Biology comes from the Greek words bio meaning

Biology comes from the Greek words “bio” meaning “life” and “logy” meaning “the study of. ” Organisms share common characteristics that, taken together, define life. Organisms: Acquire and use materials and energy Actively maintain organized complexity Perceive and respond to stimuli Grow Reproduce Have the capacity to evolve, collectively
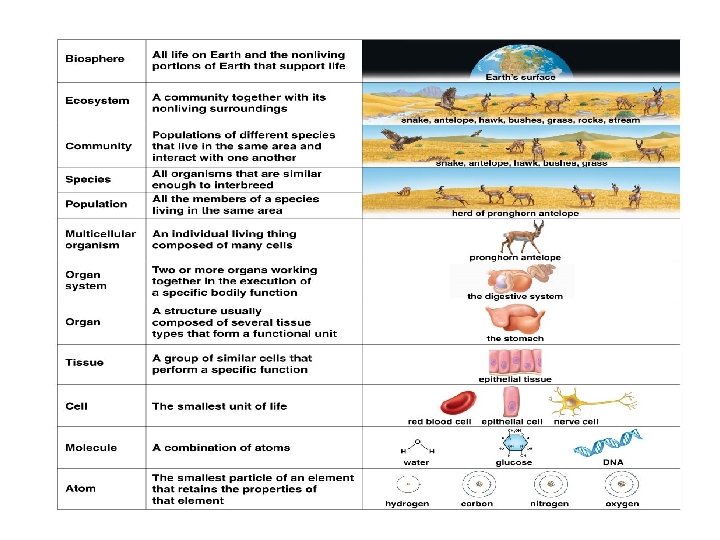

An atom is the smallest particle of an element retaining the properties of an element Atoms combine to form molecules Molecules provide the building blocks for cells, which are the smallest unit of life
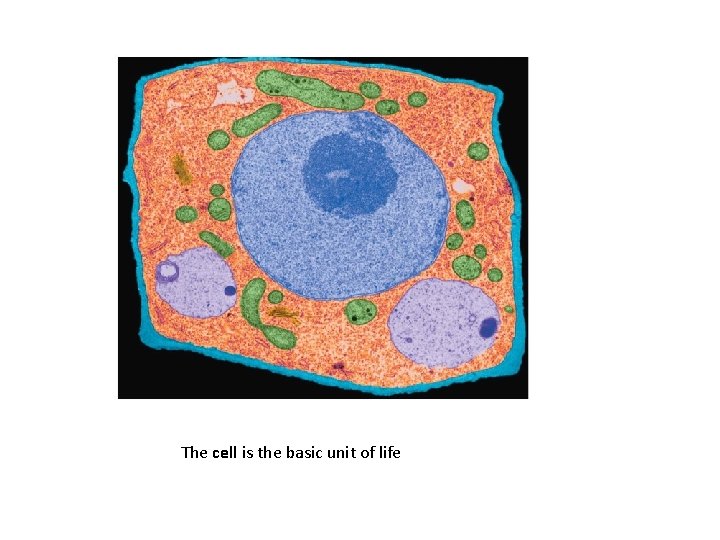
The cell is the basic unit of life

Multicellular (many-celled) organisms are found in Eukarya, within the kingdoms • Fungi • Plantae • Animalia Unicellular (single-celled) organisms are found in • Bacteria • Archaea v Multicellularity occurs only among the Eukarya

The six steps of scientific inquiry 1. Observation of a specific phenomenon 2. The observation, in turn, leads to a question 3. The question leads to formulation of a hypothesis, based on previous observations, which is offered as an answer to the question 4. 5. 6. The hypothesis leads to a prediction, which is the expected outcome of testing if the hypothesis is correct The prediction is tested by carefully designed additional observations or carefully controlled manipulations called experiments The experiments produce results that either support or refute the hypothesis, allowing the development of a conclusion

Biologists test hypotheses using controlled experiments Two types of situations are established A baseline or control situation in which all possible variables are held at a constant. An experimental situation in which one factor, variable, is manipulated to test the hypothesis to determine that this variable is the cause of an observation v Science is useless unless communicated

Science requires communication. • Scientists communicate in many ways; face to face, at symposia (scientific meetings), via journals (including primary journal articles and secondary journal articles). • Primary journal articles are those which are written by the person/persons who actually performed the experiment and tend to include sections like a materials and methods section and a results section. • Secondary journal articles tend to be written by an expert in a particular field and usually sum the conclusions of many different researchers. This will give an overview of the current state of a particular area of scientific research.

