BIOLOGY 2 E Module 5 METABOLISM Power Point

BIOLOGY 2 E Module 5 METABOLISM Power. Point Image Slideshow This work is licensed under a Creative Commons Attribution. Non. Commercial-Share. Alike 4. 0 International License.
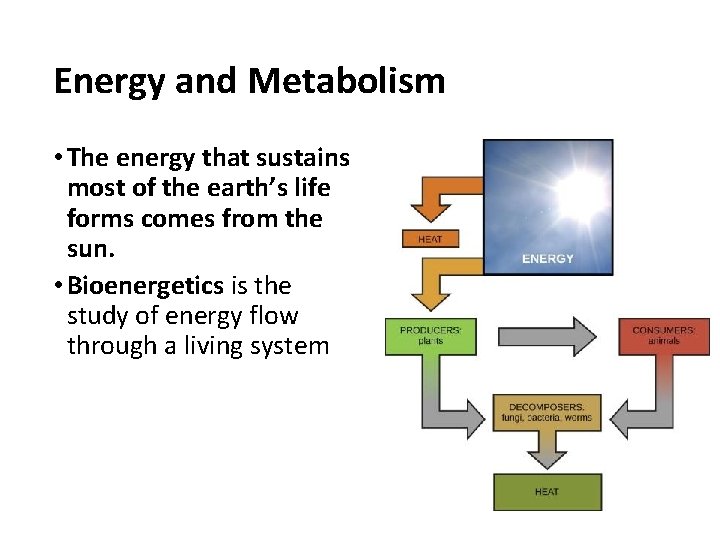
Energy and Metabolism • The energy that sustains most of the earth’s life forms comes from the sun. • Bioenergetics is the study of energy flow through a living system

Metabolism • Metabolism refers to all chemical reactions of a cell or organism. • A metabolic pathway is series of biochemical reactions that converts one or more substrates into a final product. • For example, energy from the sun is captured during photosynthesis. The captured energy is used to convert CO 2 and H 2 O into sugars, including glucose (C 6 H 12 O 6). • The chemical energy stored in glucose is released during cellular respiration, regenerating CO 2 and H 2 O. (We will discuss in subsequent lectures. )

Metabolism • Metabolism refers to all chemical reactions of a cell or organism. • A metabolic pathway is series of biochemical reactions that converts one or more substrates into a final product. Enzyme 1 A Enzyme 3 D C B Reaction 1 Starting molecule Enzyme 2 Reaction 3 Product http: //glencoe. mcgrawhill. com/sites/9834092339/student_view 0/chapter 6/a_biochemical_pathway. html
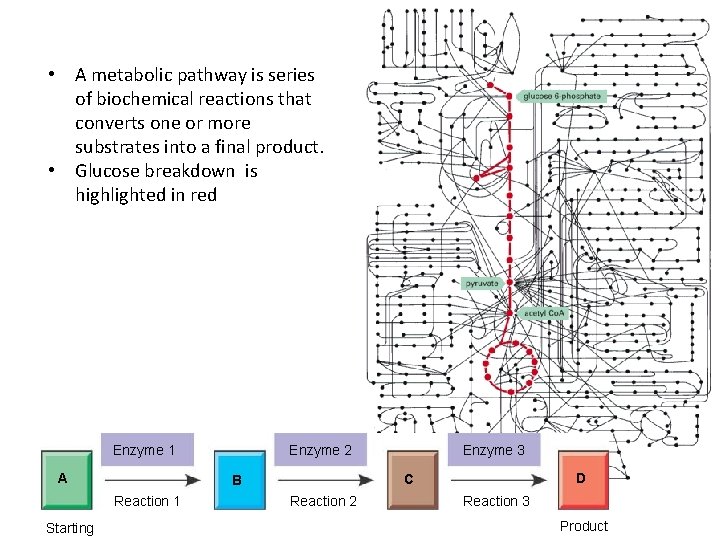
• A metabolic pathway is series of biochemical reactions that converts one or more substrates into a final product. • Glucose breakdown is highlighted in red Enzyme 1 A Enzyme 3 D C B Reaction 1 Starting Enzyme 2 Reaction 3 Product

METABOLIC PATHWAYS • Two types of reactions/pathways are required to maintain the cell’s energy balance. • Those that require energy and synthesize larger molecules are called anabolic. • Those that release energy and break down large molecules into smaller molecules are called catabolic.

