Biological Science Sixth Edition Chapter 8 Energy and

Biological Science Sixth Edition Chapter 8 Energy and Enzymes: An Introduction to Metabolism Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved
Chapter 8 Opening Roadmap Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Energy in Chemical Reactions • Two types of energy exist • Kinetic energy is energy of motion – Thermal energy: energy of molecules moving • Potential energy is energy that is stored in position or configuration – Chemical energy: energy stored in chemical bonds • Energy can be transformed from one type to another Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved
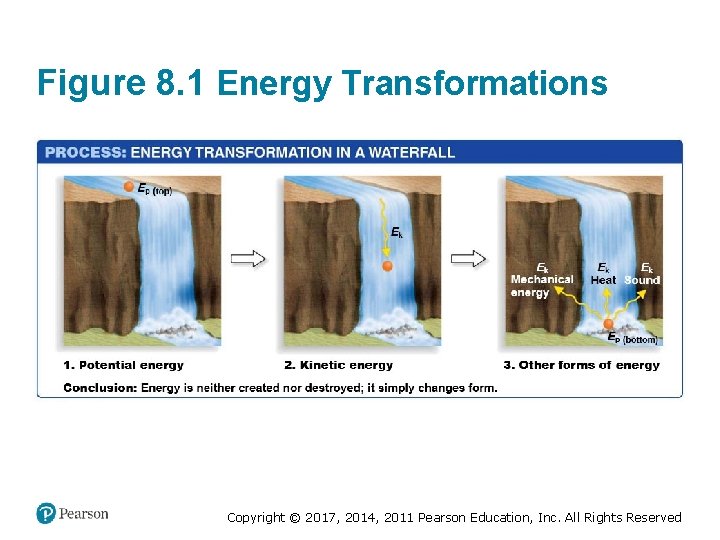
Figure 8. 1 Energy Transformations Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Chemical Reactions Involve Energy Transformations (1 of 7) • The amount of chemical energy in a covalent bond is based on – Position of shared electrons – Distance from positive charges in nuclei • The potential energy of a molecule depends on the configuration and position of its shared electrons – Weaker bonds with equally shared electrons have high potential energy – Stronger bonds with unequally shared electrons have low potential energy Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved
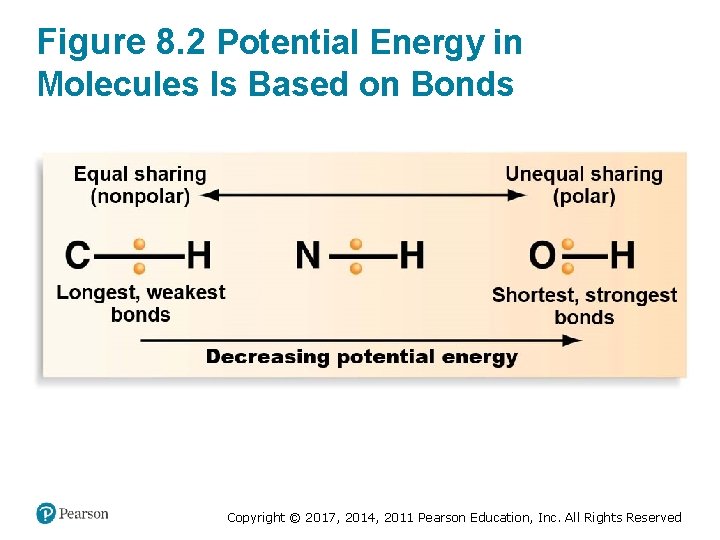
Figure 8. 2 Potential Energy in Molecules Is Based on Bonds Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Chemical Reactions Involve Energy Transformations (2 of 7) • In chemical reactions, if products have shorter, stronger covalent bonds than the reactants – The potential energy in the bonds decreases – Change in energy is transformed to thermal energy or light Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Chemical Reactions Involve Energy Transformations (3 of 7) • Energy is conserved • Energy cannot be created or destroyed • Energy can only be transferred and transformed • Enthalpy (H) includes – The potential energy of the molecule (heat content) – Effect of the molecule on surrounding pressure and volume • Changes in enthalpy (ΔH) are primarily based on the difference in potential energy Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Figure 8. 