Elements: An element is a substance that cannot be broken down or converted into another substance by ordinary chemical means. ü For example, carbon is an element, and a “perfect” diamond is made of pure carbon. If you cut the diamond into pieces, each one is still a diamond, made of carbon. If you kept cutting into the smallest pieces that still looked and felt like shiny diamonds you could theoretically get to individual carbon atoms. Atoms are the basic structural units of matter.

Atoms: The subatomic particles called protons, neutrons and electrons are the ones we shall consider in this course. • Protons – Located in the center of the atom (the nucleus), have a mass of 1 amu and are positively charged (+1) Neutrons – Located in the nucleus, have a mass of 1 amu and are neutral (have no charge) • Electrons – Orbit the nucleus, have essentially no mass and are negatively charged (-1).

Atomic number: Each particular type of atom has a specific number of protons. • Hydrogen has 1 proton, helium has 2 protons and so on. The atomic number of an element is its number of protons. So, the atomic number of hydrogen is 1, the atomic number of helium is 2, etc. Also, atoms are electrically neutral. Therefore, the number of electrons in an atom must equal the number of protons. Isotopes are atoms with an “unusual” number of neutrons. Some isotopes, but not all, are radioactive. They are unstable and release energy for a certain amount of time. The amount of energy and the time that the energy is emitted tends to be specific to the isotope. Think about cancer and medical testing.

2. 2 How Do Atoms Form Molecules?

Ionic bonds: formed by passing an electron from one atom to another • One partner becomes positive, the other negative, and they attract one another. ü Na+ + Cl– becomes Na. Cl (sodium chloride) Positively or negatively charged atoms are called ions.

Covalent bonds: bond between two atoms that share electrons in their outer electron shell ü For example, an H atom can become stable by sharing its electron with another H atom, forming H 2 gas.

Covalent bonds produce either nonpolar or polar molecules. Ø Nonpolar molecule: atoms in a molecule equally share electrons that spend equal time around each atom, producing a nonpolar covalent bond

Polar molecules: atoms in a bond unequally share electrons, producing a polar covalent bond One atom in the bond has a more positive charge in the nucleus, and so attracts electrons more strongly, becoming the negative pole of the molecule. The atom in the bond that has a less positive charge in the nucleus gives up electrons, becoming the positive pole of the molecule. ü For example, H 2 O is a polar molecule The (slightly) positively charged pole is around each hydrogen The (slightly) negatively charged pole is around the oxygen

Hydrogen bonds: weak electrical attraction between positive and negative parts of polar molecules ü Example: the negative charge of oxygen atoms in water molecules attract the positive charge of hydrogen atoms in other water molecules

Water interacts with many other molecules. Oxygen released by plants during photosynthesis comes from water. Water is used by animals to digest food and thus breakdown large biomolecules – hydrolysis reactions. Water is produced in chemical reactions that combine small biomolecules forming larger, more complex ones – dehydration synthesis reactions.

