Biology of HN Squamous Cell Carcinoma Molecular assay


Biology of H&N Squamous Cell Carcinoma Molecular assay techniques for clonal genetic changes: ---genomic hybridization, microarray tech. SNP analysis n Frequency of chromosome 3 p/9 p loss: --82. 6% in endemic regions of NPC among Chinese n Carcinoma of tonsil: --active role of HPV DNA in carcinogenesis n Incidence of HPV-related oropharyngeal carcinomas: --steadily increasing in industrialized world.

Biomarkers Three strong biomarkers (EBV DNA titer(-), HPV(+) cancer cells, low tumor EGFR expression) ---better Tx. outcome for nasopharyngeal cancer, and oropharyngeal carcinoma n Patient with nasopharyngeal carcinoma and persistent circulating EBV DNA after completion of CRT ---high distant relapse rate; intensive systemic therapy n HPV-associated oropharyngeal SCC ---much better local-regional control and overall SR n

Choice of Treatment Modalities • Cell type and degree of differentiation • Site and extent of primary lesion • Metastatic nodal disease • Characteristics of tumor---superficial vs. invasive • Presence of bone and muscle involvement • Preservation of speech and/or swallowing function • Social status and occupation of patient • Experience and skill of surgeon and radiotherapist

Management of Primary Site Early stage lesion (T 1 N 0 -1, T 2 N 0 -1): - accessible lesion: S or RT less accessible lesion: RT Moderate advanced lesion (T 2 -3 N 0 -2): - combined treatment with S and RT no chance of salvage in recurrence: aggressive Tx Advanced lesion (T 3 -4 N 2 -3): - cure rate is less than 10% with very poor prognosis operable lesion: combined treatment with S, RT, CT inoperable lesion: palliative RT or CT
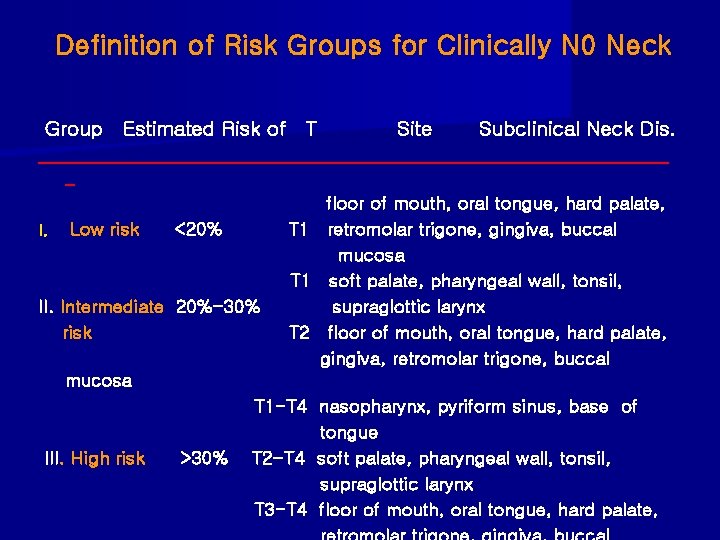
Definition of Risk Groups for Clinically N 0 Neck Group Estimated Risk of T Site Subclinical Neck Dis. ___________________________________ _ floor of mouth, oral tongue, hard palate, I. Low risk <20% T 1 retromolar trigone, gingiva, buccal mucosa T 1 soft palate, pharyngeal wall, tonsil, II. Intermediate 20%-30% supraglottic larynx risk T 2 floor of mouth, oral tongue, hard palate, gingiva, retromolar trigone, buccal mucosa T 1 -T 4 nasopharynx, pyriform sinus, base of tongue III. High risk >30% T 2 -T 4 soft palate, pharyngeal wall, tonsil, supraglottic larynx T 3 -T 4 floor of mouth, oral tongue, hard palate,

Management of Clinically (+) Node N 1 stage: 1. Surgery for primary with selective or modified RND; if p. N 0 or p. N 1 with no ECE---follow-up if > p. N 1 and/or ECE--- postop. RT or chemo-RT 2. Alternatively, RT or chemo-RT for primary with elective neck RT---surgery for persistent disease v >N 2 stage: 1. Surgery for primary with modified, or extended RND; if p. N 1 with no ECE; follow-up if >p. N 1 and/or ECE, postoperative RT or chemo-RT 2. Alternatively, RT or chemo-RT for primary with comprehensive RT for neck; surgery and/or neck dissection for persistent disease and/or node >3 cm v

Unknown Primary of Head and Neck If only 1 c. N (+): 1. Selective or modified radical neck dissection first 2. If no additional lymphadenopathy or extracapsular extension, may observe 3. If >2 LN or ECE: post-op RT or chemo-RT If > 2 c. N (+): 1. Early N 2 disease (N 2 A, early N 2 B): RT 2. Advanced N 2 -N 3: chemo-RT 3. PET/CT 8 weeks after RT or chemo-RT -- risk of residual disease ≤ 5%: observe -- risk of residual disease >5%: (nodes>15 mm, focal lucency, or enhancement or calcification in L/N, ECE or nodal rupture): neck dissection

Failure in Cancer Treatment Surgical failure: “marginal failure” - inability to remove microscopic tumor extension tumor seeding in operation wound. metastasis via lymphatic or hematogenous. Radiation failure: “central failure” - difference of oxygenation between center and margin, tumor core greater than 150 -180 micron often contains hypoxic cells.

