Biology 221 Anatomy Physiology II TOPIC 1 Circulatory

Biology 221 Anatomy & Physiology II TOPIC 1 Circulatory System – Blood Chapter 17 pp. 644 -674 E. Lathrop-Davis / E. Gorski / S. Kabrhel 1

Major Components of the Circulatory System • Blood • Heart • Blood vessels Fig. 19. 2, p. 715 Good websites: http: //www. neosoft. com/~uthman/blood_cells. html http: //www. fpnotebook. com/HEM. htm 2

Major Functions of the Circulatory System N • Transportation • Protection – against disease and toxins – against blood loss • Regulation – blood pressure – blood volume – body temperature 3

Physical Characteristics of Blood • • Specific gravity = 1. 045 -1. 065 Viscosity (relative to water) = 4. 5 -5. 5 p. H = 7. 35 – 7. 45 Volume = 7 -9% of body weight – 5 -6 L in adult males – 4 -5 L in adult females • Temperature ~ 100. 4 o. F (38 o. C) 4
Composition of Blood N • cells & cell fragments = “formed elements” – erythrocytes = RBCs (>95%) – leukocytes = WBCs – thrombocytes = platelets (cell fragments) • matrix (plasma) – ground substance (serum) – plasma proteins • Classify blood as one of the 4 major tissue types See Fig. 17. 1, p. 645; Table 17. 2, p. 656 5

N Plasma: Definition and Composition • Plasma = whole blood minus cells • Serum = plasma minus clotting proteins • Constituents (Table 17. 1, p. 647) – 92% water – 7% plasma proteins – 1% other solutes ° including electrolytes ° organic nutrients and wastes ° respiratory gases 6

Plasma Proteins Most made by liver • Albumins (~ 60% of plasma proteins) – exert osmotic force – buffer p. H • Globulins (~ 36%) – immunoglobulins (antibodies) protect against disease – transport proteins ° bind ions (e. g. , transferrin) and small molecules that would otherwise be lost ° bind fatty acids, thyroid and steroid hormones 7

Plasma Proteins (con’t) N • Fibrinogen and other clotting factors (~ 4% of all plasma proteins) • Other plasma proteins: – hormones (e. g. , insulin, glucagon) – enzymes (e. g. , renin) – antibacterial proteins (e. g. , complement) 8

Functions of Erythrocytes Main function: transport of respiratory gases • transports about 98. 5% of O 2 (oxyhemoglobin); about 23% of CO 2 (carbaminohemoglobin) • aids conversion of CO 2 to bicarbonate (HCO 3 -; covered with Topic 7: Respiratory System) http: //www. usc. edu/hsc/dental/ghisto/bld/d_5. html 9

Characteristics of Erythrocytes N • Small, biconcave disk • Anucleate, no ribosomes • No mitochondria – What type of ATP synthesis can they do? • Life span ~ 120 days (or less) http: //www. vh. org/adult/provider/pathology/CLIA/Hematology/14 Red Cell. html 10

Characteristics of Erythrocytes N • Diameter = 7 -8 micrometers (μm) • Mean corpuscular volume (MCV) – average volume of individual RBCs in sample – microcytic – macrocytic Fig. 17. 3, p. 648 11

Measuring RBC Abundance N • Normally, RBCs account for >95% of all formed elements • Red blood cell count (part of “CBC w/diff”) – males: 4. 5 -6. 3 x 106 / mm 3 (microliter) – females: 4. 2 -5. 5 x 106 / mm 3 – Polycythemia: 8 -11 x 106 / mm 3 ° renal hypoxia ° high altitude ° cancer (polycythemia vera) • Hematocrit = packed cell volume (PCV) 12

N Hematocrit (PCV) • measured by centrifuging tiny sample of blood • separates formed elements from plasma – >95% of formed elements are RBCs – “buffy coat” Fig. 17. 1, p. 645 13