Catabolic pathways and anabolic pathways Catabolic (breakdown; releases energy) Anabolic (building): stores energy
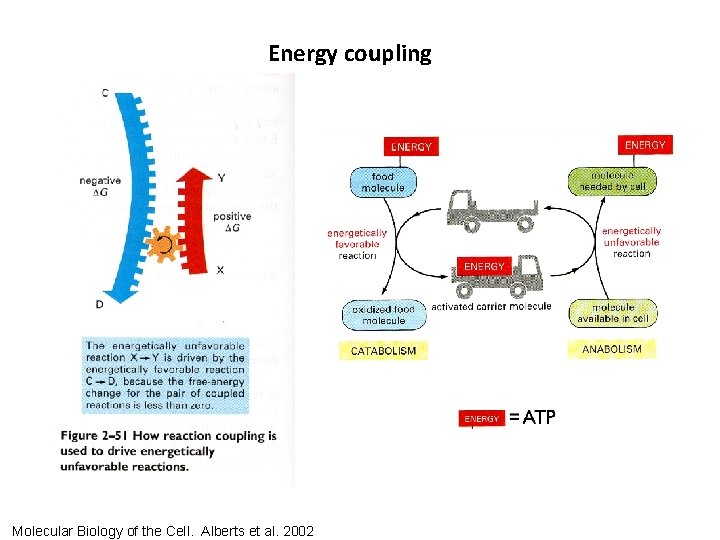
Energy coupling Molecular Biology of the Cell. Alberts et al. 2002
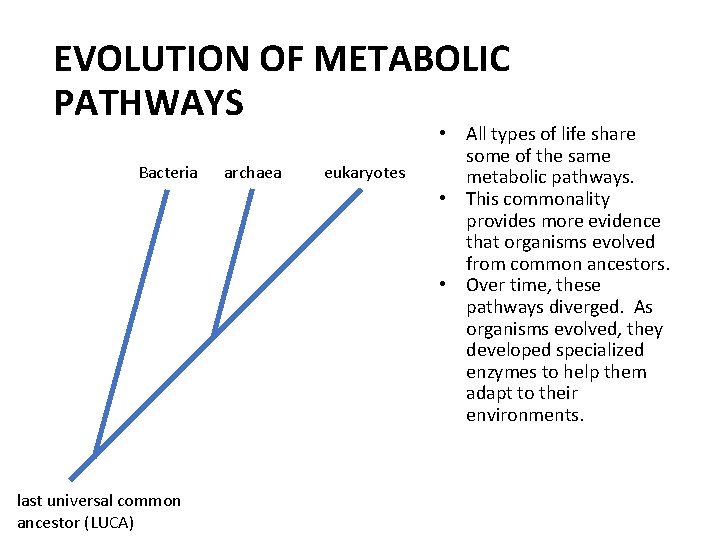
EVOLUTION OF METABOLIC PATHWAYS Bacteria last universal common ancestor (LUCA) archaea eukaryotes • All types of life share some of the same metabolic pathways. • This commonality provides more evidence that organisms evolved from common ancestors. • Over time, these pathways diverged. As organisms evolved, they developed specialized enzymes to help them adapt to their environments.

Types of energy • Energy is the ability to do work. • Energy can be classified as kinetic or potential • • Objects in motion have kinetic energy Objects that have the potential to move or change form in a way that powers work have potential energy (there is a lot of potential energy in water behind a dam, or a high concentration of a molecule on one side of a membrane)

Types of energy Potential Energy in cells • Chemical gradient and electrochemical gradients across a membrane • Chemical energy – Energy stored in chemical bonds

Free energy • To explore the bioenergetics of a system, we study the amount of energy exchanged in a metabolic reaction • Gibb’s Free Energy (G) = amount of energy available to do work (usable energy) out of the total energy of the system • All chemical reactions affect G; change in G after a reaction is abbreviated as ∆G (in science, ∆ means “change in”) ΔG = Gfinal state − Gstarting state ΔG = ΔH − TΔS ΔH is change in total energy of the system T is Temperature in Kelvin degrees ΔS is change in entropy (energy lost to entropy)

Free energy: exergonic reactions • If energy is released in a chemical reaction, then ΔG<0 (negative). • Products of these reactions will have less free energy than the substrates (starting materials) • These reactions are classified as exergonic • Exergonic reactions are spontaneous reactions because they occur without the addition of energy. • For a process to be spontaneous, it must either give off energy (decrease in H), give up order (increase in S), or both. • However, spontaneous reactions do not necessarily occur quickly! • The hump shown in the free energy diagram is the reason.

