Biologically Important Molecules I Glycine an amino acid
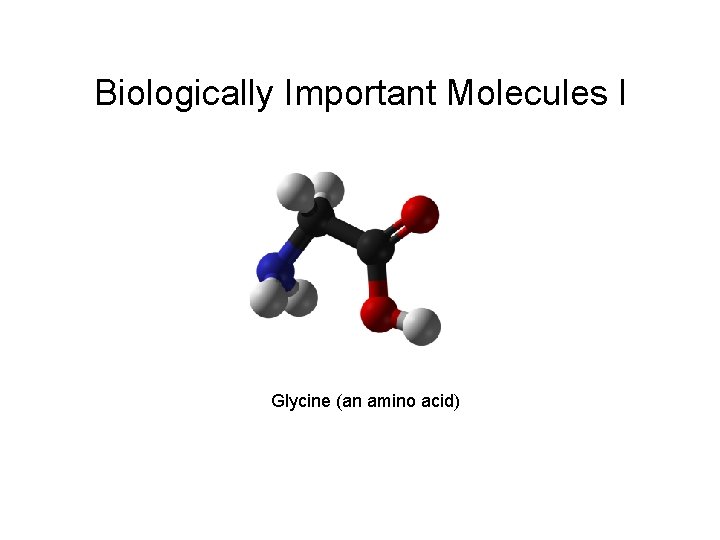
Biologically Important Molecules I Glycine (an amino acid)

I. Water A. Structure – did it B. Properties - ‘Universal’ solvent (what types of compounds? ) High specific heat (thermally stable) High heat of vaporization Adhesive/cohesive

I. Water C. Water and Life on Earth is inconceivable without water. Life requires rapid and continuous chemical reactions facilitated by a dissolution of reactants in a liquid solvent. Water’s solvent properties are ideal. Water is a liquid over a wide temperature range that is very common on Earth. (High specific heat, vaporization). Water