Biological Molecules Organic Chemistry Functional Groups Organic Compounds

Biological Molecules

Organic Chemistry & Functional Groups

Organic Compounds n Carbon-based molecules n Second most abundant type of compound found in organisms n Over 2 million known organic compounds n Properties of organic compounds depend on its size, shape, and type of functional group attached to it

What is a Functional Group? Atoms, such as nitrogen, oxygen, phosphate and sulfur covalently bonded to a carbon backbone n They are groups that change both the structure and behavior of a molecule, i. e. , solubility, reactivity with other molecules n They help chemists and biochemists classify different molecules found in living organisms n Functional groups containing nitrogen or oxygen are polar and therefore hydrophylic and thus water-soluble n

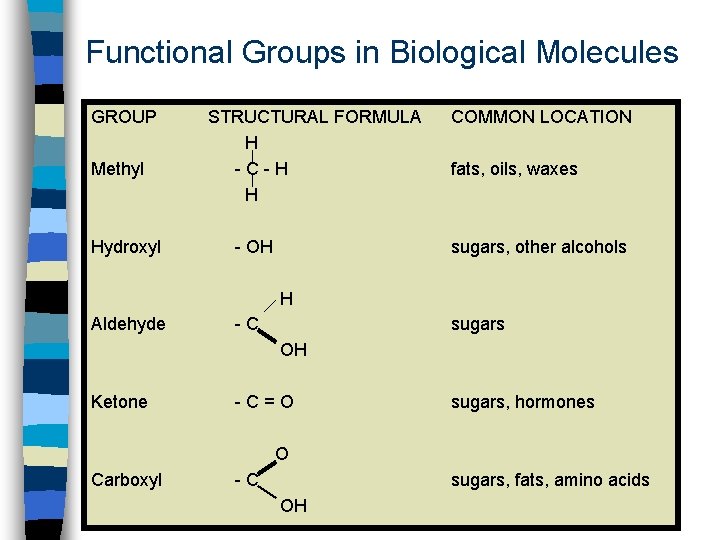
Functional Groups in Biological Molecules GROUP Methyl Hydroxyl STRUCTURAL FORMULA H -C-H H - OH COMMON LOCATION fats, oils, waxes sugars, other alcohols H Aldehyde -C sugars OH Ketone -C=O sugars, hormones O Carboxyl -C sugars, fats, amino acids OH
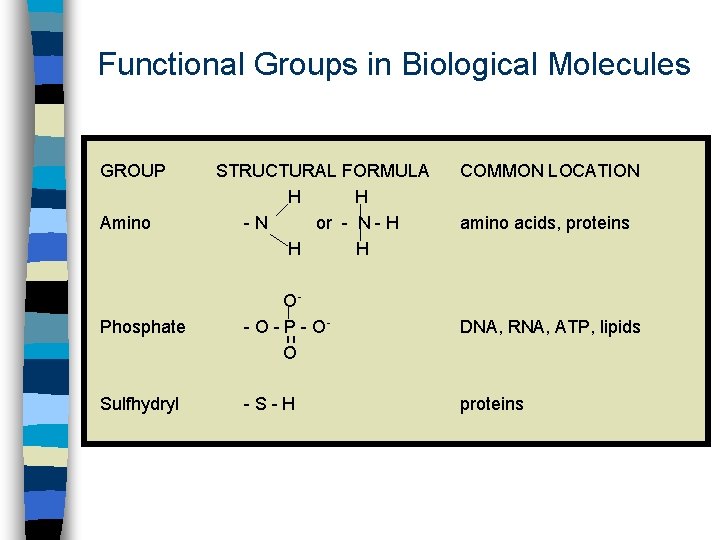
Functional Groups in Biological Molecules GROUP Amino STRUCTURAL FORMULA H H -N or - N - H H H COMMON LOCATION amino acids, proteins Phosphate O- O - P - OO DNA, RNA, ATP, lipids Sulfhydryl -S-H proteins