Failure Rates in Neck Following Neck Dissection Alone Author Institution Feature Failure Primary Site _____________________________ __ Bartelink, ’ 83 Netherland ECE(+) >40% all H&N Vikram, ’ 84 MSKCC multi-levels 71% one level 37% all H&N Huang, ’ 92 UV ECE(+) 78% all H&N Foote, ’ 93 base Mayo ≥N 2 53% tongue

Prognostic Factors for Loco-regional Relapse after Surgery Moderate Risk High Risk ___________________________ _ 1. Positive margin or 1. Extracapsular spread close margin(<5 mm) 2. Combination of two or 2. Primary tumor in oral cavity more of moderate risk 3. Perineural invasion factors 4. Two or more invaded nodes 5. Two or more invaded node levels 6. Invaded nodes more than 3 cm 7. More than 6 wks between S and RT

Linear Accelerator
Helical Tomotherapy Unit
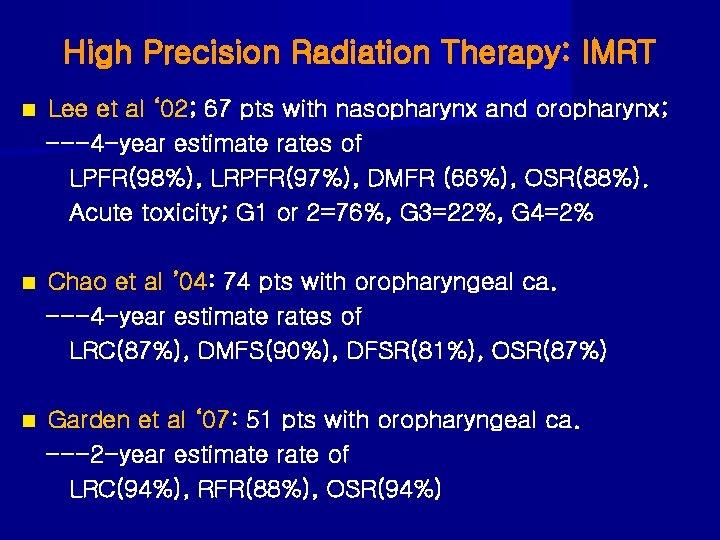
High Precision Radiation Therapy: IMRT n Lee et al ‘ 02; 67 pts with nasopharynx and oropharynx; ---4 -year estimate rates of LPFR(98%), LRPFR(97%), DMFR (66%), OSR(88%). Acute toxicity; G 1 or 2=76%, G 3=22%, G 4=2% n Chao et al ’ 04: 74 pts with oropharyngeal ca. ---4 -year estimate rates of LRC(87%), DMFS(90%), DFSR(81%), OSR(87%) n Garden et al ‘ 07: 51 pts with oropharyngeal ca. ---2 -year estimate rate of LRC(94%), RFR(88%), OSR(94%)
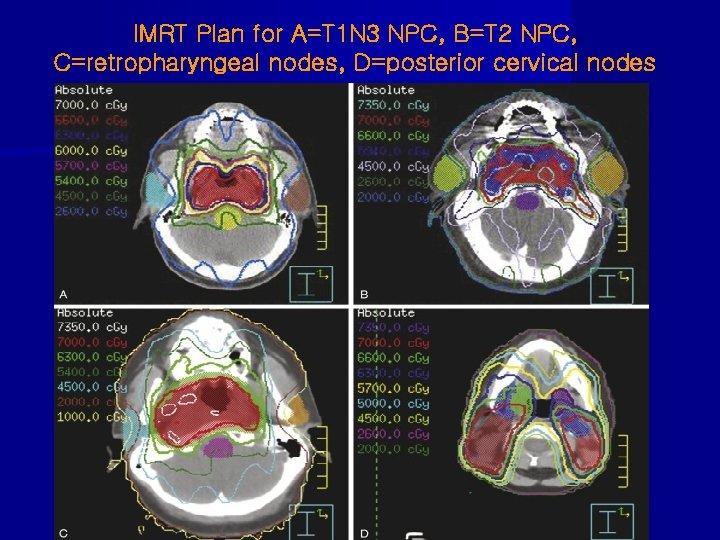
IMRT Plan for A=T 1 N 3 NPC, B=T 2 NPC, C=retropharyngeal nodes, D=posterior cervical nodes
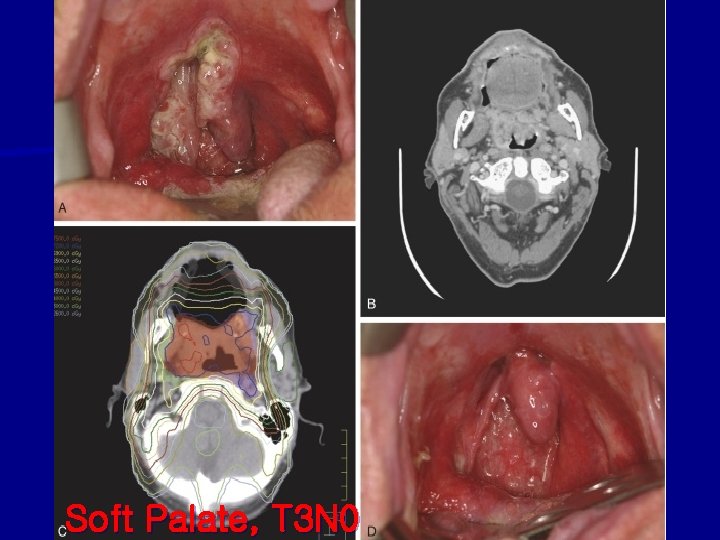
Soft Palate, T 3 N 0

Preoperative Radiotherapy Aims: 1) to prevent marginal recurrence 2) to control subclinical disease in primary or L/N 3) to convert inoperable tumor into operable ones 4) to decrease iatrogenic implant, local recurrence, distant meta Disadvantages: 1) obscured exact extent due to tumor shrinkage 2) delayed surgery-anxiety of patients 3) increased in postoperative complications Application: -medium sized or advanced tumor of oral cavity, larynx, hypopharynx with subcancerocidal dose (4, 500 c. Gy/5 weeks)

