BIOLOGY NOTES BIOLOGY BIOCHEMISTRY PART 2 PAGES 36

BIOLOGY NOTES BIOLOGY & BIOCHEMISTRY PART 2 PAGES 36 -39

Standards 1. 4 ___ Interpret formula representations of molecules and compounds in terms of composition and structure Essential Question 1. Why are carbon based molecules the foundation of life?

I. Matter = anything that occupies space _____ and mass has _____. List three different types of matter…

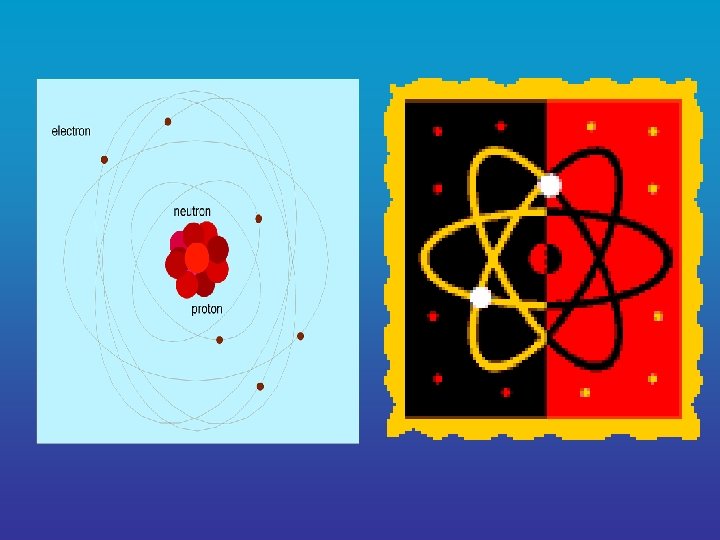
A. The Structure of Matter matter 1. Atoms = the basic units of ____ • Atoms can be broken down into smaller subatomic parts called ______ particles a. proton = subatomic particle that positive charge has a ____

b. neutron = subatomic article that no charge has ____ atom vnucleus = the center of the ______ where protons and neutrons are located c. electron = subatomic particle that has a ____ negative charge velectrons are in constant ____ motion nucleus in an area around the _____ known as the electron cloud

v. The electron cloud has different energy levels that can hold a ____ specific number of electrons Ø 1 st level (closest to the nucleus) = ______ electrons 2 Ø 8 2 nd level = ______ electrons

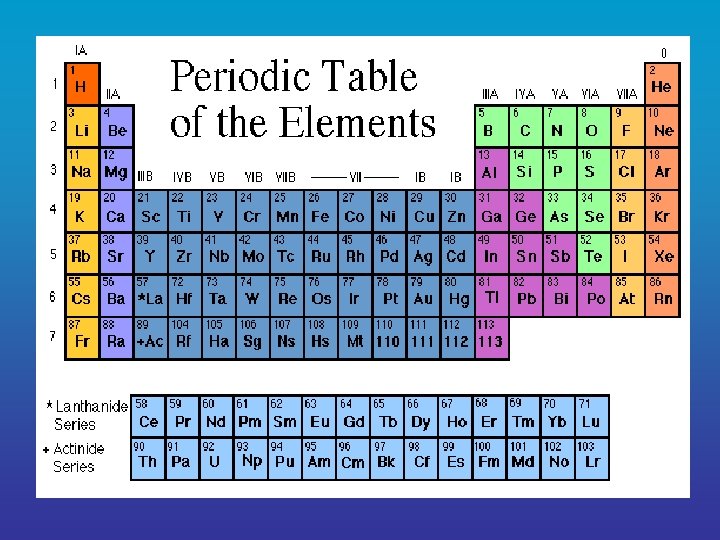
2. Elements Element = a pure substance that consists 1 entirely of _______ type of atom • elements cannot be _______ broken down into simpler substances • each element is represented by a _______ chemical symbol
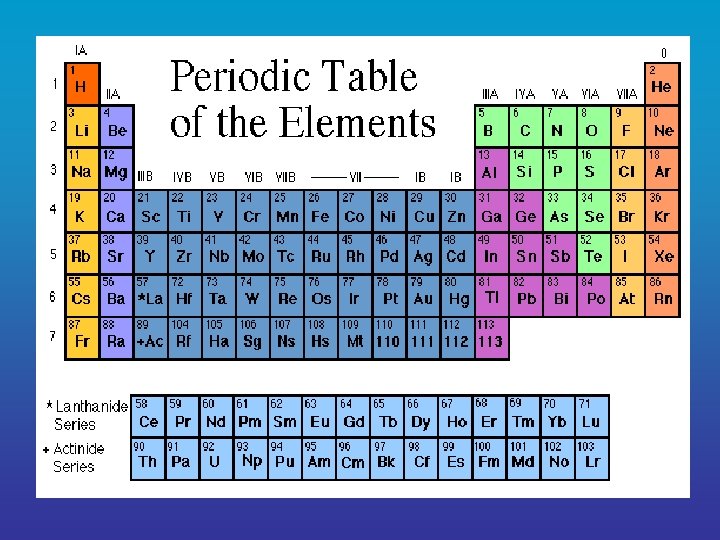

O Oxygen = ___ Na Sodium = ___ Cl Chlorine = ___ S Sulfur = ___ H Hydrogen = ___ C Carbon = ___ K Potassium = ___ Mg Magnesium = ___ Ca Calcium = ___ P Phosphorus = ___ N Nitrogen = ___ Fe Iron = ___

• Of the naturally occurring elements, only life about 25 are essential to ______. • Of these 25, four ___, C ___, H ___, O and ___ N make up 96% of the mass of a human and are essential for life. • The rest of the elements are present in small amounts. very _______

protons 1. Atomic Number = the number of _____ in an element. fingerprint because • This is the element’s _____ no two elements have the same amount of protons. e. g. C = 6, N= 7, O = 8, etc.