Hematocrit (PCV) • Ratio of formed elements to whole sample expressed as percentage – males: average 45% (range: 40 -54%) – females: average 42% (range 37 -47%) – minimum hematocrit to donate blood = 38% • Blood doping – reinfusion of packed RBCs to increase hematocrit – Why would this be advantageous to athletes? Would this help all athletes? 14

Hemoglobin (Hb) N • Accounts for > 95% of protein in RBC • Main functions: transportation of respiratory gases – carries ~ 98. 5% of all O 2 – carries ~ 20 -30% of all CO 2 15

N Hemoglobin (Hb): Structure • Globular protein with quaternary structure: 2 alpha chains & 2 beta chains • Heme - non-protein, lipid-like molecule – porphyrin ring with iron center (binds oxygen) – 1 heme per protein chain – How many heme per Hb? How many O 2 can each Hb bind? Fig. 17. 4, p. 649 16

Hemoglobin (Hb) Content of Blood N • measured as g/dl (grams per deciliter, or per 100 ml) using hemoglobinometer • average values: – male: 14 -18 g/dl – female: 12 -16 g/dl – infants: 14 -20 g/dl 17

Hemoglobin (Hb) Content of Blood N • mean corpuscular Hb = average mass of Hb in one RBC • measured as hemoglobin concentration/number of RBCs – normochromic – hyperchromic 18

Locations of Erythrocyte Formation (Erythropoiesis) • 1 st 8 weeks of fetal development, RBCs formed in yolk sac • During 2 nd to 5 th months of fetal development, RBCs formed in liver (main supplier) and spleen • From 5 th month on, RBCs formed in bone marrow 20

Locations of Erythrocyte Formation (Erythropoiesis) • Post-natal development and in adults, RBCs formed in red bone marrow (myeloid tissue) – portions of: vertebrae, ribs, scapula, skull, pelvis, proximal heads of femur and humerus – yellow marrow of medullary cavities can be converted back into red marrow, if needed 21

N Stages of Erythropoiesis Formed from hemocytoblasts proerythroblasts – differentiated RBC precursors erythroblasts – synthesize Hb normoblasts (formed after about 4 days) – lose nucleus, some mitochondria Fig. 17. 5, p. 650 22

Stages of Erythropoiesis (con’t) N reticulocytes – contains ribosomes & mitochondria (but no nucleus) – leaves bone marrow after ~ 2 days – reticulocyte count: normally ~ 0. 8% (0. 82. 0%) of RBC population mature erythrocyte Fig. 17. 5, p. 650 23

Control of Erythropoiesis N Under influence of erythropoietin • secreted by kidney under hypoxic conditions – anemia – decreased blood flow to kidney – decreased oxygen availability (e. g. , increased elevation) • stimulates: – increased cell division of stem cells and erythroblasts – increased maturation by increasing rate of Hb synthesis • Is this control negative or positive feedback? Fig. 17. 6, p. 651 24

Other Factors Influencing Rate of Erythropoiesis • Hormones - androgens (testosterone) and growth hormone - stimulate erythropoiesis • Adequate diet – amino acids – vitamins (B 12, B 6, folic acid) ° pernicious anemia – lack of Vit. B 12 due to deficiency of intrinsic factor produced by gastric mucosa and needed for uptake of B 12 – iron (Fe) ° iron-deficiency anemia – lack of sufficient iron in diet, inability to absorb iron; secondary to hemorrhagic anemia 26

Erythrocyte Recycling N • 10% hemolyzed before degradation • 90% phagocytized by macrophages in spleen, liver, bone marrow – amino acids released into blood – heme broken into Fe and porphyrin ring ° Fe transported by incorporation into transferrin to – red bone marrow for reincorporation into Hb, or – liver or spleen for storage by incorporation into ferritin or hemosiderin See Fig. 17. 7, p. 652 27

N Erythrocyte Recycling (con’t) – porphyrin ring of heme converted to bilirubin (or other forms) ° excreted in bile and released in feces ° excreted in urine ° jaundice – deposition of bilirubin in skin due to failure to accumulation in blood (hyperbilirubinemia) – caused by: » liver dysfunction » obstruction of bile passageways » excessive rupture of RBCs (e. g. , neonatal jaundice or transfusion reaction) 28