is abundant on Earth, covering over 70% of the surface. Water is a thermally stable internal/external environment. No surprize that life probably originated in water (3. 8 bya), and did not adapt to exploit the desiccating terrestrial environments until the last ~10% of Life’s history (0. 4 bya).

Biologically Important Molecules I. Water II. Carbohydrates
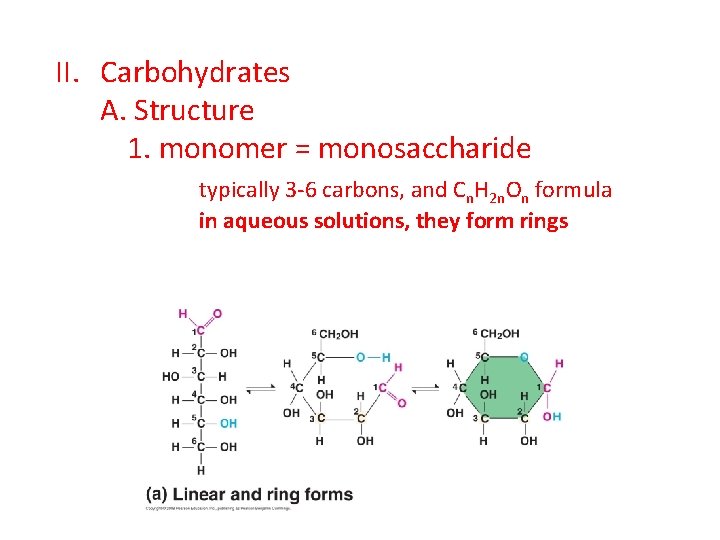
II. Carbohydrates A. Structure 1. monomer = monosaccharide typically 3 -6 carbons, and Cn. H 2 n. On formula in aqueous solutions, they form rings
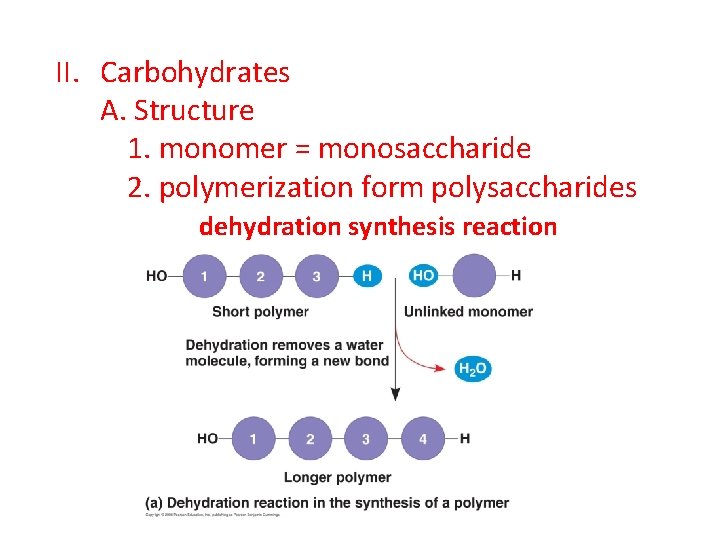
II. Carbohydrates A. Structure 1. monomer = monosaccharide 2. polymerization form polysaccharides dehydration synthesis reaction