II. The Combination of Matter A. Compounds Compound = a substance formed by the chemical 2 or more ____ combination of ___ elements. e. g. Na. Cl (salt) H 2 O (water) C 6 H 12 O 6 (glucose)

• chemical formula = a shorthand method compounds of writing compositions of ______ e. g. salt = Na. Cl water = H 2 O • subscript = in chemical formulas, the small number _______ next to the element that tells how many ______ atoms of that element are in the compound e. g. H 2 O = 1 atom O, 2 atoms H C 6 H 12 O 6 = 6 atoms C, 12 atoms H, 6 atoms O

How many atoms of each element are there in…. . C 7 O 3 H 2 = 7 atoms carbon, 3 atoms oxygen, 2 atoms hydrogen C 57 H 110 O 6 = 57 atoms carbon, 110 atoms hydrogen, 6 atoms oxygen

C 7 N 4 O 2 H 9 = 7 atoms carbon, 4 atoms nitrogen, 2 atoms oxygen, 9 atoms hydrogen 1 atom carbon, CO 2 Ca. Na = 2 atoms oxygen, 1 atom calcium, 1 atom sodium

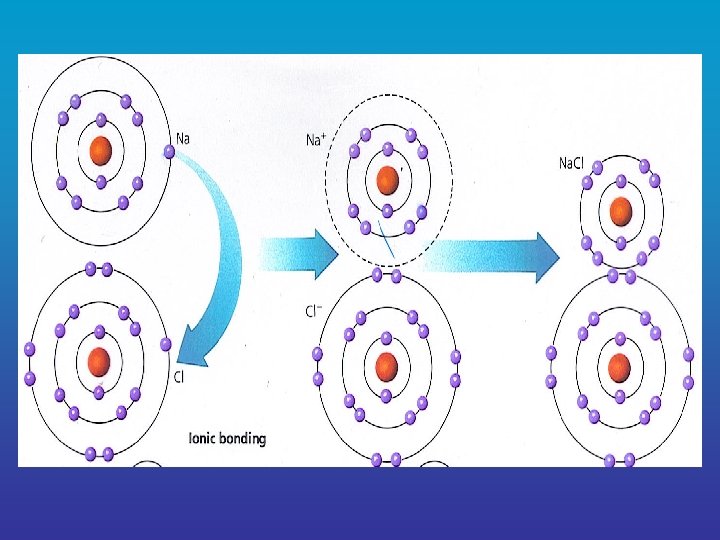
B. Chemical Bonds – the formation of compounds Elements will only combine to form compounds when they become more stable by forming a compound ______ • Atoms of different elements become eight more stable when they have ______ electrons in their outer ______ energy level (valence shell = outer energy level)

1. Types of Chemical Bonds a. Ionic Bond = a chemical bond formed between two atoms ______ when one or electrons are transferred more _____ between the atoms resulting in atoms of _____ opposite charge which are attracted to each other e. g. salt

b. Covalent Bond = a chemical bond formed between two atoms _____ when they electrons share two or more _____ e. g. water, carbohydrates, & fats
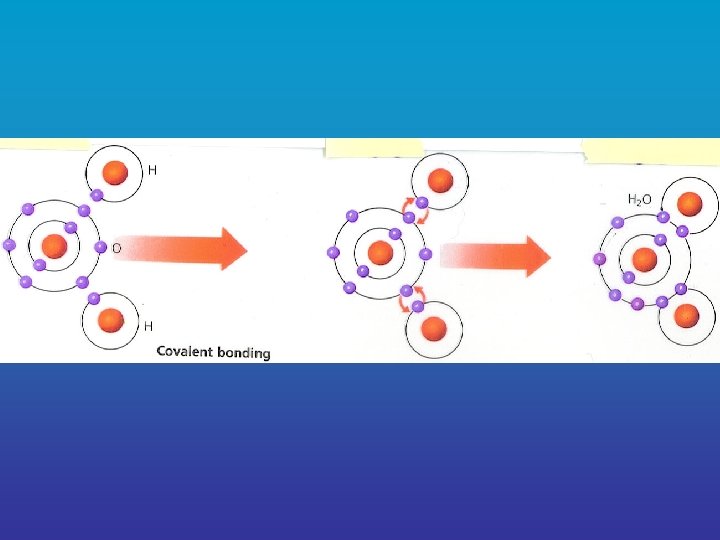

• molecule = the structure that results when covalent bonds atoms are joined by _____ vmolecules make up most of the living things compounds found in ______ vsuperscript = in chemical formulas, the front of large number found in ____ compounds that tells how many molecules of that compound exist ______ e. g. 2 H 20 = 2 molecules of water (4 atoms of Hydrogen, 2 atoms of Oxygen)

How many molecules? vs. How many atoms? 5 molecules of polyester vs. 50 atoms carbon, 5 C 10 O 4 H 4 = 20 atoms oxygen, 20 atoms hydrogen 3 molecules of bug spray vs. 3 C 12 H 13 ON = 36 atoms of carbon 39 atoms of hydrogen 3 atoms of oxygen 3 atoms of nitrogen
- Slides: 25