N Blood Typing • Based on surface antigens • At least 50 kinds of proteins may be used • Most common – ABO blood group ° ° with A protein = Type A blood with B protein = Type B blood with neither protein = Type O blood with both proteins = Type AB blood – Rh factor (D) ° with D protein = Rh+ ° without D protein = Rh– Fig. 17. 15, p. 670 29
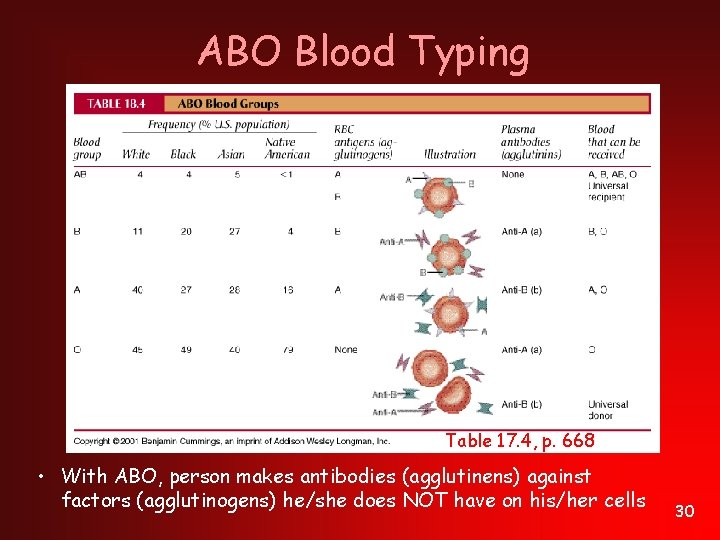
ABO Blood Typing Table 17. 4, p. 668 • With ABO, person makes antibodies (agglutinens) against factors (agglutinogens) he/she does NOT have on his/her cells 30
![ABO Blood Types Blood Type A B AB[1] O[2] Agglutinogens (antigen proteins) Present A ABO Blood Types Blood Type A B AB[1] O[2] Agglutinogens (antigen proteins) Present A](http://slidetodoc.com/presentation_image/18493b6a22e9846a0857f421db398dbc/image-29.jpg)
ABO Blood Types Blood Type A B AB[1] O[2] Agglutinogens (antigen proteins) Present A B A&B (neither) Makes Agglutinins (antibodies) Against B A (neither) A&B May Receive Blood From: A, O B, O A, B, AB, O O May Give Blood To: A, AB B, AB AB A, B, AB, O Genotype IAIA or IAi IBIB or IBi IA IB ii Rh Factor Present or Absent (A+ or A-) Present or Absent (B+ or B-) Present or Absent (AB+ or AB-) Present or Absent (O+ or O-) See also Table 17. 4, p. 668 [1] Universal Recipient [2] Universal Donor 31

Rh Blood Types Blood Type Rh+ Rh- Agglutinogen D (antigen proteins) Present or Absent Present Absent Makes Agglutinins (antibodies) Against Agglutinogen No Yes[1] May Receive Blood From: Rh+ or Rh-[2] May Give Blood To Without Reaction[2]: Rh+ or Rh‑ Genotype DD or Dd dd [1] Only makes antibodies (agglutinens) after exposure to Rh+ blood cells (via transfusion or during birth process) [2] Transfusion of Rh- individual with Rh+ blood results in production of anti-D agglutinens; sensitizes person to Rh factor and may result in anaphylaxis if exposed a second time. Erythroblastosis fetalis arises when Rh- mother has been exposed to Rh+ blood and is carrying Rh+ child. 32

Blood Typing N • Cross reactions – caused by giving blood type to which recipient has antibodies – cause agglutination (clumping) • Erythroblastosis fetalis – occurs when Rhmother who has been exposed to Rh+ blood is carrying Rh+ fetus antibodies from mother cross placenta and attack fetal blood cells – Rho. GAM® agglutinates anti-Rh antibodies 33