Postoperative Radiotherapy Indications: 1) close or positive resection margins 2) extensive perineural spead 3) extensive vascular invasion 4) multiple positive nodes(T 2/3) 5) presence of extracapsular extension(ECE) Dosage: 6, 000 c. Gy/7 weeks for microscopic residual 7, 000 c. Gy/8 weeks for gross residual lesion
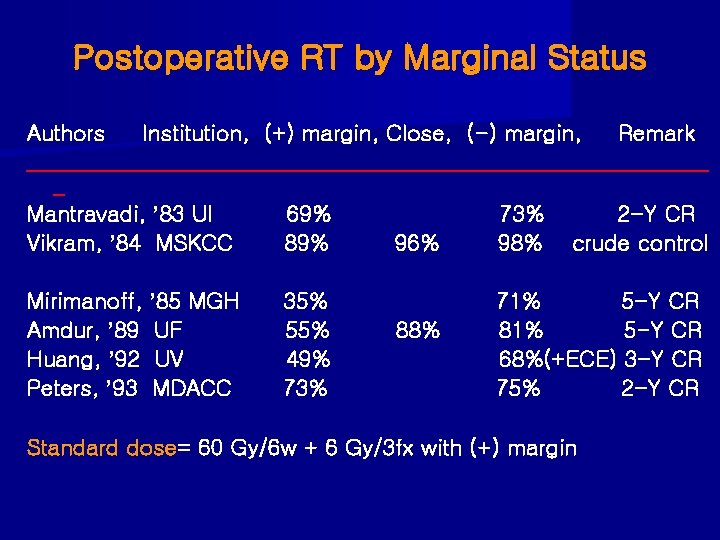
Postoperative RT by Marginal Status Authors Institution, (+) margin, Close, (-) margin, Remark _______________________________ _ Mantravadi, ’ 83 UI 69% 73% 2 -Y CR Vikram, ’ 84 MSKCC 89% 96% 98% crude control Mirimanoff, ’ 85 MGH Amdur, ’ 89 UF Huang, ’ 92 UV Peters, ’ 93 MDACC 35% 55% 49% 73% 88% 71% 5 -Y CR 81% 5 -Y CR 68%(+ECE) 3 -Y CR 75% 2 -Y CR Standard dose= 60 Gy/6 w + 6 Gy/3 fx with (+) margin
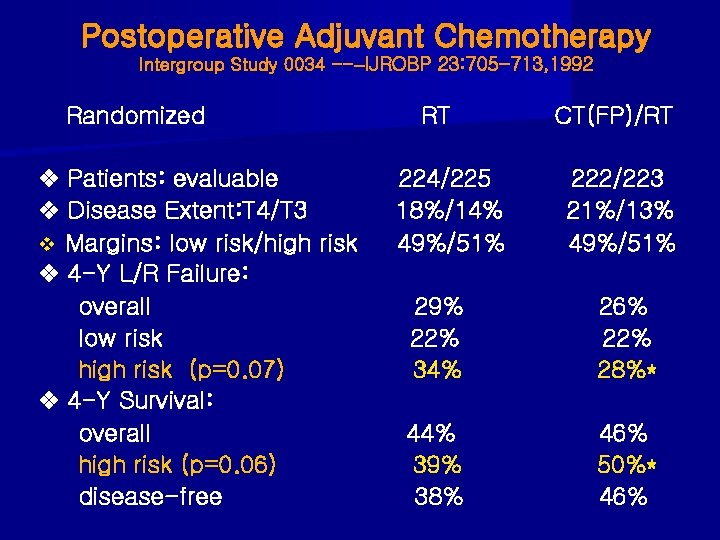
Postoperative Adjuvant Chemotherapy Intergroup Study 0034 --–IJROBP 23: 705 -713, 1992 Randomized Patients: evaluable Disease Extent: T 4/T 3 v Margins: low risk/high risk 4 -Y L/R Failure: overall low risk high risk (p=0. 07) 4 -Y Survival: overall high risk (p=0. 06) disease-free RT 224/225 18%/14% 49%/51% CT(FP)/RT 222/223 21%/13% 49%/51% 29% 22% 34% 26% 22% 28%* 44% 39% 38% 46% 50%* 46%
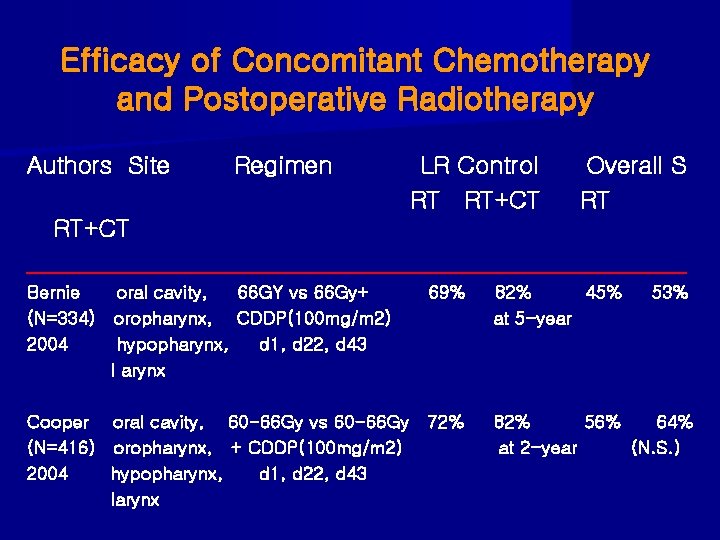