Dehydration synthesis The construction of large molecules yields water. Small molecules are joined together to form large molecules. During the joining of small molecules, water is released. This water-releasing reaction is called dehydration synthesis
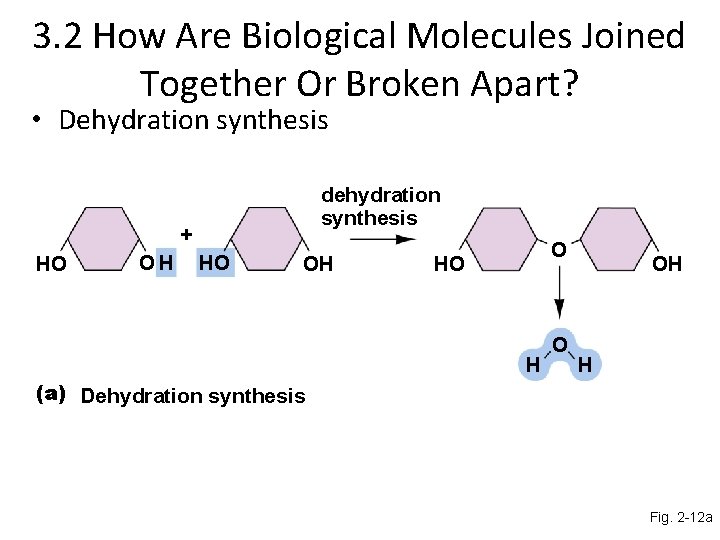
3. 2 How Are Biological Molecules Joined Together Or Broken Apart? • Dehydration synthesis dehydration synthesis + HO OH O HO H O OH H (a) Dehydration synthesis Fig. 2 -12 a

Hydrolysis reactions During the breakdown of large molecules, covalent bonds are broken, separating the subunits
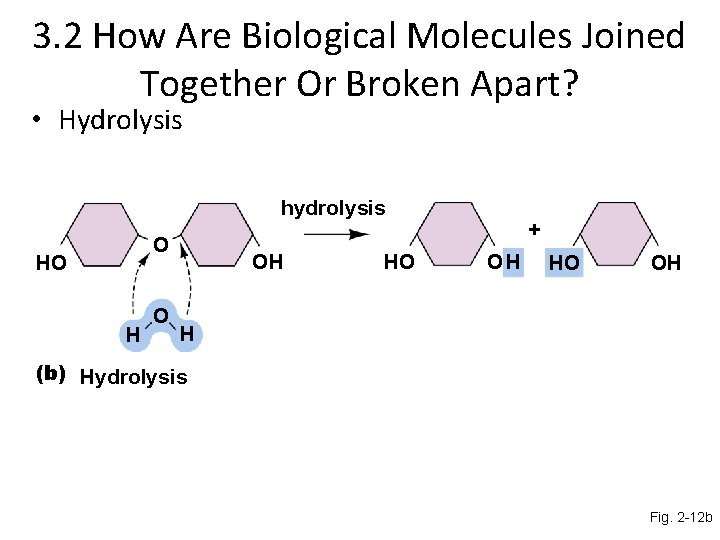
3. 2 How Are Biological Molecules Joined Together Or Broken Apart? • Hydrolysis hydrolysis O HO H O OH HO + OH HO OH H (b) Hydrolysis Fig. 2 -12 b
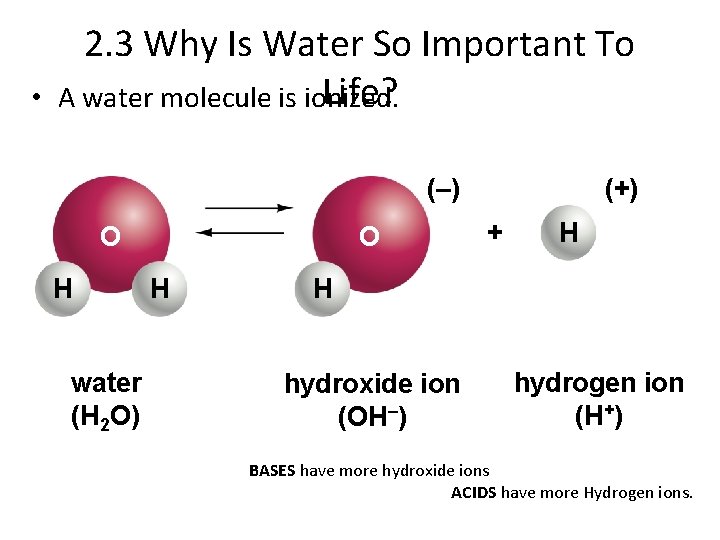
• 2. 3 Why Is Water So Important To Life? A water molecule is ionized. (–) O H water (H 2 O) O H (+) + H H hydroxide ion (OH–) hydrogen ion (H+) BASES have more hydroxide ions ACIDS have more Hydrogen ions.

p. H measures acidity. Acids have a p. H below 7. Bases have a p. H above 7. Neutral solutions have a p. H of 7. Buffers are substances that maintain a constant p. H in a solution BASES have more hydroxide ions. ACIDS have more Hydrogen ions.