Free energy: endergonic reactions • If a chemical reaction requires an input of energy, then ΔG>0 (energy is being gained by the system). • Products of these reactions will have more free energy than the substrates. • These reactions are classified as endergonic. They are NOT spontaneous.

Activation energy • Activation energy is the energy required for a reaction to proceed (the “hump” in the diagram). • It causes reactant(s) to become contorted and unstable, which allows the bond(s) to be broken or made. • This unstable state is called the transition state. • Once in the transition state, the reaction occurs very quickly. • Heat energy is the main source for activation energy in a cell • Heat helps reactants reach their transition state
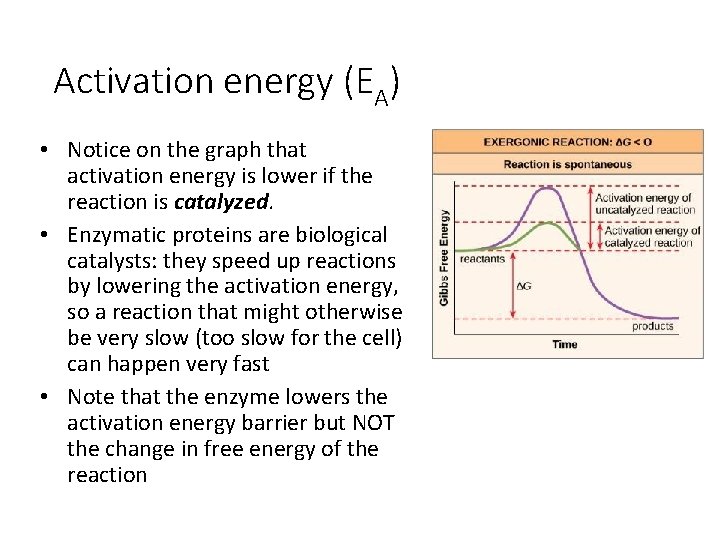
Activation energy (EA) • Notice on the graph that activation energy is lower if the reaction is catalyzed. • Enzymatic proteins are biological catalysts: they speed up reactions by lowering the activation energy, so a reaction that might otherwise be very slow (too slow for the cell) can happen very fast • Note that the enzyme lowers the activation energy barrier but NOT the change in free energy of the reaction

(a) Exergonic reaction: energy released, spontaneous Reactants Free energy Amount of energy released (DG < 0) Energy Products Progress of the reaction (b) Endergonic reaction: energy required, nonspontaneous Free energy Products Energy Reactants Progress of the reaction Amount of energy required (DG > 0)

Exergonic reaction example • The breakdown of gasoline is an example of an exergonic reaction. • A spark is required to provide sufficient heat to overcome the activation energy barrier. • Once the reaction begins, enough heat is released to drive additional reactions.

The Laws of Thermodynamics is the study of energy and energy transfer involving physical matter. a) The First Law of Thermodynamics states that the total amount of energy in the universe is constant: energy cannot be created or destroyed, only transferred or transformed. b) The Second Law of Thermodynamics states that the transfer or transformation of energy is not completely efficient. With each chemical reaction, some energy is lost in a form that is unusable, such as heat energy. The result is increased entropy: entropy is the logarithm of the multiplicity of states, or the degree of dispersion of energy in a system. Less precisely entropy is sometimes stated as a measure of randomness (i. e. , disorder). The second law of thermodynamics says that energy of all kinds tends to disperse or spread out if it is not hindered from doing so. Entropy is the quantitative measure of that kind of spontaneous process: how much energy has flowed from being localized to becoming more widely spread out.