Efficacy of Concomitant Chemotherapy and Postoperative Radiotherapy Authors Site Regimen LR Control RT RT+CT Overall S RT RT+CT ____________________________ Bernie oral cavity, 66 GY vs 66 Gy+ (N=334) oropharynx, CDDP(100 mg/m 2) 2004 hypopharynx, d 1, d 22, d 43 l arynx Cooper (N=416) 2004 69% oral cavity, 60 -66 Gy vs 60 -66 Gy 72% oropharynx, + CDDP(100 mg/m 2) hypopharynx, d 1, d 22, d 43 larynx 82% 45% at 5 -year 53% 82% 56% 64% at 2 -year (N. S. )
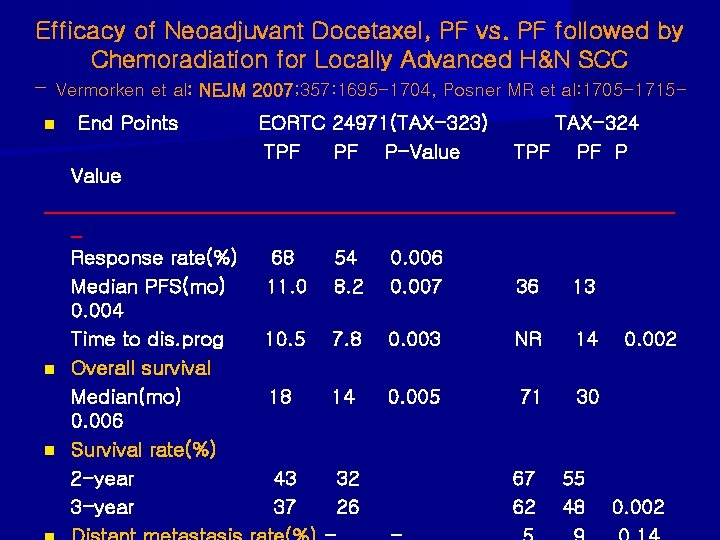
Efficacy of Neoadjuvant Docetaxel, PF vs. PF followed by Chemoradiation for Locally Advanced H&N SCC - Vermorken et al: NEJM 2007; 357: 1695 -1704, Posner MR et al: 1705 -1715 n End Points EORTC 24971(TAX-323) TPF PF P-Value TAX-324 TPF PF P Value ________________________________ _ Response rate(%) 68 54 0. 006 Median PFS(mo) 11. 0 8. 2 0. 007 36 13 0. 004 Time to dis. prog 10. 5 7. 8 0. 003 NR 14 0. 002 n Overall survival Median(mo) 18 14 0. 005 71 30 0. 006 n Survival rate(%) 2 -year 43 32 67 55 3 -year 37 26 62 48 0. 002
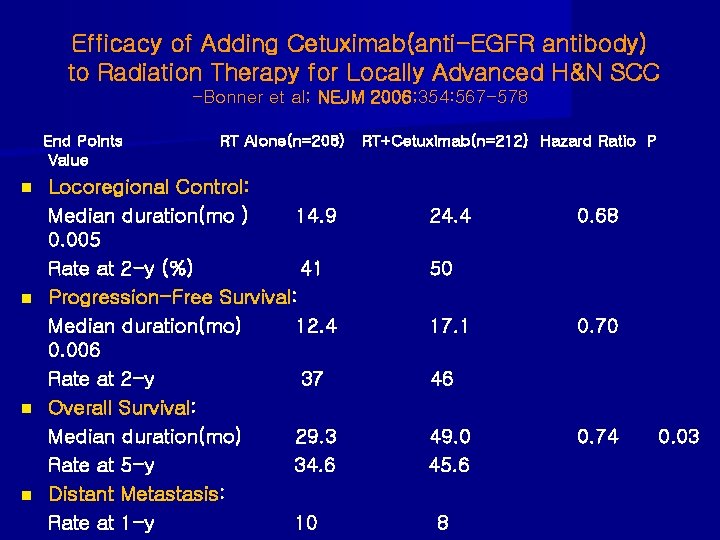
Efficacy of Adding Cetuximab(anti-EGFR antibody) to Radiation Therapy for Locally Advanced H&N SCC -Bonner et al; NEJM 2006; 354: 567 -578 End Points Value n n RT Alone(n=208) RT+Cetuximab(n=212) Hazard Ratio P Locoregional Control: Median duration(mo ) 14. 9 0. 005 Rate at 2 -y (%) 41 Progression-Free Survival: Median duration(mo) 12. 4 0. 006 Rate at 2 -y 37 Overall Survival: Median duration(mo) 29. 3 Rate at 5 -y 34. 6 Distant Metastasis: Rate at 1 -y 10 24. 4 0. 68 50 17. 1 0. 70 46 49. 0 45. 6 8 0. 74 0. 03