0 100 1 10– 1 2 10– 3 3 p. H value 10– 4 5 10– 5 H+ 6 10– 6 7 10– 7 8 10– 8 (H+ > OH–) 10– 9 increasingly acidic 10 10– 10 11 10– 11 neutral (H+ = OH–) (H+ < OH–) 12 13 10– 12 10– 13 drain cleaner (14. 0) 1 molar sodium hydroxide (Na. OH) oven cleaner (13. 0) household ammonia (11. 9) washing soda (12) phosphate detergents chlorine bleach (12. 6) toothpaste (9. 9) seawater (7. 8– 8. 3) baking soda (8. 4) water from faucet milk (6. 4) pure water (7. 0) blood, sweat (7. 4) normal rain (5. 6) urine (5. 7) black coffee (5. 0) orange (3. 5) tomatoes beer (4. 1) vinegar, cola (3. 0) stomach acid (2) lemon juice (2. 3) 1 molar hydrochloric acid (HCl) • 2. 3 Why Is Water So Important To Life? The p. H scale 4 9 14 10– 14 increasingly basic concentration in moles/liter Fig. 2 -11

An organic molecule is one that contains carbon. This is possible because carbon has four electrons in its outermost shell, leaving room for four more electrons from other atoms. Therefore, carbon can form many bonds with other atoms.
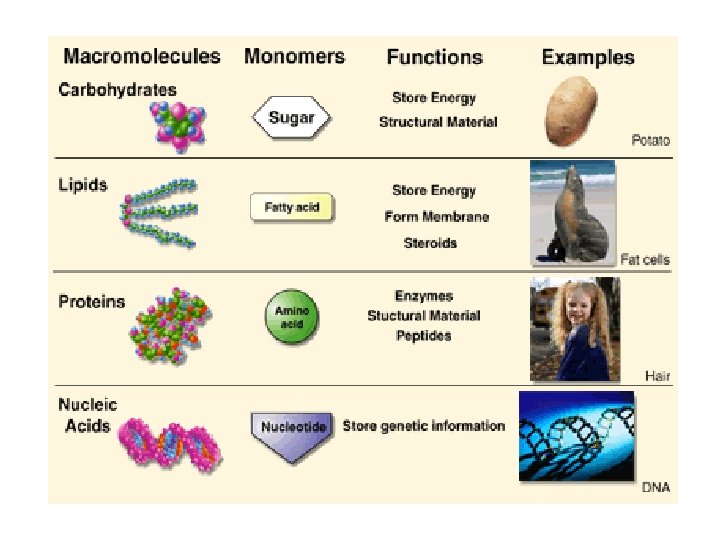

3. 3 What Are Carbohydrates? • Carbohydrates are molecules composed of carbon, hydrogen, and oxygen in the ratio of 1: 2: 1. • They can be small single sugar molecules or long chains of single sugar molecules strung together.

Monosaccharide: a carbohydrate consisting of one sugar molecule Disaccharide: two sugars linked together Polysaccharide: three or more sugars linked together

3. 3 What Are Carbohydrates? – A variety of simple sugars occurs in organisms. – Disaccharides store energy and are two simple sugars bonded together by a dehydration synthesis reaction, such as lactose (milk sugar) and sucrose (table sugar). – – Polysaccharides store energy for long periods of time or can be structural. Starch in plants and glycogen in animals are energy storage molecules. Chitin (lobster shells) and cellulose (plant call walls) are polysaccharides which are structural.

3. 4 What Are Lipids? • Molecular characteristics of lipids – Lipids are molecules with long regions composed almost entirely of carbon and hydrogen. – The nonpolar regions of carbon and hydrogen bonds make lipids hydrophobic and insoluble in water.