3 Reactions May Be Explosive Due to Changes in Enthalpy Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Chemical Reactions Involve Energy Transformations (4 of 7) • Exothermic reactions – Release heat energy – ΔH < 0 – Products have less potential energy than reactants • Endothermic reactions – Heat energy is taken up – ΔH > 0 – Products have higher potential energy than reactants Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Chemical Reactions Involve Energy Transformations (5 of 7) • Entropy (S) is the amount of disorder • When the products of a chemical reaction become less ordered than the reactant molecules – Entropy increases – ΔS > 0 • Second law of thermodynamics states that total entropy always increases in a system Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Chemical Reactions Involve Energy Transformations (6 of 7) • Gibbs free energy (G) determines whether a reaction is spontaneous or requires added energy to proceed • Calculate the change in G (ΔG) during the reaction • Standard free-energy change equation: – ΔH = change in enthalpy – ΔS = change in entropy – T = temperature in degrees Kelvin Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Chemical Reactions Involve Energy Transformations (7 of 7) • ΔG < 0 = a spontaneous reaction – These reactions are exergonic • ΔG > 0 = a nonspontaneous reaction that requires energy input to occur – These reactions are endergonic • ΔG = 0 = a reaction that is at equilibrium Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Temperature and Concentration Affect Reaction Rates • For most reactions to proceed – One or more chemical bonds have to break – Others have to form • Substances must collide in a specific orientation that brings the electrons involved near each other • Higher concentrations and higher temperature increase the number of collisions • Higher concentrations and higher temperature therefore increase reaction rate Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved
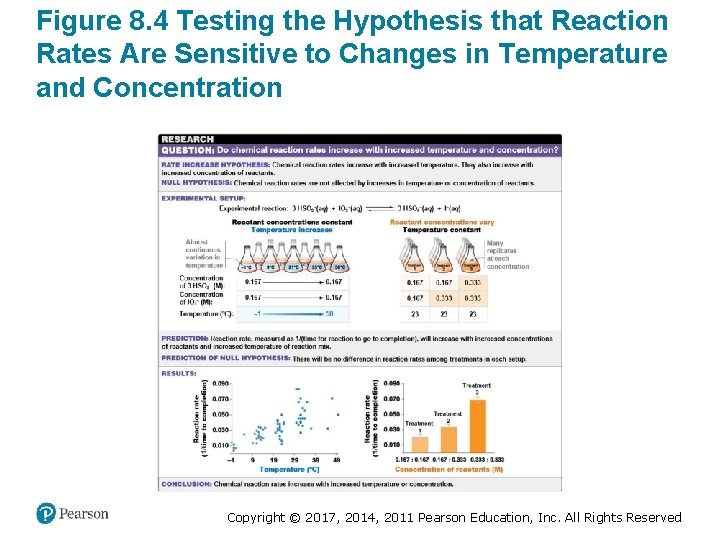
Figure 8. 4 Testing the Hypothesis that Reaction Rates Are Sensitive to Changes in Temperature and Concentration Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Nonspontaneous Reactions May Be Driven Using Chemical Energy • Energetic coupling – Between exergonic and endergonic reactions – Allows chemical energy released from one reaction – To drive another reaction Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Figure 8. 5 Energetic Coupling Allows Endergonic Reactions to Proceed Using the Free Energy Released from Exergonic Reactions Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Redox Reactions Transfer Energy via Electrons (1 of 2) • Reduction–oxidation reactions (redox reactions) – Are chemical reactions that involve electron transfer § Oxidation: loss of an electron(s) § Reduction: gain of an electron(s) – Always occur together – Represent energetic coupling of two half-reactions § Oxidation is exergonic § Reduction is endergonic Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Web Activity: Redox Reactions Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Redox Reactions Transfer Energy via Electrons (2 of 2) • During a redox reaction, electrons can – Be transferred completely from one atom to another – Simply shift their position in covalent bonds Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Figure 8. 6 Redox Reactions Involve the Gain or Loss of One or More Electrons Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Another Approach to Understanding Redox (1 of 2) • Electrons can be transferred from an electron donor to an electron acceptor • Most electron acceptors gain potential energy as they are reduced • Electrons are usually accompanied by a proton – Reduction often “adds Hs” – Oxidation often “removes Hs” Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Another Approach to Understanding