Combining Radiation with Chemotherapy(1) Meta-analysis of 16, 485 patients in 87 trials adding concurrent chemotherapy to radiation --increased absol. 5 -Y SR than non-taxane-containing induction chemotherapy with radiation(6. 5% vs. 2. 4%) n n No significant difference in benefit between concomitant chemotherapy trials and adjuvant therapy, as conventional and altered fractionation, or usingle-agent and multi-agent regimens n Benefit of concurrent chemotherapy was due to improvement in local-regional control, and induction chemotherapy due to better effect on distant meta.

Combining Radiation with Chemotherapy(2) n Adding platinum-based concurrent chemotherapy to radiation increases the incidence of late morbidity such as swallowing dysfunction and soft tissue fibrosis or necrosis n Postoperative adjuvant radiation with concurrent cisplatin is recommended only for patients with histologically proven ECE, or (+)surgical margin n Postoperative radiotherapy without chemotherapy remains standard care for patients with intermediaterisk features; such as perineural invasion, close margins, and positive lymph nodes

Complications of Radiotherapy Minor complications: - xerostomia, loss of appetite, dental caries epilation abnormal facial growth in children Major Complications: - soft tissue ulceration oro-cutaneous fistula radiation-induced endocrine functional changes

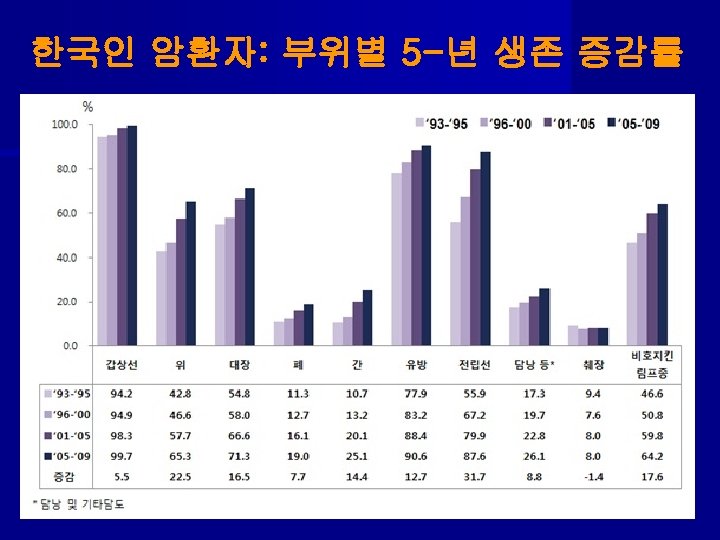
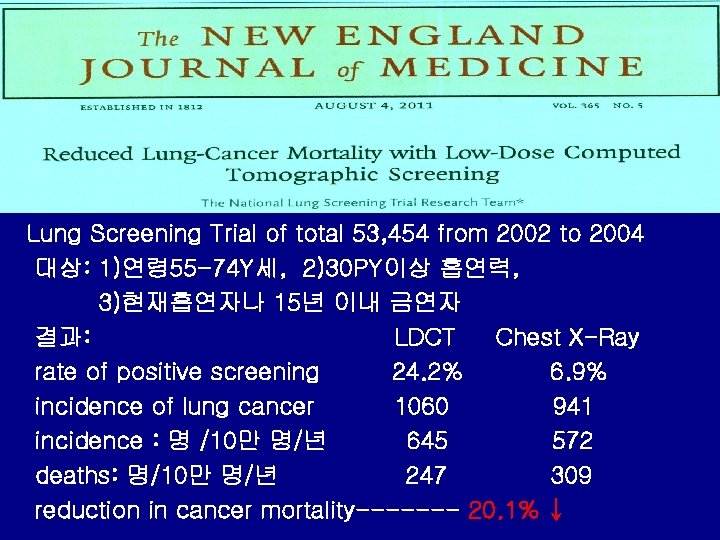
Lung Screening Trial of total 53, 454 from 2002 to 2004 대상: 1)연령 55 -74 Y세, 2)30 PY이상 흡연력, 3)현재흡연자나 15년 이내 금연자 결과: LDCT Chest X-Ray rate of positive screening 24. 2% 6. 9% incidence of lung cancer 1060 941 incidence : 명 /10만 명/년 645 572 deaths: 명/10만 명/년 247 309 reduction in cancer mortality------- 20. 1% ↓
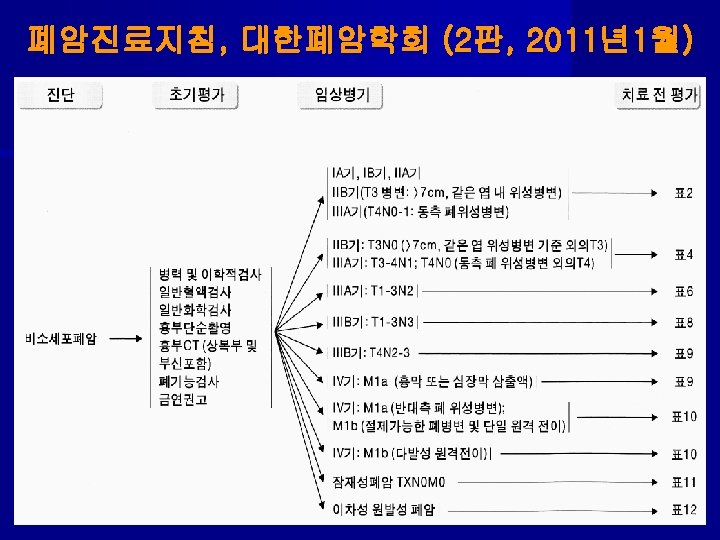
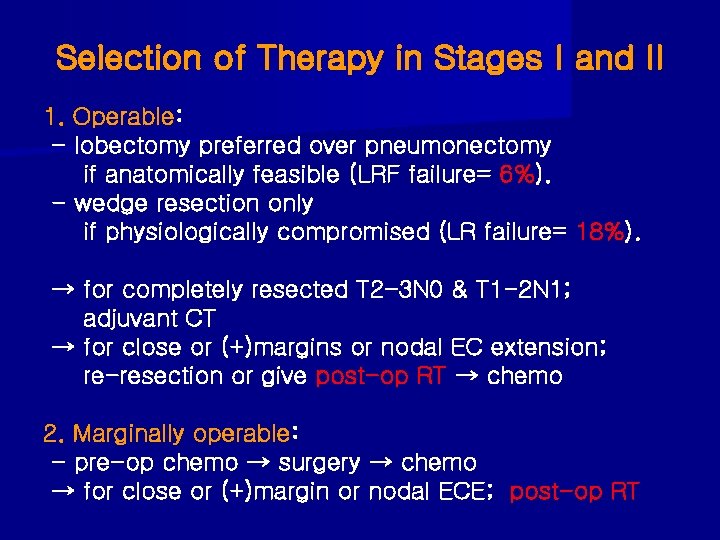
Selection of Therapy in Stages I and II 1. Operable: - lobectomy preferred over pneumonectomy if anatomically feasible (LRF failure= 6%). - wedge resection only if physiologically compromised (LR failure= 18%). → for completely resected T 2 -3 N 0 & T 1 -2 N 1; adjuvant CT → for close or (+)margins or nodal EC extension; re-resection or give post-op RT → chemo 2. Marginally operable: - pre-op chemo → surgery → chemo → for close or (+)margin or nodal ECE; post-op RT

Selection of Therapy in Stages I and II 3. Inoperable: - definite RT to primary ± involved nodes - 1. 8 -2 Gy/fraction to ≥ 65 Gy - if peripheral tumor or poor PS, may hypofractionated with 4 Gy /fraction to 48 Gy to primary tumor only - dose escalation >70 Gy& radiosurgical technique (~60 Gy/3 fx) to provide improved LC than standard - if pt. can tolerate it, give concurrent chemo-RT for T 3 No or N 1 disease.