3. 4 What Are Lipids? • Lipid classification – Group 1: Oils, fats, and waxes (Triglycerides) – Group 2: Phospholipids – Group 3: Steroids
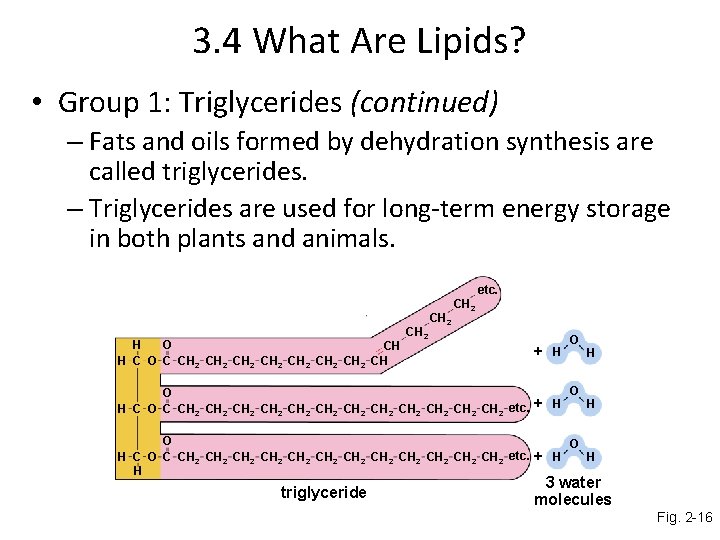
3. 4 What Are Lipids? • Group 1: Triglycerides (continued) – Fats and oils formed by dehydration synthesis are called triglycerides. – Triglycerides are used for long-term energy storage in both plants and animals. etc. H O CH H C O C CH 2 CH 2 CH 2 + H O H C O C CH 2 CH 2 CH 2 CH 2 CH 2 CH 2 etc. H + H triglyceride O O O H H H 3 water molecules Fig. 2 -16
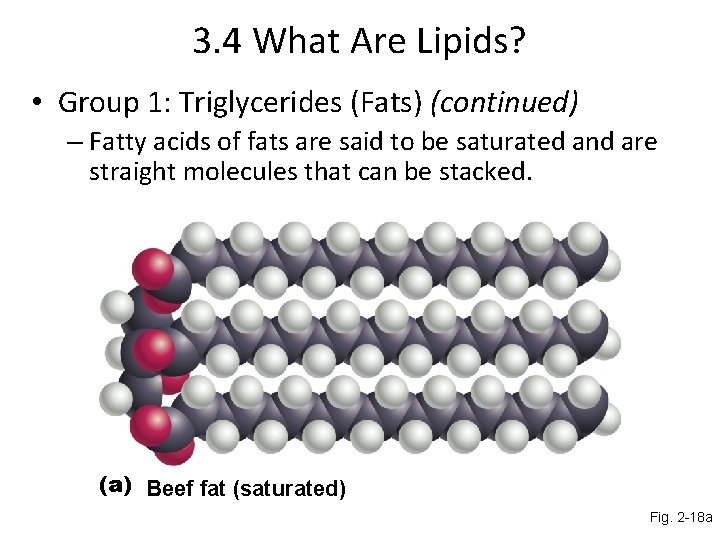
3. 4 What Are Lipids? • Group 1: Triglycerides (Fats) (continued) – Fatty acids of fats are said to be saturated and are straight molecules that can be stacked. (a) Beef fat (saturated) Fig. 2 -18 a
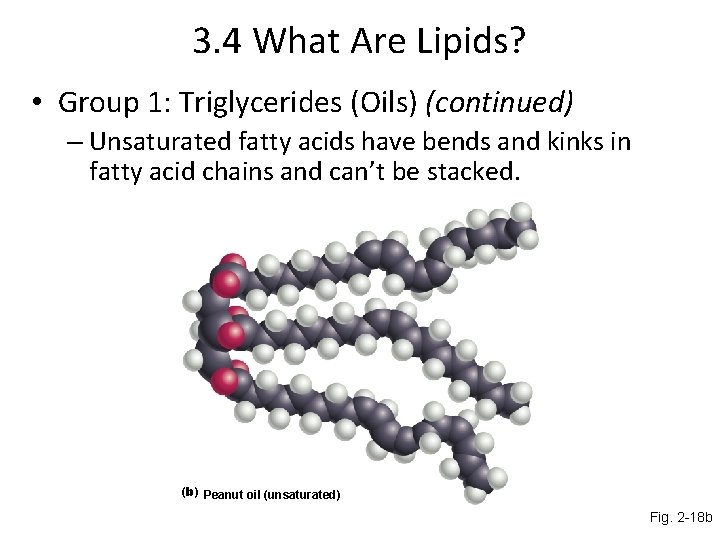
3. 4 What Are Lipids? • Group 1: Triglycerides (Oils) (continued) – Unsaturated fatty acids have bends and kinks in fatty acid chains and can’t be stacked. (b) Peanut oil (unsaturated) Fig. 2 -18 b