Five Classes of Chemical Reactions in Biological Systems

Five Classes of Reactions 1. 2. 3. 4. 5. Functional-group transfer Electron transfer Rearrangement Condensation (dehydration) Cleavage (hydrolysis)

1. Functional-Group Transfer n one molecule gives up a functional group, which another molecule accepts n seen in metabolic reactions, ex. , glycolysis

2. Electron Transfer n one or more electrons stripped from one molecule are donated to another molecule n seen in metabolic reactions, ex. , Kreb’s cycle, glycolysis

3. Rearrangement na juggling of internal bonds converts one type of organic compound into another n seen in many metabolic pathways ex. , glycolysis, Kreb’s cycle, etc.

4. Condensation (Dehydration) n through covalent bonding, two molecules combine to form a larger molecule n many successive condensation reactions leads to polymerization n cells undergo condensation reactions to produce complex carbohydrates, lipids and proteins
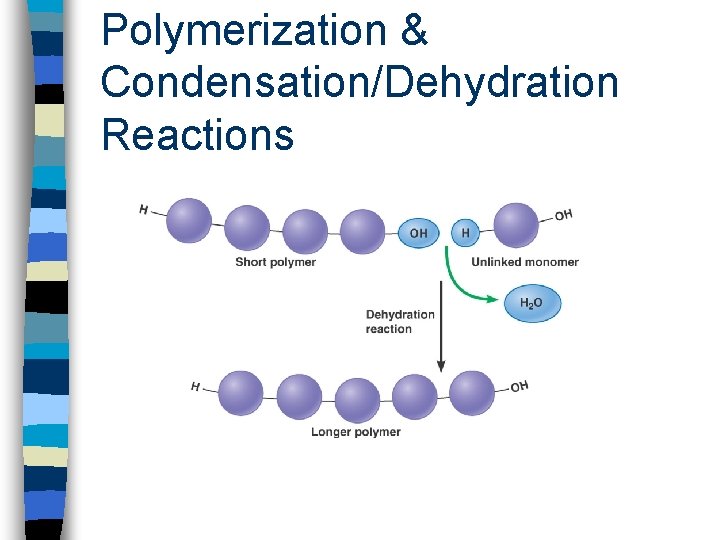
Polymerization & Condensation/Dehydration Reactions

5. Cleavage (Hydrolysis) na molecule splits into two smaller ones n hydrolysis, is a very common biological cleavage. It is like condensation in reverse n cells hydrolyze large polymers like starch and proteins, then use the released subunits as building blocks or energy sources
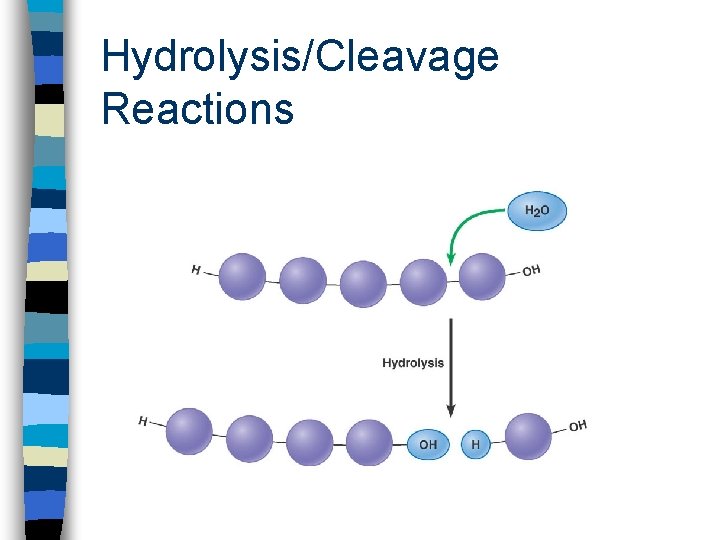
Hydrolysis/Cleavage Reactions

Carbohydrates

Carbohydrates na simple sugar or a molecule composed of two or more sugar units n can be used as either an immediate energy source, stored energy source or structural material n 3 classes are: 1) monosaccharides, 2) oligosaccharides, 3) polysaccharides