Redox (2 of 2) • Flavin adenine dinucleotide (FAD) accepts two electrons plus two protons to form FADH 2 • Nicotinamide adenine dinucleotide (NAD+) accepts two electrons plus one proton to form NADH • These electron carriers readily donate electrons to other molecules Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved
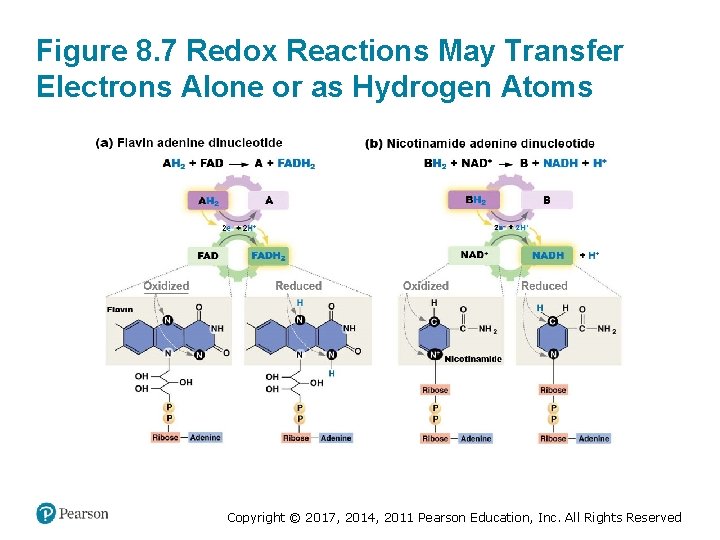
Figure 8. 7 Redox Reactions May Transfer Electrons Alone or as Hydrogen Atoms Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

ATP Transfers Energy via Phosphate Groups • Adenosine triphosphate (ATP) is the energy currency for cells – It provides the fuel for most cellular activities • ATP forms bonds between three negatively charged phosphate groups – Negative charges repel each other – High-energy bonds store a large amount of potential energy Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved
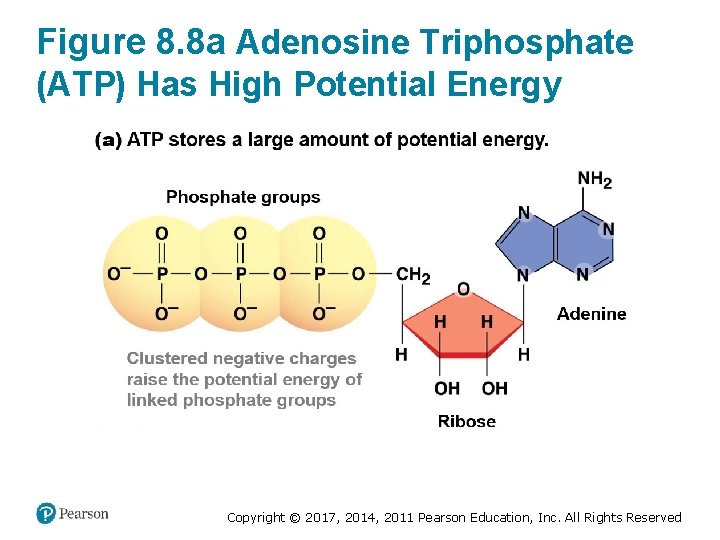
Figure 8. 8 a Adenosine Triphosphate (ATP) Has High Potential Energy Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved
Cell Biology Video: Structure of ATP (Space Filling Model) Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved
Cell Biology Video: Structure of ATP (Stick Model) Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

ATP Hydrolysis Releases Free Energy • Hydrolysis of the bond between the two outermost phosphate groups results – In formation of ADP and Pi (inorganic phosphate, ) – In a highly exergonic reaction § Releases 7. 3 kilocalories of energy per mole of ATP Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved
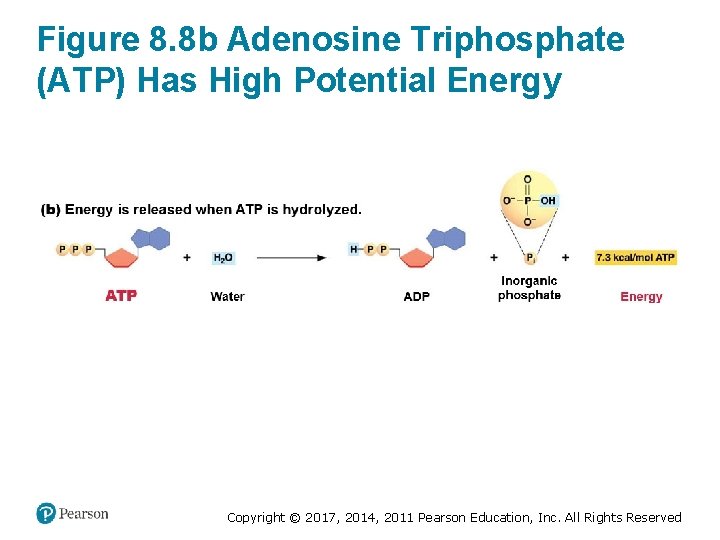
Figure 8. 8 b Adenosine Triphosphate (ATP) Has High Potential Energy Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

How Does ATP Drive Endergonic Reactions? • Energy released during ATP hydrolysis is transferred to a substrate by phosphorylation – Phosphorylation is adding a phosphate group – Usually causes a change in the protein’s shape • Exergonic phosphorylation reactions are coupled to endergonic reactions Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Figure 8. 9 Exergonic Phosphorylation Reactions Are Coupled to Endergonic Reactions Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