Check These Blood Typing Links • http: //sln. fi. edu/biosci/blood/types. html • http: //www. biology. arizona. edu/human_bio/ problem_sets/blood_types/Intro. html 34

RBC & Associated Disorders • Thalassemia – genetic inability to produce adequate amounts of alpha or beta chains – results in limited production of fragile, short-lived RBCs often with odd shapes http: //www. bloodline. net/stories/story. Reader$2344 35

RBC & Associated Disorders • Sickle-cell anemia – genetic mutation in which 7 th amino acid in beta chain is changed – causes Hb. S molecules to stick when oxygen is not bound leading to characteristic sickle shape of RBCs http: //hematology. lastmaldives. com/Sicklecellanemia. htm 36

N RBC & Associated Disorders • Anemias – hemorrhagic anemia – heavy bleeding (RBCs are normal but fewer than normal in number) – iron-deficiency anemia – lack of iron in diet (RBCs are hypochromic and microcytic); may be secondary to hemorrhagic anemia – pernicious anemia – lack of Vit. B 12 due to lack of intrinsic factor (RBCs are macrocytic) 37

N RBC & Associated Disorders • Hemolytic anemia – RBCs break faster than they can be replaced – Causes include transfusion reactions, hemolytic anemia, severe burns, reactions to certain toxins • Hemoglobinuria - Hb in urine 38

Leukocytes: Functions N • Fight pathogens and provide both innate and adaptive immunity (see Topic 6) – pathogens = disease-causing agents ° “microbes” include: bacteria, fungi (yeasts and molds), viruses, protozoa, algae • Clear debris from damaged areas • Fight cancer cells 39

Leukocyte Abundance N • Normal = 4, 800 -10, 800 cells / mm 3 – Leukopenia < 4, 800 cells / mm 3 ° Response to some drugs and some autoimmune disorders – Leukocytosis > 11, 000 cells / mm 3 ° normal with disease ° > 100, 000 WBCs / mm 3 not uncommon with leukemia • Measured by: (as part of a “CBC w/diff”) – White blood cell count – Differential WBC Count 40

Leukocyte Abundance: Differential WBC Count • relative abundance of different kinds of WBCs • count number of each different type in a total of 100 WBCs Fig. 17. 9, p. 655 41

N Granulocytes: Neutrophils • • 50 -70% (Marieb, 6 th Ed. ) Phagocytic, especially against bacteria Large number of lysosomes in cytoplasm Highly mobile 10 -12 µm in diameter Short life spans (~ 10 hrs; less if highly active) Neutrophilia – associated with acute bacterial infections http: //www. usc. edu/hsc/dental/ghisto/bld/d_1. html 43

Granulocytes: Eosinophils N • 2 -4% (Marieb, 6 th ed. ) • 10 -14 µm in diameter • Phagocytize antibody-covered objects (bacteria, cellular debris, parasitic worms and protozoa); release cytotoxic enzymes onto target parasites • Lessen severity of allergic reactions by • Eosinophilia –associated with parasitic worm infections http: //www. usc. edu/hsc/dental/ghisto/bld/d_3. html 44

Granulocytes: Basophils • < 1% • 8 -10 µm in diameter (Marieb, 6 th ed. ) • Accumulate in damaged tissues where they release histamine and heparin • Associated with chronic inflammatory diseases • Basophilia – increase in number of basophils associated with allergic reactions http: //www. usc. edu/hsc/dental/ghisto/bld/d_4. html 45

Agranulocytes: Lymphocytes N • • 25 -45% (Marieb, 6 th ed. ) 5 -17 µm in diameter Most remain in lymphatic tissue Increase associated with several types of infections, especially viral • Circulating lymphocytes (see Topic 6) – T cells – B cells – Natural killer cells http: //www. usc. edu/hsc/dental/ghisto/bld/d_5. html 46