Selection of Therapy in Stages IIIA • Operable or marginally operable: - concurrent chemo-RT(45 Gy) → restage → if no progression → surgery → chemo for N 2 nodes - alternatively, chemo alone → restage → if no progression → surgery → chemo± post-op RT for close or (+)margin, nodal ECE, or N 2 disease - if unresectable after restaging → complete definitive concurrent chemo-RT(63 Gy) • Inoperable: - concurrent chemo-RT(63 Gy) → adjuvant chemo, or induction chemo → concurrent chemo-RT
Selection of Therapy in Stages IIIB • concurrent chemo-RT(63 Gy) → adjuvant chemo, or induction chemo → concurrent chemo-RT • if T 4 N 0 -1, may treat with surgery → chemo ± RT, or chemo ± RT → surgery → chemo • if pleural effusion, local treatment as necessary (e. g. pleurodesis) & treat as stage IV

Selection of Therapy in Stages IV • ECOG Performance Status 0 -2: --- chemo ± palliative RT. First-line chemo uses 2 agents for 3 -4 cycles. Addition of bevazumab to carboplatin-paclitaxel improves survival • ECOG Performance Status 3 -4: --- best supportive care

Surgical Treatment of Lung Cancer Lobectomy---superior to limited resection as wedge resection or segmentectomy n Following surgical resection, 5 -year survival --- 73%(IA), 58%(IB), 46%(IIA), 36%(IIB) n ● ● Failure patterns after surgery: ---70%(distant), 20%(local), 10%(local&distant) Requires significant recovery time unlike breast cancer surgery.

Unresectable Reasons of Tumor 1. cytologically positive effusion 2. vertebral body invasion 3. invasion/encasement of great vessels 4. tumor extensively involving carina or trachea 5. recurrent laryngeal nerve paralysis 6. extensive mediastinal L/N metastasis 7. N 3 disease

Preoperative Radiation Therapy Advantages: - improve resectability of larger tumors, - sterilize cells beyond margin of resection, - prevent dissemination by surgical manipulation, - use of lower radiation dose n Disadvantages: - not known precise surgical stage, - not appreciated exact anatomic extent of tumor, - increased risk of postoperative complications n

Radical RT in Medically Inoperable NSCLC Author No. Dose(Gy) 5 -Y SR(%) LF(%) ___________________________ _ Standard fractionation: Morita ’ 97 149 65 22 44 Sibley ’ 98 141 64 15 50 SEER ’ 05 4, 300 RT improve MS, but not 5 YSR ● Stereotactic body radiation therapy: Nagata ‘ 05 31 12 x 4 83(5 -y) Timmerman ’ 06 7 0 20 x 3 55(2 -y) Fakiris ‘ 09 20 -22 x 3 43(3 -Y) RTOG-0236 ’ 09 55 20 x 3 72(2 -y) ● 6 9 12 6

Indications of Post-operative RT • Microscopic disease: - bronchial resection margin, - vascular margin, - chest wall • High risk disease: - mediastinal lymph node involvement, - inadequate mediastinal resection, - extracapsular extension, - close margin
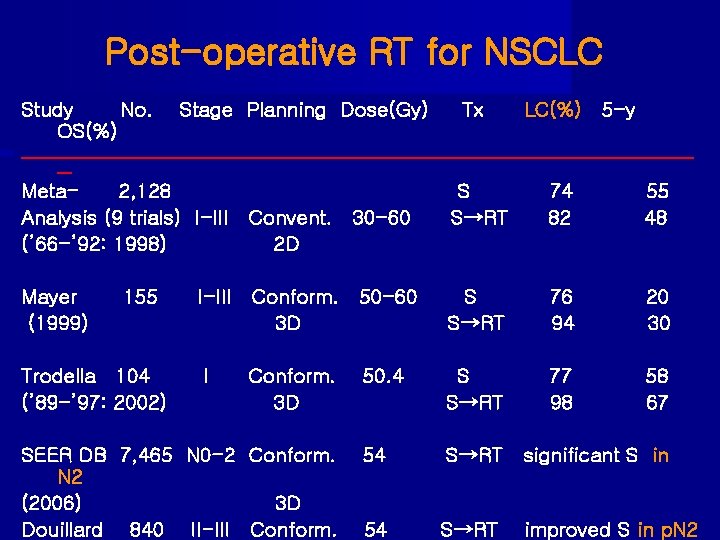
Post-operative RT for NSCLC Study No. OS(%) Stage Planning Dose(Gy) Tx LC(%) 5 -y ________________________________________________ __ Meta 2, 128 Analysis (9 trials) I-III Convent. 30 -60 (’ 66 -’ 92: 1998) 2 D S S→RT 74 82 55 48 Mayer (1999) I-III Conform. 50 -60 3 D S S→RT 76 94 20 30 I 50. 4 S S→RT 77 98 58 67 54 S→RT significant S in 54 S→RT 155 Trodella 104 (’ 89 -’ 97: 2002) Conform. 3 D SEER DB 7, 465 N 0 -2 Conform. N 2 (2006) 3 D Douillard 840 II-III Conform. improved S in p. N 2

Indications of Adjuvant Chemotherapy following Complete Resection of NSCLC n Poorly differentiated n Lympho-vascular invasion n Over 4 cm mass in size n Invasion of visceral pleura n Close resection margin n Incomplete resection(R 1, R 2) in stage IB -re-resection or radiation therapy + chemo