On the ground a climber has less potential energy. Climbing up converts kinetic energy of muscle movement to potential energy. On the platform, the climber has more potential energy. . Leaping off the platform converts potential energy to kinetic energy. http: //mattkkelley. wordpress. com/2008/05/29/calypso/

Laws Of Thermodynamics • These kids convert the chemical energy from the ice cream cone to kinetic energy of riding a bike. Some heat energy is released, too. • Heat energy is also lost when plants use sunlight during photosynthesis to produce sugars. • No transfer or transformation of energy is completely efficient. • (credit “ice cream”: modification of work by D. Sharon Pruitt; credit “kids on bikes”: modification of work by Michelle Riggen-Ransom)

ATP: Adenosine triphosphate • What provides the energy for a cell’s endergonic (energy-requiring) reactions? An endergonic reaction is not spontaneous. For example, anabolic reactions like building a polymer don’t just happen, they require energy.

Energy coupling ATP hydrolysis endergonic process being driven by ATP hydrolysis The hydrolysis of ATP releases energy that can be used to drive (make energetically favorable) what would otherwise be an endergonic processes. Molecular Biology of the Cell. Alberts et al. 2002
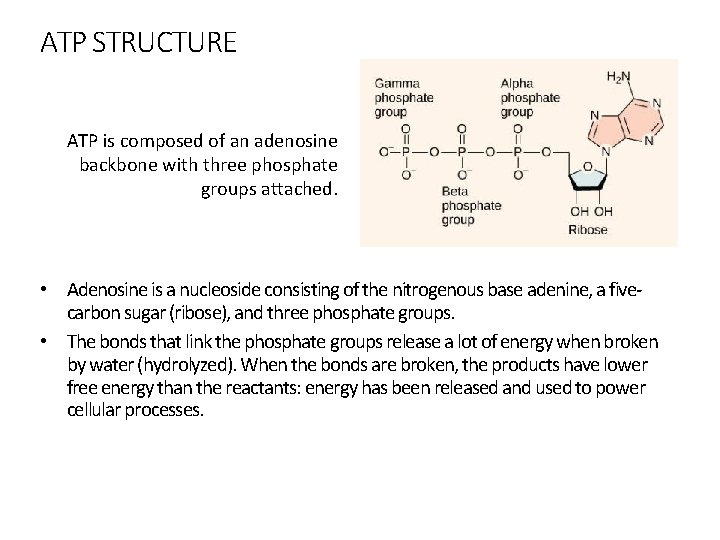
ATP STRUCTURE ATP is composed of an adenosine backbone with three phosphate groups attached. • Adenosine is a nucleoside consisting of the nitrogenous base adenine, a fivecarbon sugar (ribose), and three phosphate groups. • The bonds that link the phosphate groups release a lot of energy when broken by water (hydrolyzed). When the bonds are broken, the products have lower free energy than the reactants: energy has been released and used to power cellular processes.

ATP hydrolysis ATP + H 2 O ADP + Pi + free energy • ΔG = -7. 3 kcal/mole (it’s negative because energy is released) • ATP is an unstable molecule and will hydrolyze quickly. • If it is not coupled with an endergonic reaction this energy is lost as heat. • If it is coupled with an endergonic reaction, much of the energy can be transferred to drive that reaction. • ATP hydrolysis is reversible: a phosphate group can be added to ADP to make ATP again, storing energy derived from food. • The process of cellular respiration achieves this.

The sodium-potassium pump • The sodium-potassium pump (involving an integral transmembrane channel protein) is an example of energy coupling. • The energy derived from exergonic ATP hydrolysis is used by the integral protein to pump 3 sodium ions out of the cell and 2 potassium ions into the cell.

What you need to know • Explain what metabolic pathways are and describe the two major types of metabolic pathways • Discuss how chemical reactions play a role in energy transfer • Define “energy” • Explain the difference between kinetic and potential energy • Discuss the concepts of free energy and activation energy • Describe endergonic and exergonic reactions • Discuss the concept of entropy • Explain the first and second laws of thermodynamics • Explain the role of ATP as the cellular energy currency • Describe how energy is released through hydrolysis of ATP • Describe the role of enzymes in metabolic pathways • Explain how enzymes function as molecular catalysts • Discuss enzyme regulation by various factors
- Slides: 27