Agranulocytes: Monocytes • • 3 -8% (Marieb, 6 th ed. ) 18 µm in diameter Some become fixed macrophages within tissues Phagocytize viruses, debris, bacteria; enhance scar tissue formation http: //www. usc. edu/hsc/dental/ghisto/bld/d_6. html 47

Agranulocytes: Infectious Mononucleosis • Highly contagious viral disease • Symptoms include large numbers of atypical agranulocytes, fatigue, soreness, chronic sore throat, low-grade fever http: //image. bloodline. net/stories/story. Reader$680 http: //www. wadsworth. org/chemheme/microscope/imono. htm 48

Leukocyte Production Arise from hemocytoblasts lymphoid stem cells lymphoblasts prolymphocytes Fig. 17. 11, p. 659 49

Leukocyte Production Arise from hemocytoblasts myeloid stem cells monoblast promonocyte or myeloblast differentiated myelocytes various band cells various granulocytes Fig. 17. 11, p. 659 50

Regulation of Leukocyte Production • Cytokines (molecules released by one cell that affects the growth or activity of another) – Colony stimulating factors (CSFs) ° stimulate production and development of WBCs ° named according to type(s) of cells stimulated – Interleukins ° released by WBCs to affect activity of other WBCs; most important to lymphocyte production • Thymic hormones promote differentiation and maintenance of T cells • Presence of antigens stimulates lymphocyte production (see Topic 6) 51

Leukemias • Cancer of WBC producing cells • Named according to type of cell(s) involved – e. g. , myelocytic leukemia • Acute leukemia – comes from “-blast” cells (i. e. , myeloblasts, monoblasts, lymphoblasts) – occurs more often in children • Chronic leukemia – comes from later stages (e. g. , myelocytes, promonocytes, prolymphocytes) – more common in elderly 52

Platelets • Small (2 -4 µm in diameter), anucleate cell fragments • Short-lived (5 -10 days) • 150, 000 – 400, 000 platelets / mm 3 of plasma – Thrombocytopenia < 80, 000/mm 3 ° excess platelet destruction ° inadequate production ° symptoms include bleeding in digestive tract, skin, CNS – Thrombocytosis > 500, 000 / mm 3 ° infection ° inflammation ° cancer 53

Platelet Functions N • Hemostasis – platelet plug formation – enhance clotting – clot retraction 54
N Platelet Formation and Regulation • Formation: – hemocytoblasts megakaryocyte platelet • Regulation – thrombopoietin (TPO or thrombocytestimulating factor) from kidneys – multi-CSF Fig. 17. 12, p. 660 55

Hemostasis • Stoppage of bleeding • 3 Phases (Mechanisms): – Vascular phase – Platelet phase (platelet plug formation) – Coagulation (clotting ) 56

Vascular Phase • Vascular spasm – contraction of smooth muscle of vessel wall • Endothelial cells – contract to pull vessel walls closer together – release chemical factors and local hormones stimulates vascular spasm & division of endothelial cells, smooth muscle cells and fibroblasts – in capillaries, endothelial cells on opposite sides become sticky and adhere to each other to close vessel 57

Platelet Phase – Platelet Plug Formation Stages: • Platelet adhesion – platelets stick to exposed collagen fibers in broken vessel – Aided by von Willebrand factor (VWF) from endothelial cells • Platelet aggregation – activated platelets change shape & develop processes to reach out to other platelets 58

Platelet Phase – Platelet Plug Formation N Activated platelets release chemicals that enhance hemostasis: – ADP (stimulates platelet activation) – thromboxane A 2 & serotonin (stimulate vascular spasm) – protein clotting factors (important to clotting) – platelet-derived growth factor (stimulates vessel repair) – calcium ions (non-protein clotting factor) 59

N Natural Limits to Platelet Plug Formation • Prostacyclin (PGI 2; local prostaglandin) inhibits platelet aggregation • Inhibiting compounds secreted by WBCs • Clotting (isolates platelet plug from circulation) 60