3. 4 What Are Lipids? • Group 2: Phospholipids – Phospholipids have water-soluble heads and waterinsoluble tails. – They are found in cell membranes in a double layer. – They are like oils except one fatty acid is replaced by a phosphate group attached to glycerol.

3. 4 What Are Lipids? • Group 3: Steroids – Steroids contain four carbon rings fused together. – Various functional groups protrude from the basic steroid “skeleton”. – Cholesterol is a steroid found in cell membranes.
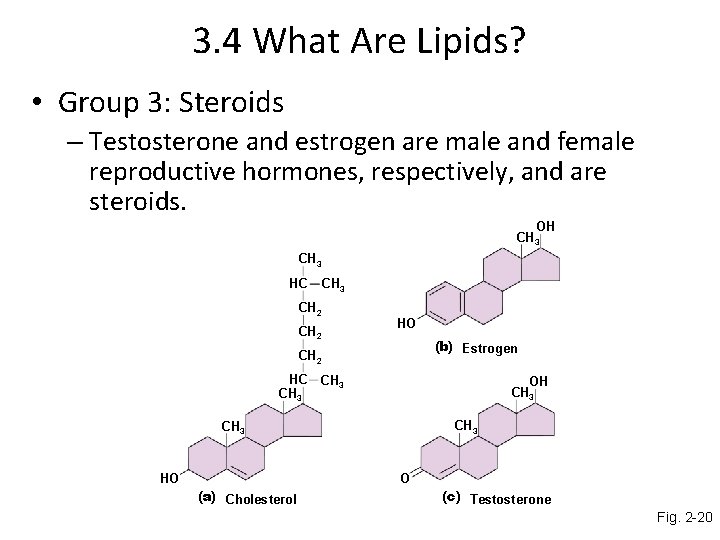
3. 4 What Are Lipids? • Group 3: Steroids – Testosterone and estrogen are male and female reproductive hormones, respectively, and are steroids. OH CH 3 HC CH 3 CH 2 HO (b) Estrogen CH 2 HC CH 3 OH CH 3 HO O (a) Cholesterol (c) Testosterone Fig. 2 -20

3. 5 What Are Proteins? • Functions of proteins – Proteins act as enzymes to catalyze many biochemical reactions. – They can act as energy stores. – They are involved in carrying oxygen around the body. – They are involved in muscle movement.
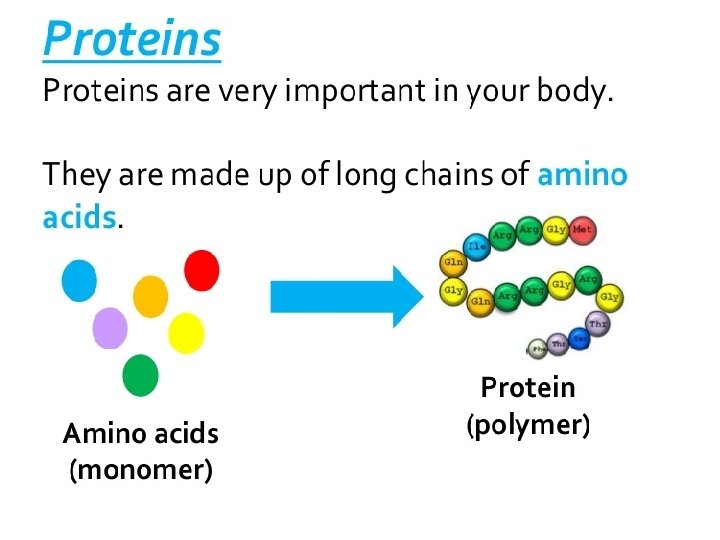