Monosaccharides n n n n Simplest carbohydrates Also called simple sugars Suffix - ose Glucose, Fructose, Galactose (hexoses) Ribose (pentose) Empirical formula is generally CH 2 O Usually form ring structures in aqueous solution functional groups include: hydroxyl and aldehyde or ketone
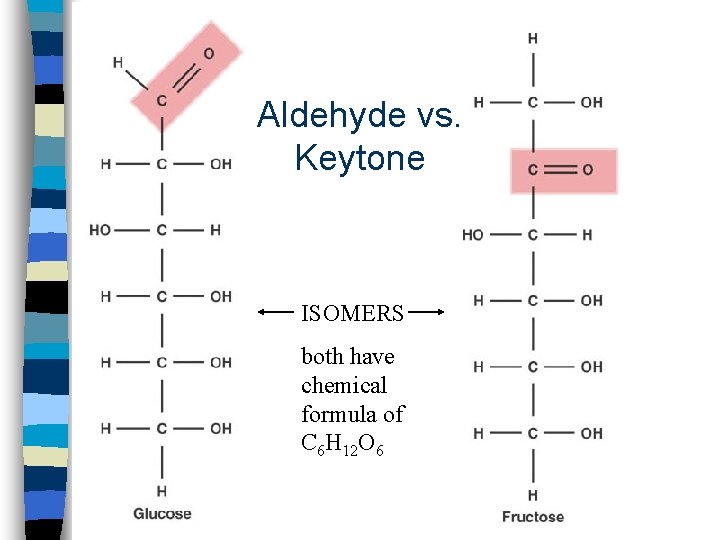
Aldehyde vs. Keytone ISOMERS both have chemical formula of C 6 H 12 O 6
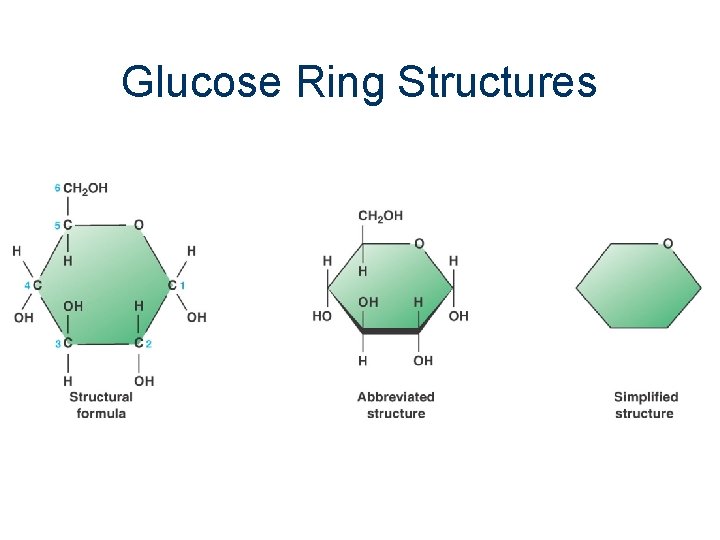
Glucose Ring Structures