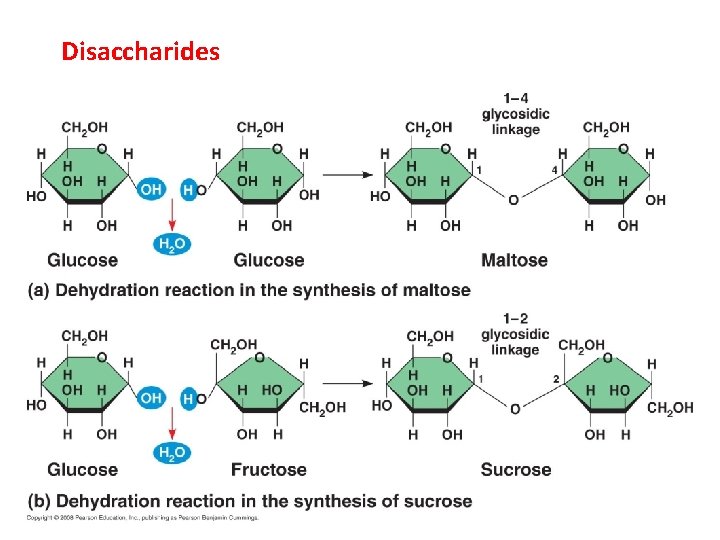
Disaccharides
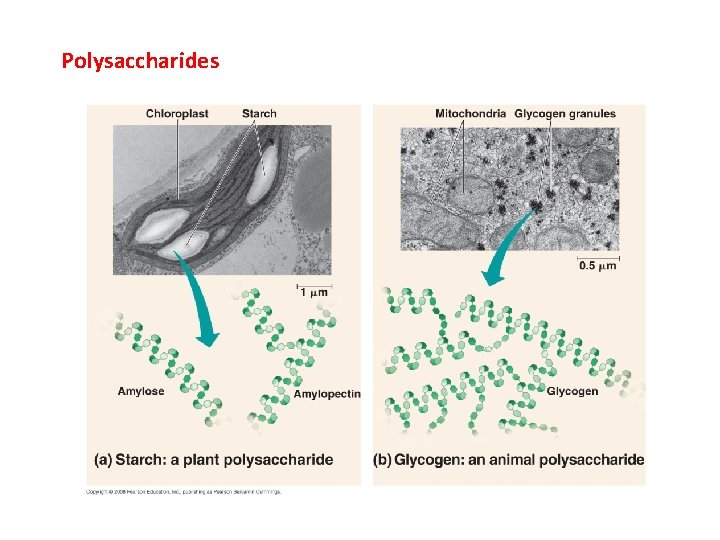
Polysaccharides
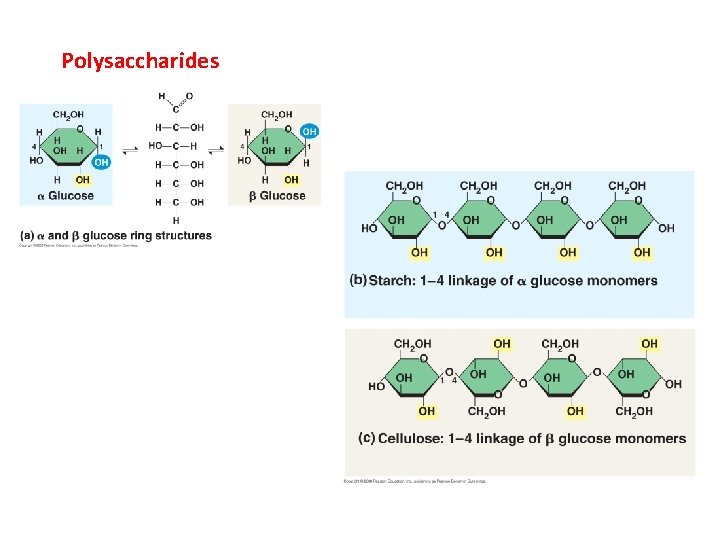
Polysaccharides
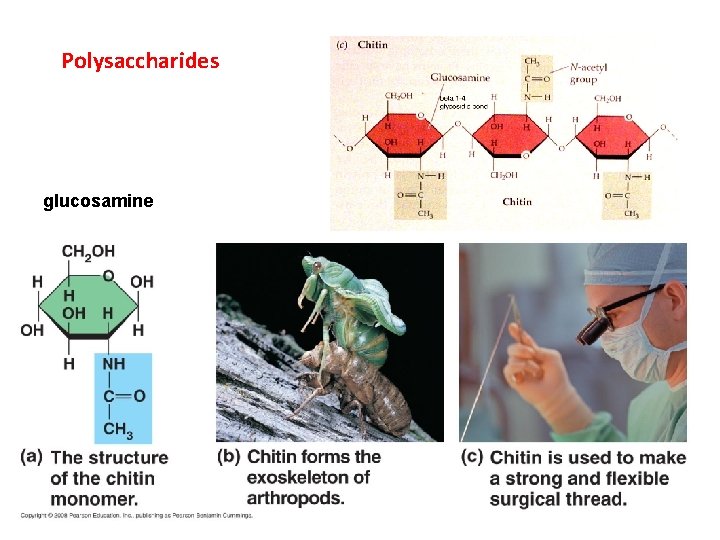
Polysaccharides glucosamine
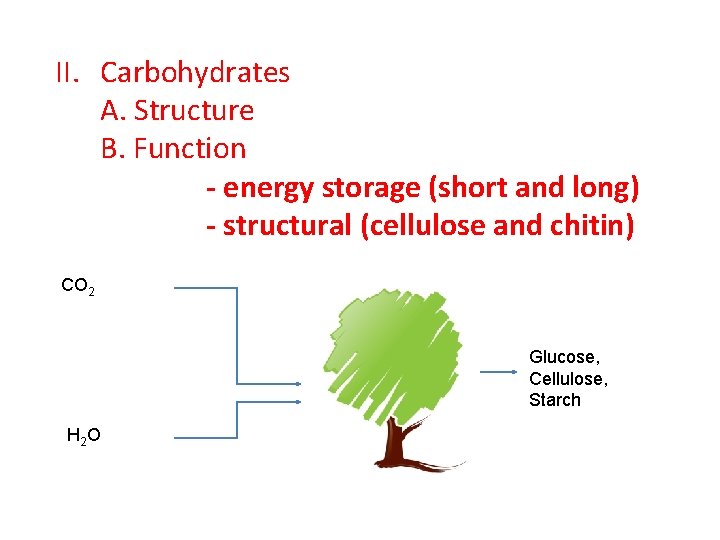
II. Carbohydrates A. Structure B. Function - energy storage (short and long) - structural (cellulose and chitin) CO 2 Glucose, Cellulose, Starch H 2 O
Biologically Important Molecules I. Water II. Carbohydrates III. Lipids

III. Lipids - not true polymers; an assortment of hydrophobic, hydrocarbon molecules classes as fats, phospholipids, waxes, or steroids.