3. 5 What Are Proteins? • Proteins are formed from chains of amino acids. – All amino acids have the same basic structure: – – A central carbon An attached amino group An attached carboxyl group An attached variable side group

3. 5 What Are Proteins? • Some proteins are structural and provide support in hair, horns, spider webs, etc. Fig. 2 -21

Types of nucleotides Those that contain the sugar ribose. Those that contain the sugar deoxyribose. Nucleotides string together in long chains as nucleic acids with the phosphate group of one nucleotide bonded to the sugar group of another. DNA and RNA, the molecules of heredity, are nucleic acids. There are two types of nucleic acids. Deoxyribonucleic acid (DNA): contains the genetic code of cell Ribonucleic acid (RNA): is used in the synthesis of proteins

3. 6 What Are Nucleic Acids? • Structure of nucleic acids (continued) – All nucleotides have three parts. • A five-carbon sugar (ribose or deoxyribose) • A phosphate group • A nitrogen-containing molecule called a base
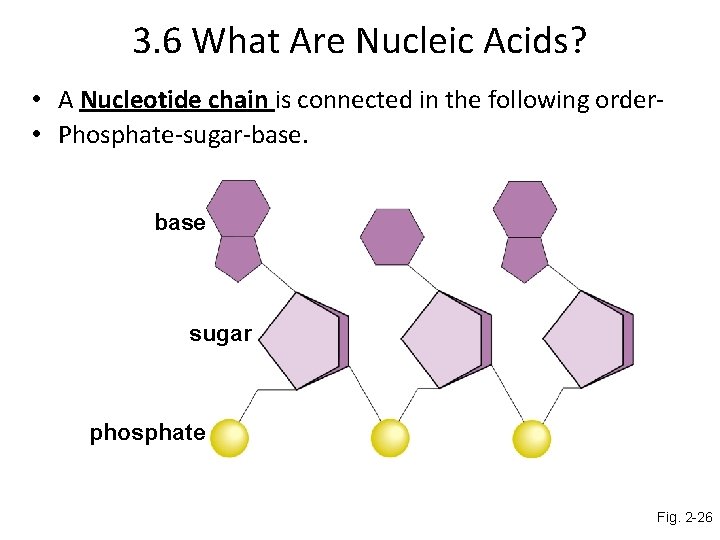
3. 6 What Are Nucleic Acids? • A Nucleotide chain is connected in the following order • Phosphate-sugar-base sugar phosphate Fig. 2 -26
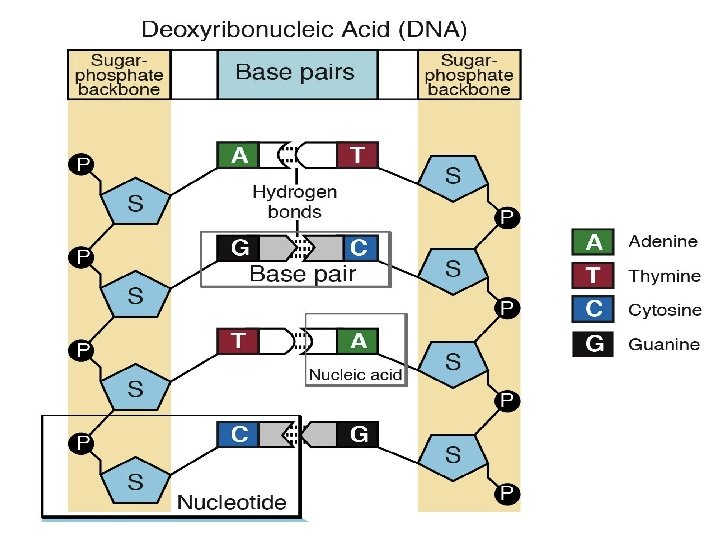