Oligosaccharide n Is a short chain of two or more covalently bonded sugar units n Two or more monosaccharides join by dehydration reactions n Disaccharide = two sugars – lactose - milk sugar (glucose + galactose) – sucrose - fruit sugar (glucose + fructose) – maltose - beer, seeds (glucose + glucose)
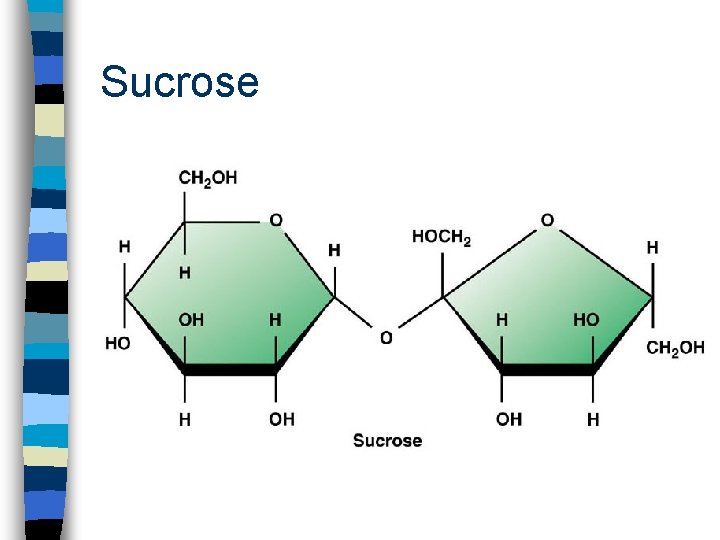
Sucrose
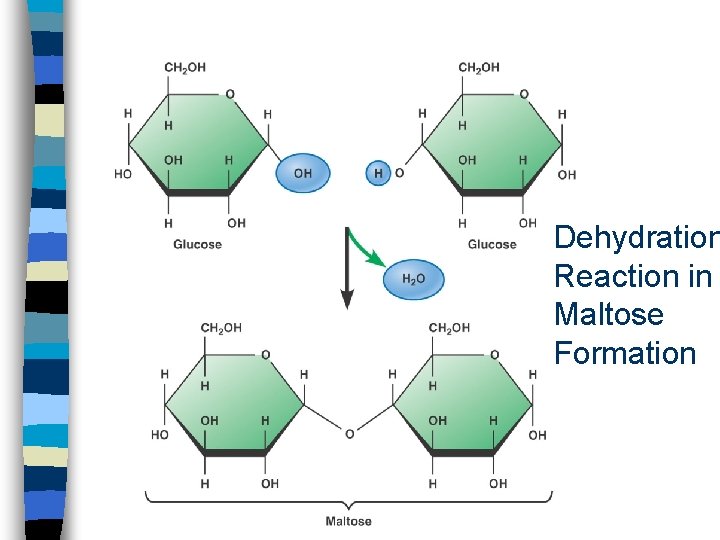
Dehydration Reaction in Maltose Formation

Complex Carbohydrates Polysaccharides n straight or branched chain of hundreds or thousands of the same or different sugar units (monomers) n glucose-based polysaccharides: – starch - plant energy storage – cellulose - plant structural form – glycogen - animal energy storage
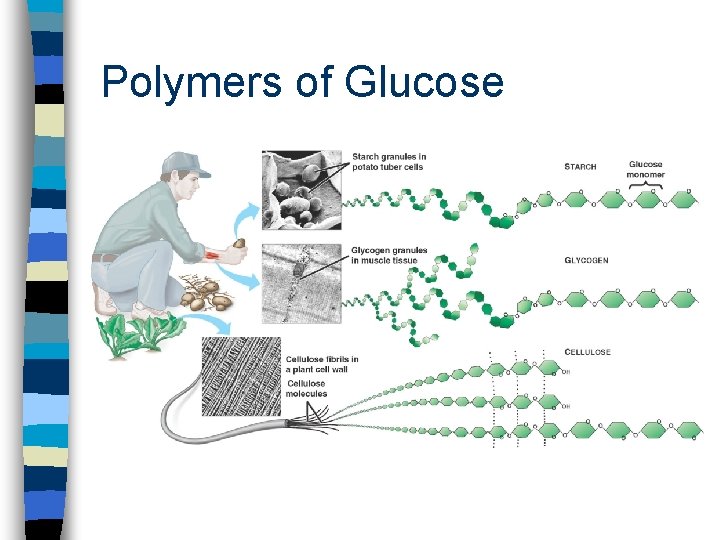
Polymers of Glucose

Fats & Lipids

Five Classes of Lipids n Fatty Acids n Triglycerides n Phospholipids n Waxes n Sterols