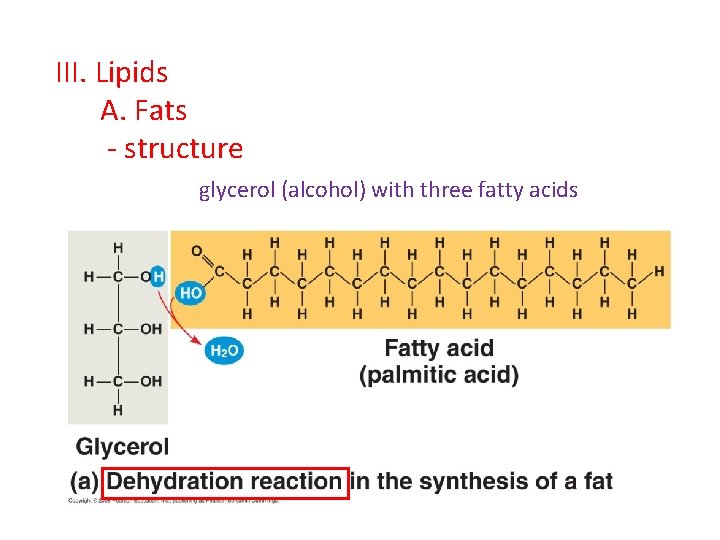
III. Lipids A. Fats - structure glycerol (alcohol) with three fatty acids
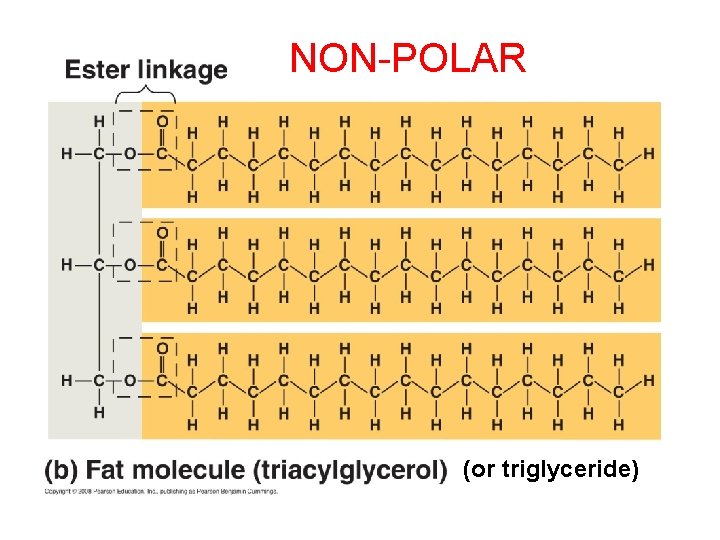
NON-POLAR (or triglyceride)
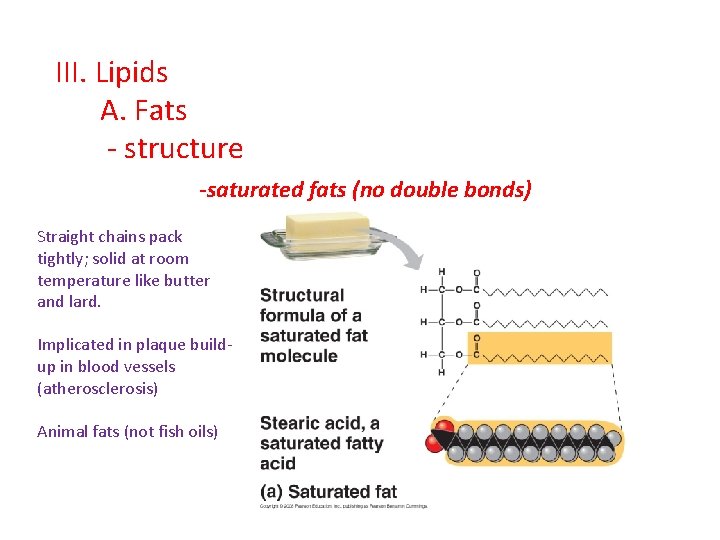
III. Lipids A. Fats - structure -saturated fats (no double bonds) Straight chains pack tightly; solid at room temperature like butter and lard. Implicated in plaque buildup in blood vessels (atherosclerosis) Animal fats (not fish oils)
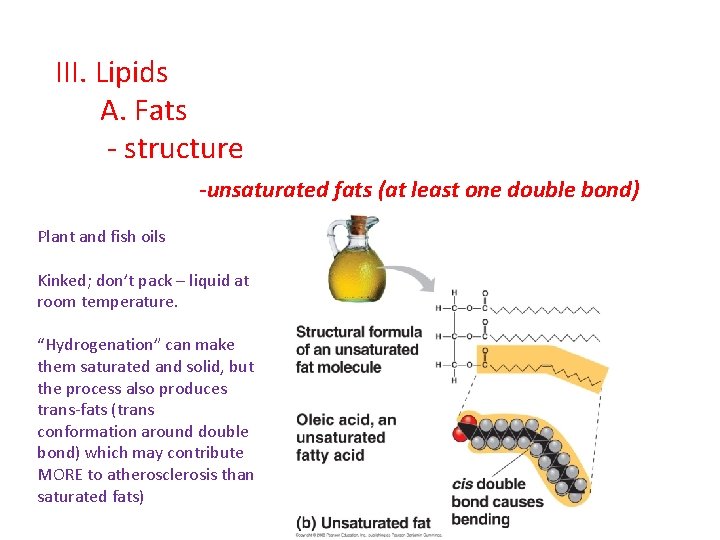
III. Lipids A. Fats - structure -unsaturated fats (at least one double bond) Plant and fish oils Kinked; don’t pack – liquid at room temperature. “Hydrogenation” can make them saturated and solid, but the process also produces trans-fats (trans conformation around double bond) which may contribute MORE to atherosclerosis than saturated fats)