�Mutations are the original source of differences in DNA Variations are due to the differences in genes, which are components of DNA and the basic units of heredity Mutations occur when changes in genes are mistakenly copied

There are two kinds of cells. Prokaryotic cells q Are found only in two groups of singlecelled organisms—the bacteria and archaea Eukaryotic cells q Are structurally more complex cells Possess a membrane-enclosed nucleus Probably arose from prokaryotic cells

Prokaryotic cells • are much smaller than eukaryotic cells, are more abundant, and are very reproductively successful. • The are very small and have a simple internal structure. • They are surrounded by a stiff cell wall, which provides shape and protection. • Some move with a whiplike flagellum. • A single, circular strand of DNA is attached to the plasma membrane and is concentrated in an area called the nucleoid, which lacks a membrane. • There are no membrane-enclosed organelles. • The cytoplasm contains ribosomes used for protein synthesis.

Eukaryotic cells • differ from prokaryotic cells because the former possess a number of membrane-enclosed organelles that perform specific cell functions. o Nucleus: contains DNA o Mitochondria: produce energy o Endoplasmic reticulum: synthesizes membrane components and lipids o Golgi apparatus: molecule sorting center o Lysosomes: digest cellular membranes or defective organelles o Microtubules: make up the cytoskeleton

Membrane proteins v Transport proteins: allow the movement of water-soluble molecules through the plasma membrane by forming channels or by carrying them across v Receptor proteins: possess a binding site on the outer surface for binding specific chemicals that may alter overall cell function v Recognition proteins: with sugar groups attached to the exterior of the cell, are used by the immune system to identify cells as belonging to “self”

Diffusion is the movement of molecules down a gradient from high concentration to low concentration. • The greater the concentration gradient, the faster the rate of diffusion. • If no other processes intervene, diffusion will continue until the concentration gradient is eliminated.

Osmosis: the diffusion of water molecules from a high water concentration to a low water concentration across a biological membrane • Pure water has the highest water concentration. • The addition of dissolved solutes to pure water reduces the number of water molecules and thus lowers the water concentration.

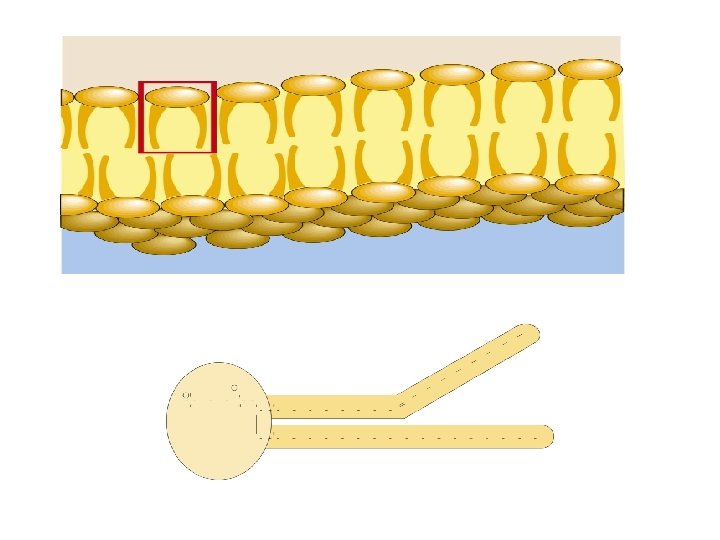
Plasma membranes are “fluid mosaics”. • Membranes are composed of a double lipid layer that is highly fluid without breaking. • Proteins are embedded in this double lipid layer and give the membrane its mosaic character. The phospholipid bilayer is the fluid portion of the membrane. • Phospholipid molecules have a polar head group and a pair of nonpolar tails Ø The head groups are hydrophilic and point toward the aqueous environment on both sides of the membranes. Ø The tail groups are hydrophobic and point away from the aqueous environments and toward each other
- Slides: 57