Fatty Acids n Contains a carbon backbone of up to 36 carbon atoms n Contains a carboxyl group at one end n Can be saturated (only single bonds) n Can be unsaturated (may have one or more double or triple bonds) n Saturated are solid at room temperature n Unsaturated are liquid

Saturated vs. Unsaturated Fatty Acids

Saturated & Unsaturated Fatty Acids

Trans Fatty Acids n Increases the « bad » LDL cholesterol (low-density lipoprotein) and decreases the good HDL (high-density lipoprotein) n Suggested that you should not consume more than 2 g of trans fat/day in a 2000 kcal/day diet
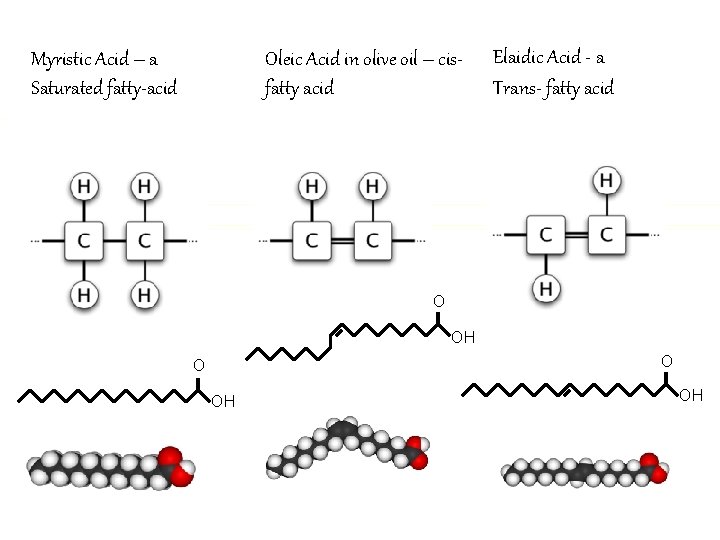
Oleic Acid in olive oil – cisfatty acid Myristic Acid – a Saturated fatty-acid Elaidic Acid - a Trans- fatty acid O OH OH

Other Effects of Trans Fats n Cancer n Type 2 diabetes n Obesity n Liver Dysfunction n Ovulatory Infertility

What if Trans fats are not labelled? n add up the values for saturated, polyunsaturated and monounsaturated fats. If the number is less than the "Total fats" shown on the label, the unaccounted is trans fat.

Triglycerides n Composed of a glycerol molecule and three fatty acids n Body’s most abundant lipid & best source of energy n FAT!!! n Adipose tissue contains high concentrations of triglycerides
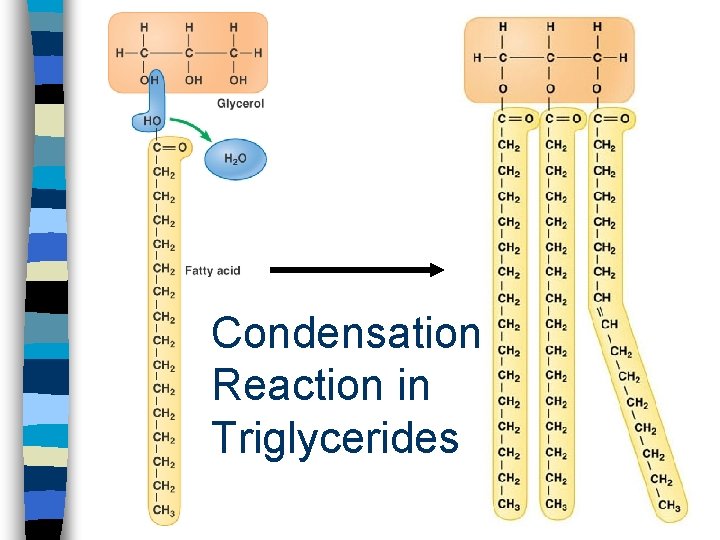
Condensation Reaction in Triglycerides