III. Lipids A. Fats - structure - functions - long term energy storage (dense) not vital in immobile organisms (mature plants), so it is metabolically easier to store energy as starch. But in seeds and animals (mobile), there is selective value to packing energy efficiently, in a small space. In animals, fat is stored in adipose cells

III. Lipids A. Fats - structure - functions - long term energy storage (dense) - insulation (subcutaneous fat) - cushioning
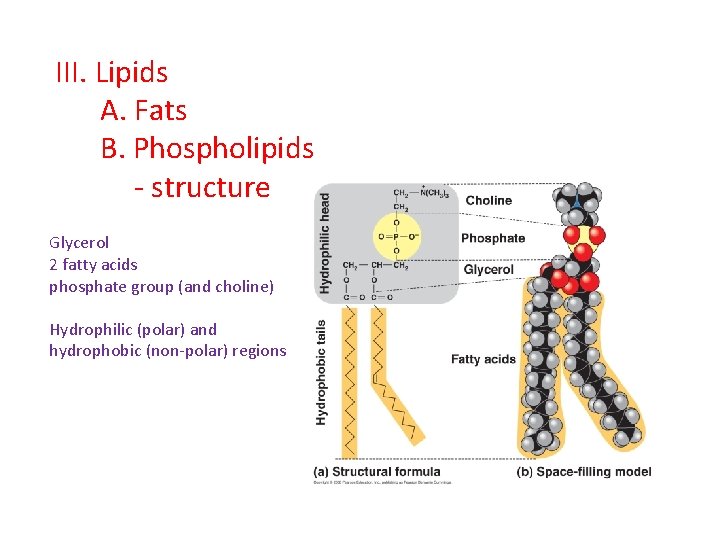
III. Lipids A. Fats B. Phospholipids - structure Glycerol 2 fatty acids phosphate group (and choline) Hydrophilic (polar) and hydrophobic (non-polar) regions
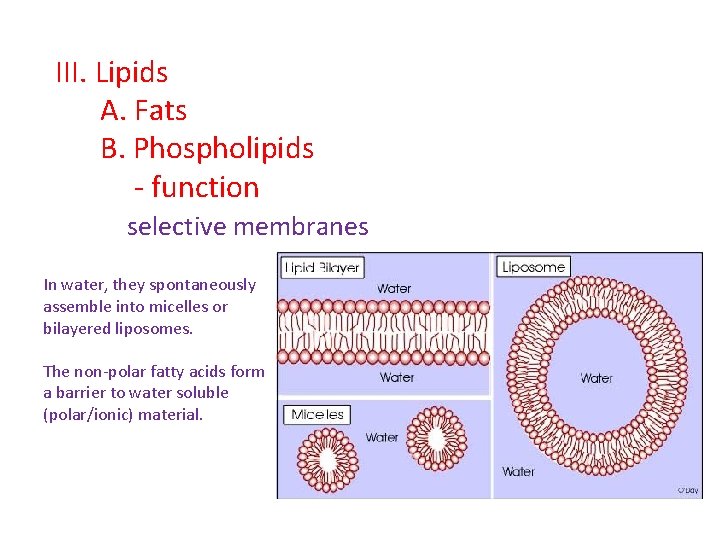
III. Lipids A. Fats B. Phospholipids - function selective membranes In water, they spontaneously assemble into micelles or bilayered liposomes. The non-polar fatty acids form a barrier to water soluble (polar/ionic) material.