Adjuvant Chemotherapy following Complete Resection of NSCLC • 1995, Non-Small Cell Lung Cancer Collaborative Group; - metaanalysis of 52 randomized trials of 4, 467 patients trend toward improved survival by 5% (p=NS) when cisplatin-based chemotherapy was used. • 2004, International Adjuvant Lung Cancer Trial (IALT); -1, 867 patients after S vs S +cisplatinum-based CT gained 40. 4% vs 44. 5% 5 -Y SR small, improvement (4%) with use of postoperative chemotx. in stage I, III pts. • 2005, NCIC BR. 10(NEJM); 482 pts with completely resected stage I-II, observation vs cispl/vinorelibone x 4 cycles, chemo improved RFS (49%→ 61%) , 5 -y OS (54%→ 69%), • 2006, ANITA in 841 pts: ---MS=44 to 66 Mo, 5 -y SR=8. 6%↑
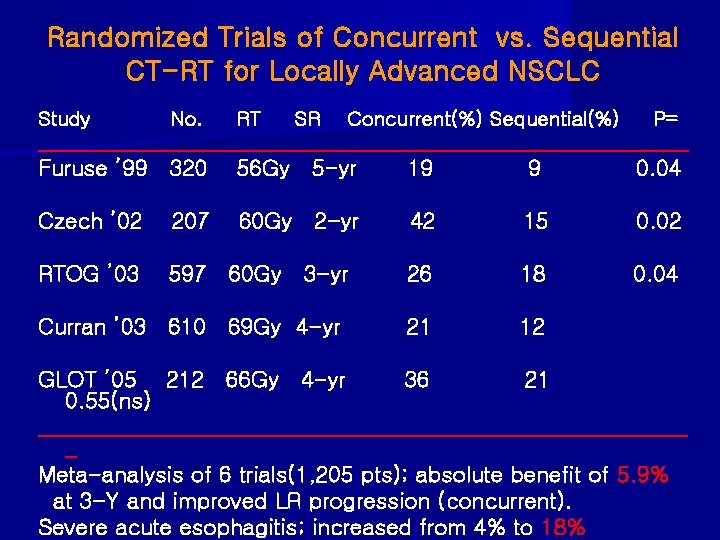
Randomized Trials of Concurrent vs. Sequential CT-RT for Locally Advanced NSCLC Study No. RT SR Concurrent(%) Sequential(%) P= _________________________________ Furuse ’ 99 320 56 Gy 5 -yr 19 9 0. 04 Czech ’ 02 207 60 Gy 2 -yr 42 15 0. 02 RTOG ’ 03 597 60 Gy 3 -yr 26 18 0. 04 21 12 Curran ’ 03 610 69 Gy 4 -yr GLOT ’ 05 212 66 Gy 4 -yr 36 21 0. 55(ns) ______________________________ _ Meta-analysis of 6 trials(1, 205 pts); absolute benefit of 5. 9% at 3 -Y and improved LR progression (concurrent). Severe acute esophagitis; increased from 4% to 18%
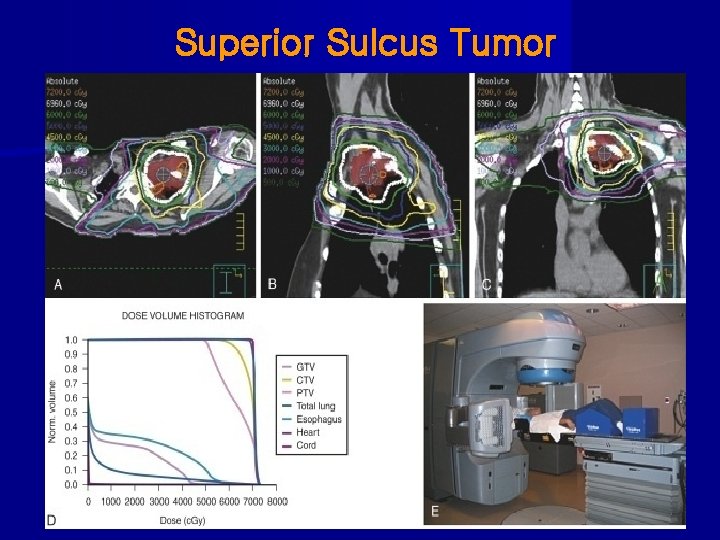
Superior Sulcus Tumor

Superior Sulcus Tumor • Int-0160 (Rush, 2001); phase II trial of 111 pts with T 3 -4 N 0 -1 SST treated with concurrent chemo-RT(45 Gy) → restaging → surgery (if no progression) → chemo(platinum/etoposide) x #2 • 86% of pts had surgery 65% of pts had p. CR or minimum residual dis. 2 -year OS was 55%(70% for pts with p. CR) local relapse was only 23% the most common site of relapse ---brain

Treatment Recommendation of Small Cell Lung Cancer • Limited: concurrent cisplatin + etoposide (every 3 wks x 4 c) with early RT(45 Gy/1. 5 Gy bid). -- If CR, prophylactic cranial RT(25 Gy/2. 5 Gy/fx). -- consider resection and chemo for T 1 N 0. • Extensive: combination chemo+palliative RT to symptomatic sites. For patients with PR or CR, consider prophylactic cranial RT(25 Gy/10 Fx). If brain metastasis(+), WBRT(30 -37. 5 Gy/10 -15 Fx)

Role of RT in Small Cell Lung Cancer, LS • Pignon et al (’ 92): meta-analysis of RT for SCLC - 3 yr survival: CT=8. 9% vs CT+RT=14. 3%(p=0. 001) - improved 3 -year OS by 5. 4% • Turrisi (‘ 99): 417 pts with limited stage SCLC - randomized to concurrent cisplatin, etopiside with 45 Gy/1. 8 Gy QD or 45 Gy/1. 5 Gy bid. - bid arm increased 5 -yr OS (23 vs 16%) and decreased local failure (36 vs 52%) compared to QD • Komaki (‘ 09): accelerated high-dose RT with EP CT - RT was given to large field to 28. 8 Gy/1. 8 Gy QD, then 32. 4 Gy/1. 8 Gy bid(am, pm). Total RT dose 61. 2 Gy in 5 weeks. Two-y OS=37%, 2 -y LC=80%.
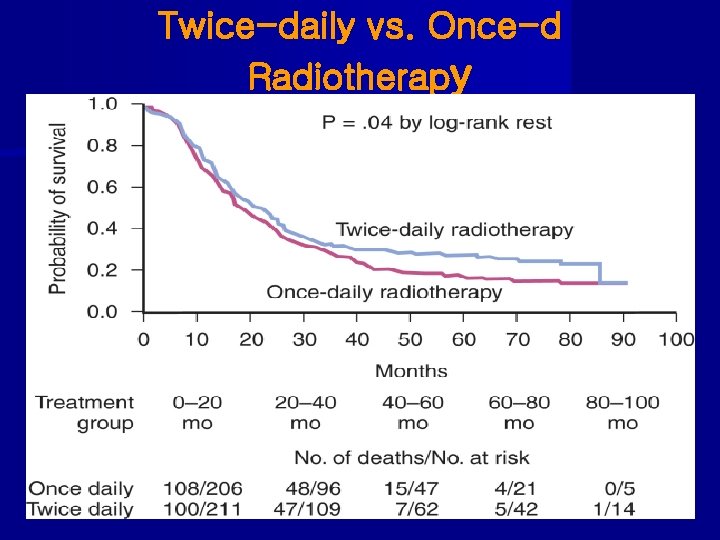
Twice-daily vs. Once-d Radiotherapy