How Enzymes Work • Most biological chemical reactions occur fast enough only in the presence of an enzyme • Enzymes are protein catalysts – Bring reactants together in precise orientations – Make reactions more likely – Are specific for a single type of reaction Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Enzymes Help Reactions Clear Two Hurdles • Before a reaction can take place, reactants need to 1. Collide in a precise orientation 2. Have enough kinetic energy to overcome repulsion between electrons that come into contact as a bond forms Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Enzymes Bring Substrates Together (1 of 2) • Enzymes bring substrates together • Substrates bind to the enzyme’s active site – Enzymes help them collide in a precise orientation – Bonds break and form to generate products • Many enzymes undergo a conformational change – When the substrates are bound to the active site – This change is called an induced fit Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved
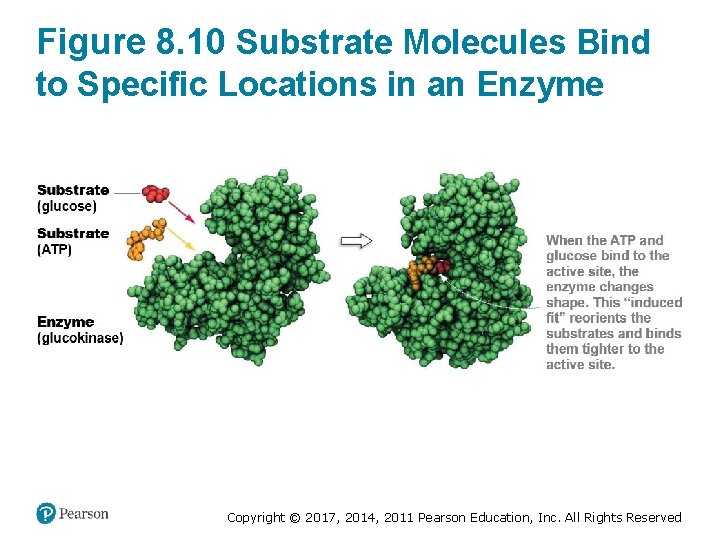
Figure 8. 10 Substrate Molecules Bind to Specific Locations in an Enzyme Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Enzymes Bring Substrates Together (2 of 2) • Substrates bind via hydrogen bonding or other interactions with amino acid residues in the active site • An unstable intermediate condition called the transition state is formed • Activation energy (Ea) is required to strain substrates’ bonds so they can reach the transition state Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Enzymes Lower the Activation Energy (1 of 3) • Reactions occur when – Reactants have enough kinetic energy to reach the transition state – The kinetic energy of molecules is a function of their temperature • Thus, reaction rates depend on – The kinetic energy of the reactants – The activation energy of particular reaction (the free energy of the transition state) Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved
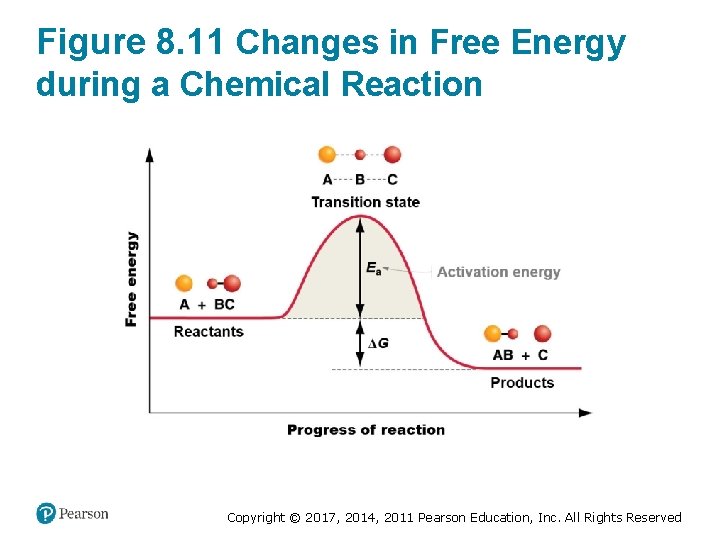
Figure 8. 11 Changes in Free Energy during a Chemical Reaction Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Enzymes Lower the Activation Energy (2 of 3) • Interactions between the enzyme and the substrate – Stabilize the transition state – Lower the activation energy required for the reaction to proceed • Enzymes are not consumed during the reaction Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved
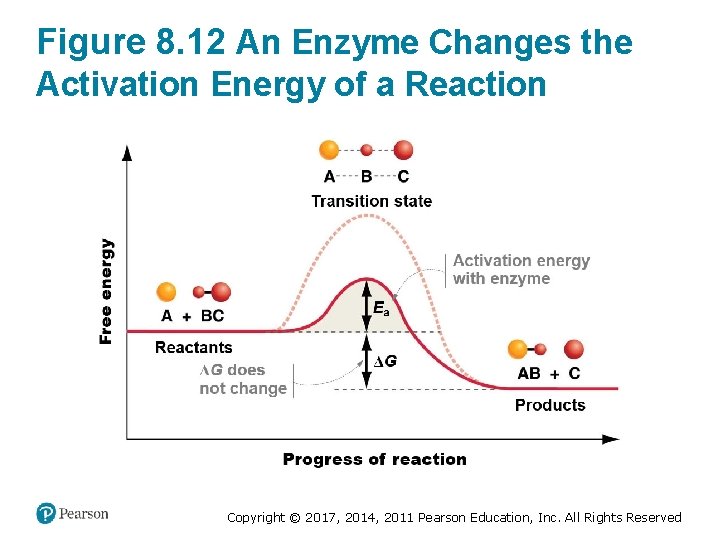
Figure 8. 12 An Enzyme Changes the Activation Energy of a Reaction Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Enzymes Lower the Activation Energy (3 of 3) • Enzyme catalysis has three steps: 1. Initiation—substrates are precisely oriented as they bind to the active site 2. Transition state facilitation—interactions between the substrate and active site R-groups lower the activation energy 3. Termination—reaction products are released from the enzyme Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved
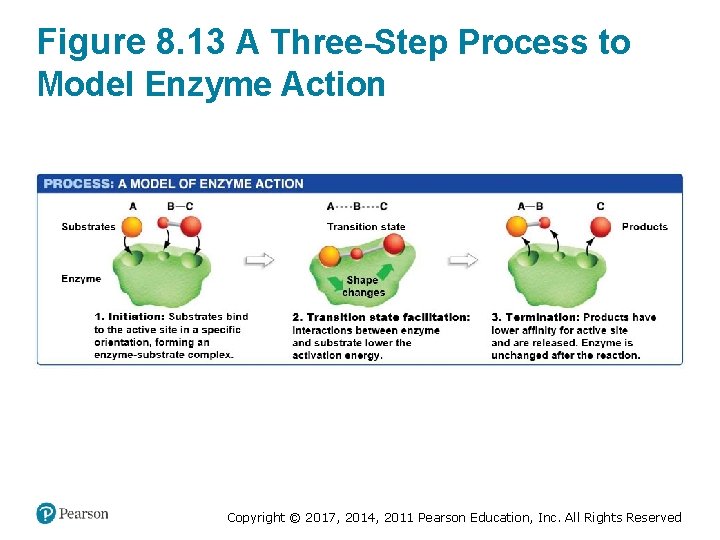
Figure 8. 13 A Three-Step Process to Model Enzyme Action Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

What Limits the Rate of Catalysis? (1 of 2) • The speed of an enzyme-catalyzed reaction 1. Increases linearly at low substrate concentrations 2. Slows as substrate concentration increases 3. Reaches maximum speed at high substrate concentrations Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved
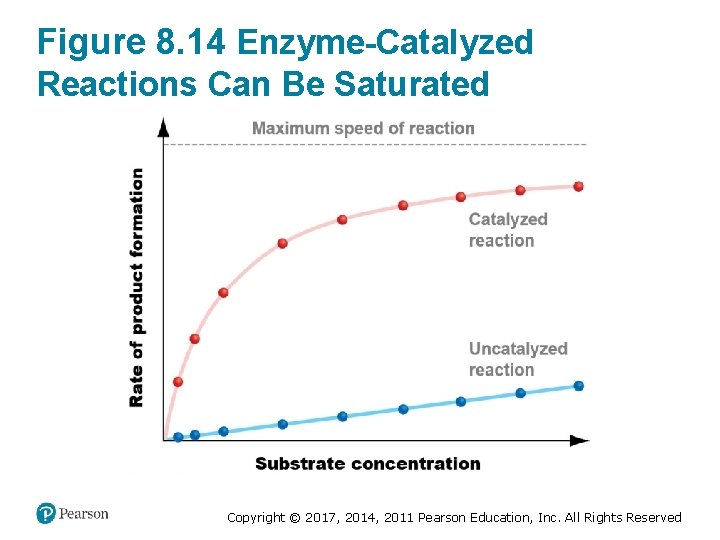
Figure 8. 14 Enzyme-Catalyzed Reactions Can Be Saturated Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