N Coagulation (Clotting) Phase • Series of reactions resulting in formation of insoluble fibrin fibers • Reactions occur as cascades resulting in large amount of fibrin formed from small amount of initial reactants • Positive feedback loop in which thrombin stimulates formation of tissue factor and release of PF-3 from platelets • Two initial pathways that share a common pathway at the end; differ in starting point and stimulus 61

Coagulation Requirements N • clotting factors (procoagulants) – protein enzymes – synthesis of 4 factors by liver requires vitamin K • fibrinogen • Ca 2+ ions 62

Measuring Coagulation N • Partial thromboplastin time (PTT) – Tests intrinsic and common pathways – 30– 45 seconds • Prothrombin Time – Tests extrinsic and common pathways – 15 -35 seconds • Bleeding time – Time for small puncture wound to stop bleeding (in vivo) – 1 -9 minutes 63

N Pathways of Coagulation • Extrinsic pathway – starts with tissue factor (factor III) – fewer steps • Intrinsic pathway – starts with activation of proenzymes in blood • Common pathway – from prothrombin activator formation to formation of fibrin from fibrinogen Fig. 17. 13 a and b, p. 663 64

N Clot Retraction • accomplished by platelets that adhere to fibrin fibers • contraction of platelets pulls torn edges of vessel together • reduces size of damaged area 65

Fibrinolysis N Breakdown of fibrin fibers by plasmin • plasmin is formed from inactive precursor called plasminogen • plasminogen is activated by: – thrombin and activated factor XII produced by common pathway of clotting – tissue plasminogen activator (TPA) produced by damaged tissues 66

N Natural Control of Clotting restricted by: • dilution of procoagulants • plasma anticoagulants (e. g. , antithrombin III produced by platelets) that inactivate thrombin • heparin (released by basophils and mast cells; accelerates activity of antithrombin III) 67

Clinical Control of Clotting N • Heparin interferes with conversion of prothrombin to thrombin; enhances action of antithrombin III • Warfarin (Coumadin) interferes with production of clotting factors that require vit. K for synthesis • Aspirin – interferes with platelet aggregation 68

N Bleeding Disorders • Hemophilia – include recessive, X-linked genetic (more common) and autosomal diseases – clotting factors (most often factor VIII) are not made in adequate amounts • von Willebrand disease – common genetic bleeding disorder (autosomal dominant) – failure to make adequate amounts of von Willebrand’s factor, which stabilizes factor VIII and stimulates platelet adhesion 69

Bleeding Disorders N • Thrombus – clot formed in intact vessel wall; often occurs where cholesterol plaques are present; may break free or completely block vessel • Embolus – abnormal mass (especially a clot) in blood – may result in embolism (blockage of vessel) and cause infarct (tissue damage) ° stroke ° myocardial infarct ° pulmonary emboli 70

N Bleeding Disorders • Disseminated Intravascular Coagulation – a pathological reaction to sepsis, massive trauma, transfusion reactions, abruptio placentae, certain toxins (e. g. , certain snake venoms), ebola – clotting is widespread resulting in overuse of platelets and procoagulants – leads to tissue damage due to blockage of small vessels by fibrin deposition – leads to abnormal bleeding due to decrease in available 71

Genetics • Recessive traits - only seen when individual is homozygous for the allele (or in male if trait is X-linked) • Dominant traits – seen when individual has at least 1 copy of the allele • Autosomal traits – caused by genes on 22 chromosomes other than X or Y • Homozygous – has two of same allele for a gene • Heterozygous – has two different alleles for gene 72

Genetics • X-linked traits - caused by genes on X chromosome – female has 2 X chromosomes so can be heterozygous (2 different alleles) ° when trait is abnormality, heterozygous female appears normal but is said to be carrier because she can pass on abnormality ° mother and father must have allele for abnormality for daughter to show trait – male has 1 X chromosome so X-linked traits are seen 73
- Slides: 70