Role of RT in Small Cell Lung Cancer, ES • Jeremis (’ 99): 210 Ex. S-SCLC pts treated with 3 cycles of EP with local PR or CR; accelerated hyperfx. RT(54 Gy/1. 5 Gy bid) and CT vs. 4 cycles of CT alone. - chemo-RT improved 5 -y OS(9. 1 vs 3. 7%) and MS (17 vs 11 months) vs. those treated with CT alone. • Slotman (‘ 07): 286 pts with ES-SCLC with response to CT randomized to PCI vs. no treatment. - PCI reduced 1 -year incidence of symptomatic brain mets(14. 6 vs 40. 4%) and improved OS(27. 1 vs 13. 3%) compared to control group.
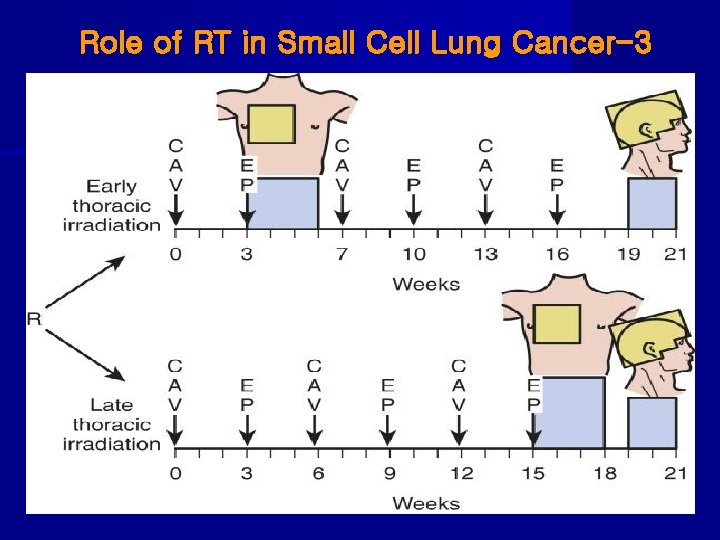
Role of RT in Small Cell Lung Cancer-3

Timing of CRT: Early vs. Late in Small Cell Lung Cancer • Murray (’ 93 NCI of Canada): concurrent CT+TRT - median survival: early(#2)=21. 2 mo, late=16. 0 mo 5 -year survival: early=22%, late=13%(p<0. 013) • Tanaka et al (’ 99, JCOG): concurrent vs. sequential - median survival: early=31. 3 mo, late=20. 8 mo 5 -year survivial: early=30%, late=15% • Fried (’ 04, meta-analysis with 1, 524): improved 2 -y SR beginning before 9 weeks after CT, or before 3 rd CT • De Ruysscher (’ 06, meta-analysis with 1, 056): higher 5 -y SR in early RT
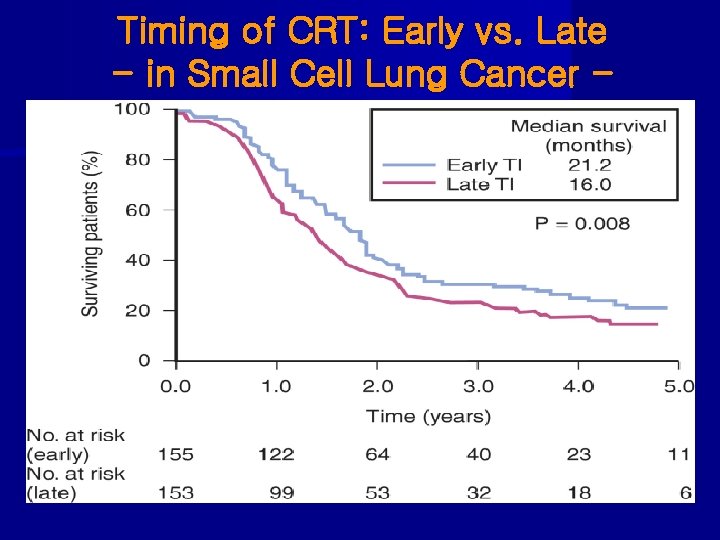
Timing of CRT: Early vs. Late - in Small Cell Lung Cancer -

Prophylactic Cranial Irradiation - Auperin ’ 99: PCI Overview Collaborative Group 대상: CR 보인 소세포 폐암 환자를 전향적 연구로 metaanaly 방법: 25 Gy/10 Fx~30 Gy/15 Fx 예방적 전뇌 치료 결과: 1. decreased brain metastasis; 24% to 6% (p<0. 001) 2. increased disease-free survival (p<0. 001) 3. increased overall survival (15% vs 21% at 3 years) 4. no difference in neuropsychologic function 권장: 1. 항암제완료 후 2주 지나서 CR이 된 환자 , 2. 방사선량=25 -30 Gy/2. 5 -3 Gy Slotman ’ 07 (EORTC trial with 286 pt. PCI in extensive stage: 1 -y brain meta-free= 14. 6% to 40. 4%,
- Slides: 56