What Limits the Rate of Catalysis? (2 of 2) • All enzymes show this type of “saturation kinetics” – Active sites cannot accept substrates any faster – No matter how large the concentration of substrates gets – Reaction rates level off because all available enzyme molecules are being used Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Do Enzymes Work Alone? • Many enzymes are regulated by molecules that are not part of the enzyme itself: 1. Cofactors are inorganic ions, such as that reversibly interact with enzymes 2. Coenzymes are organic molecules, such as NADH or FADH 2, that interact with enzymes 3. Prosthetic groups are non-amino acid atoms or molecules that are permanently attached to proteins Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

What Factors Affect Enzyme Function? • An enzyme’s structure is critical to its function • Protein structure is dependent on folding • Enzyme function is dependent on certain conditions – Temperature affects the folding and movement of the enzyme and its substrates – p. H affects the enzyme’s shape and reactivity • Each enzyme has optimal temperature and p. H Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved
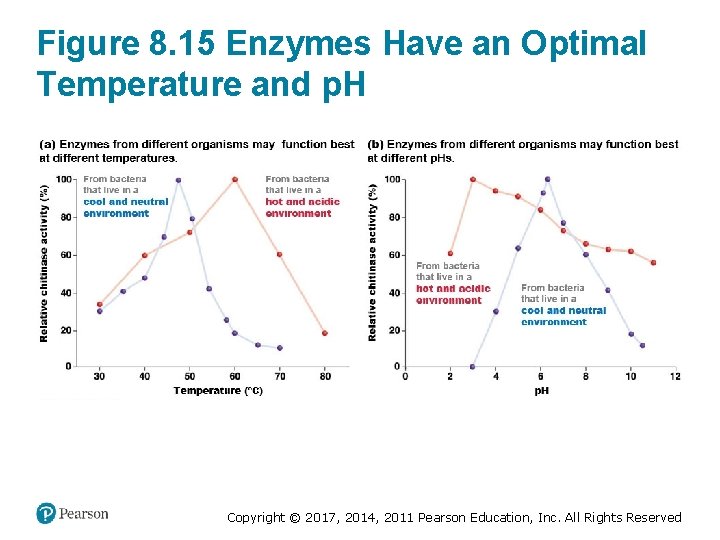
Figure 8. 15 Enzymes Have an Optimal Temperature and p. H Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Most Enzymes Are Regulated • Regulatory molecules may control when and where an enzyme functions – May change the enzyme’s structure – Or its ability to bind its substrate – May either activate or inactivate the enzyme’s function Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Regulating Enzymes via Noncovalent Interactions • Regulation via noncovalent interactions – Does not permanently affect the enzyme structure – Is referred to as “reversible” • Competitive inhibition occurs when a molecule competes with the substrate for the active site • Allosteric regulation occurs when a molecule binds at a location other than the active site – Causes a change in enzyme shape – Can activate or deactivate the enzyme Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Figure 8. 