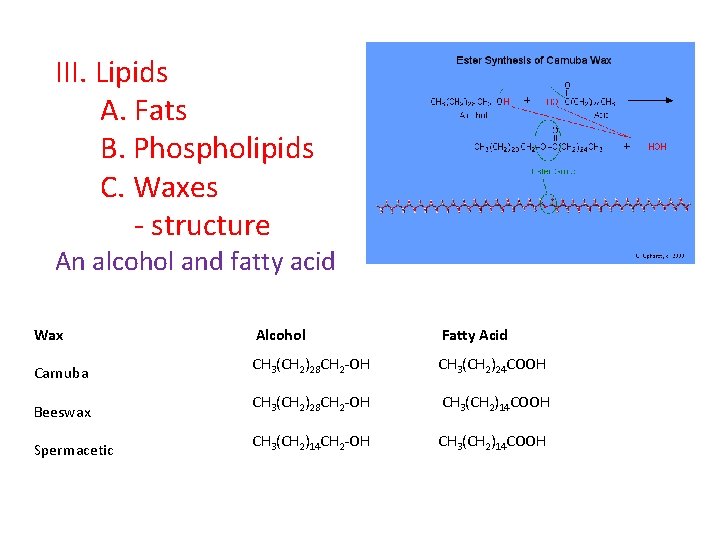
III. Lipids A. Fats B. Phospholipids C. Waxes - structure An alcohol and fatty acid Wax Alcohol Fatty Acid Carnuba CH 3(CH 2)28 CH 2 -OH CH 3(CH 2)24 COOH Beeswax CH 3(CH 2)28 CH 2 -OH CH 3(CH 2)14 COOH Spermacetic CH 3(CH 2)14 CH 2 -OH CH 3(CH 2)14 COOH

III. Lipids A. Fats B. Phospholipids C. Waxes - structure - function Retard the flow of water (plant waxes) Structural (beeswax in honeycomb) Signals – waxes on the exoskeleton can signal an insect’s identity and sexual receptivity.

III. Lipids A. Fats B. Phospholipids C. Waxes D. Steroids - structure typically a four-ring structure with side groups cholesterol and its hormone derivatives
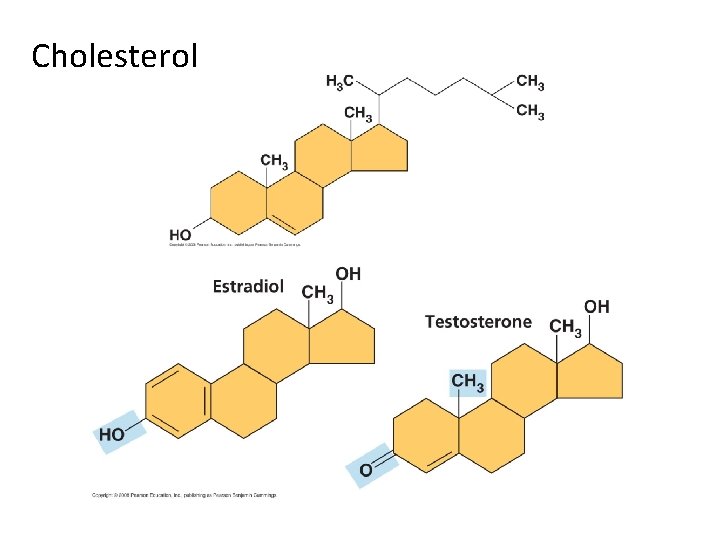
Cholesterol
- Slides: 25