storage Cushions organs Make up cell membrane Make hormones • Nucleic Acids- ▫ DNA and RNA

• Describe the properties of the carbon atom that make the diversity of carbon compounds possible • Explain how enzymes speed up the rate of a biochemical reaction by lowering the energy needed to start reaction. • Identify and/or describe the effect of environmental factors on enzyme activity. • Explain the difference between organic and inorganic compounds.
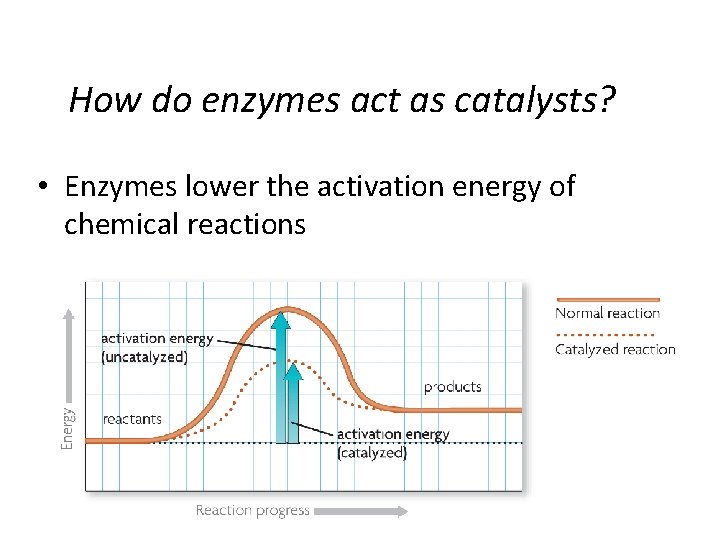
How do enzymes act as catalysts? • Enzymes lower the activation energy of chemical reactions
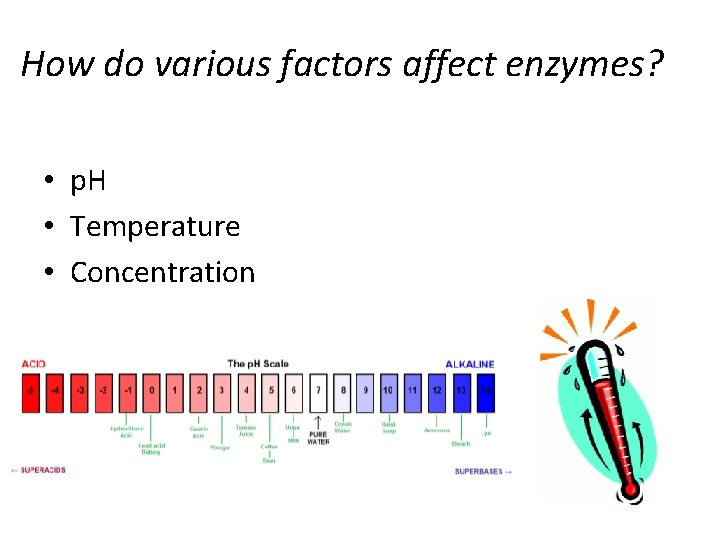
How do various factors affect enzymes? • p. H • Temperature • Concentration

Biology Review L. 18. 12 Properties of Water Discuss the special properties of water that contribute to Earth's suitability as an environment for life.

What are the properties of water and how do they influence life on Earth? List 4 properties of water • Water is a versatile solvent. • Less dense as a solid. • High specific heat – Does not change temperature very fast. • Hydrogen Bonding makes cohesion, adhesion, and surface tension – Allow water to move from roots to leaves in plants

• Identify examples of water's properties that sustain life (e. g. ice floats, water movement through plants, water cycle). • Explain the properties of water at a conceptual level. • Explain how the properties of water make water essential for life on Earth.

How does water dissolve substances? • Water is polar it has a slight positive and negative charge that allows it to separate and dissolve other substances.

Water • Key Vocabulary: adhesion, cohesion, endothermic, exothermic heat capacity, polarity, surface tension, solute, solution, solvent • Hypotonic, Hypertonic, isotonic and Osmosis • http: //www. schooltube. com/video/b 36 a 222 fc dfef 2 db 9 af 8/Properties-Of-Water
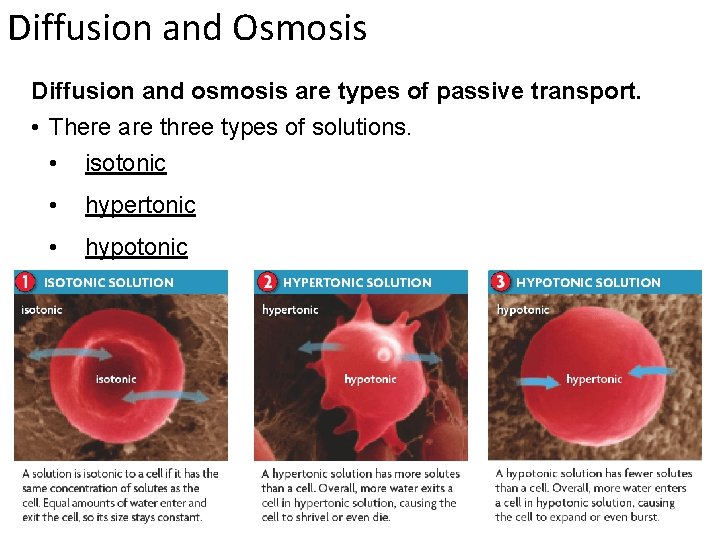
Diffusion and Osmosis Diffusion and osmosis are types of passive transport. • There are three types of solutions. • isotonic • hypertonic • hypotonic

Biochemistry • Key Vocabulary: acid, activation energy, amino acid, atom, base, carbohydrate, catalyst, compound, DNA, element, energy, enzyme, inorganic, lipid, macromolecule, nucleic acid, organic, p. H, protein

Review Questions 1. Water is essential for life. Its special properties make water the single most important molecule in plant life. Which of the following properties of water enables it to move from the roots to the leaves of plants? A. Water expands as it freezes. B. Water is an excellent solvent. C. Water exhibits cohesive behavior. D. Water is able to moderate temperature.
- Slides: 14