16 An Enzyme’s Activity Is Precisely Regulated Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Regulating Enzymes via Covalent Modifications • Regulation may involve covalent modifications – Changes the enzyme’s primary structure – Can be reversible or irreversible • Irreversible changes often result from cleavage of peptide bonds • Most common reversible modification of enzymes is the addition of phosphate groups (phosphorylation) – Causes a change in shape – May activate or inactivate the enzyme Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved
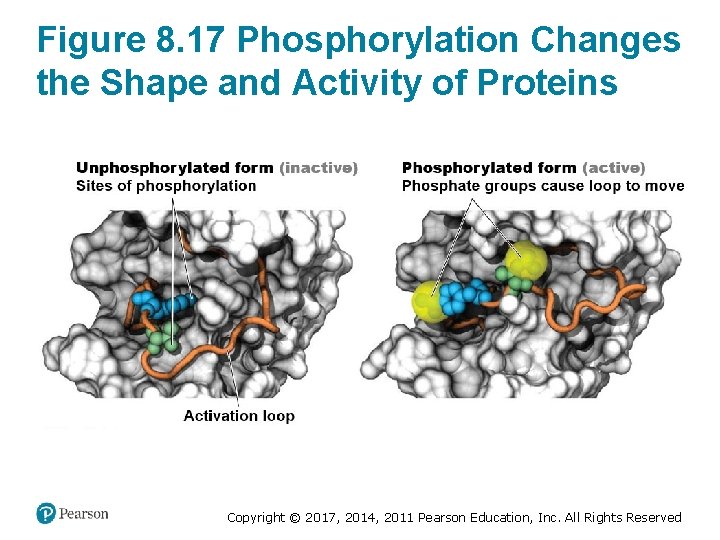
Figure 8. 17 Phosphorylation Changes the Shape and Activity of Proteins Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Enzymes Can Work Together in Metabolic Pathways • Metabolic pathways – A series of reactions – Each catalyzed by a different enzyme – To build biological molecules Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Metabolic Pathways Are Regulated • Feedback inhibition occurs – When an enzyme in a pathway is inhibited – By the final product of that pathway • Pathway can shut down when – Products are no longer needed by the cell Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved
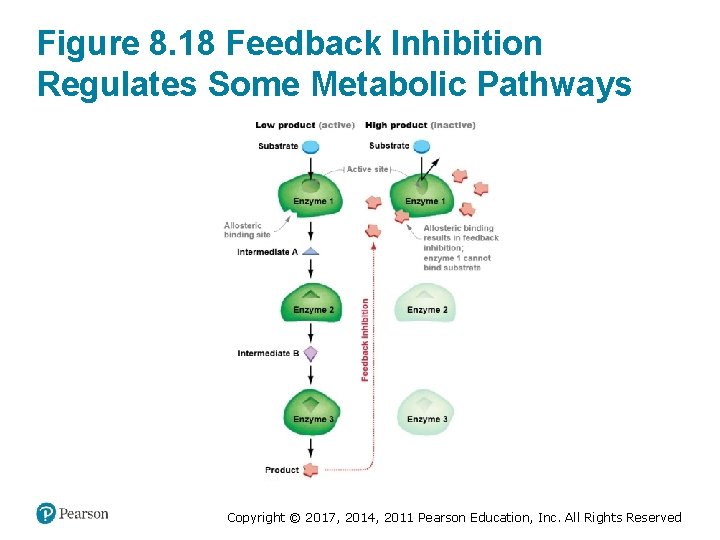
Figure 8. 18 Feedback Inhibition Regulates Some Metabolic Pathways Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Metabolic Pathways Evolve (1 of 2) • Enzymes evolved to make building blocks of life • If substrates decline, new enzymes evolve to make more of the substrates • Retro-evolution—repetition of backward process produces multistep metabolic pathway • Patchwork evolution—new enzymes are recruited to new pathways • Bioremediation—engineering new pathways to clean up pollutants Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved
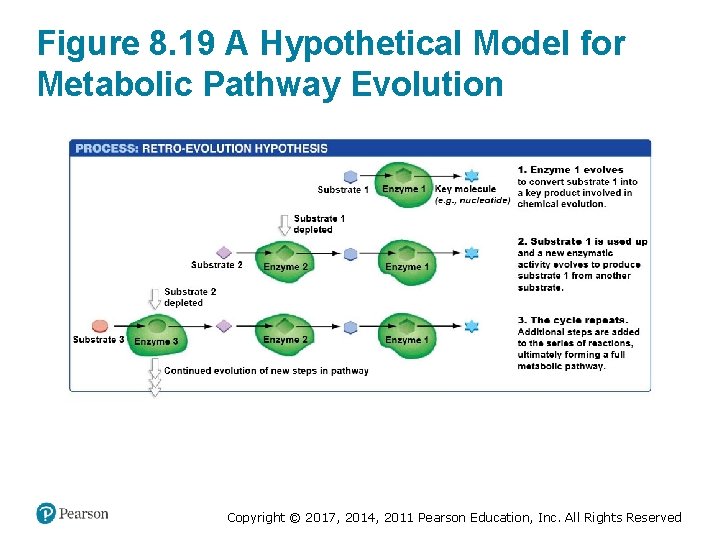
Figure 8. 19 A Hypothetical Model for Metabolic Pathway Evolution Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved

Metabolic Pathways Evolve (2 of 2) • Metabolic pathways are vital to all cells – Catabolic pathways break down molecules – Anabolic pathways synthesize molecules Copyright © 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved
- Slides: 60