Phospholipids n Main component of cell membrane n Hydrophilic head - glycerol + phosphate n Hydrophobic tail - 2 fatty acids
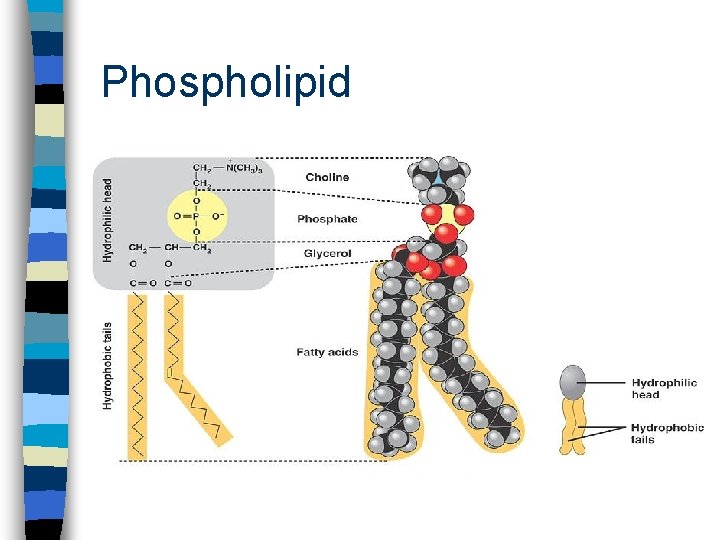
Phospholipid

WAXES

Waxes n Long-chain fatty acids linked to an alcohol or carbon rings n Very water repellent
Paraffin Wax n Breathing in paraffin candle wax may be carcinogenic
Bee’s Wax

Sterols/Steroids n No fatty acid tails! n Backbone of 4 fused carbon rings n Sterols differ functional group types and positions n Examples: cholesterol and hormones
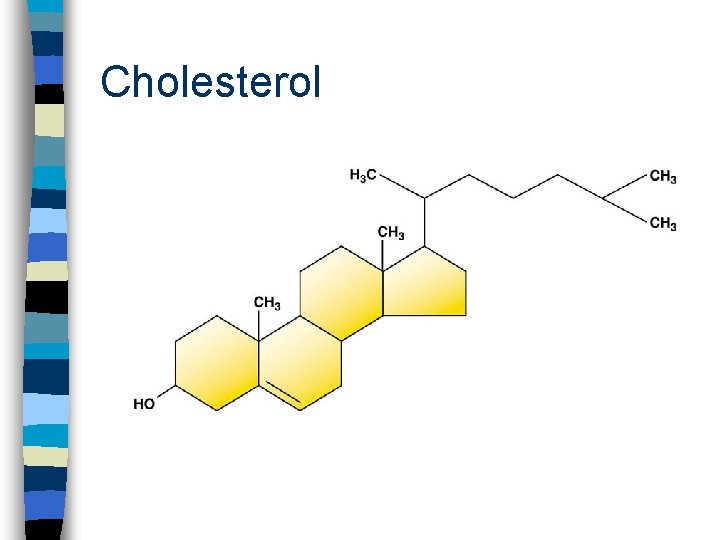
Cholesterol

Anabolic Steroids Synthetic variants of testosterone n Overdosing causes: n • • Mood swings Liver damage leading to cancer High blood pressure Shrinks testicles, reduces sex drive & causes infertility & breast enlargement in men • Disrupts menstral cycle & leads to male characteristics in females • Stunts growth & stops bone growth in teens

Proteins
Silk Proteins

Protein n Biological polymer constructed from amino acid monomers n Tens of thousands of proteins found in the human body n Each protein has its unique threedimensional structure that corresponds to a specific function n Seven classes of proteins

Classes of Proteins Protein n Structural – silk, hair, fibers, ligaments n Contractile – muscle n Storage – ovalbumin n Defensive – antibodies n Transport – hemoglobin, membrane n Signal – certain hormones n Enzymes - catalyst

Protein Shape n Consists chains n Either n Can of one or more polypeptide globular or fibrous in shape be denatured (unraveled) by heat, changes in salt concentration and p. H
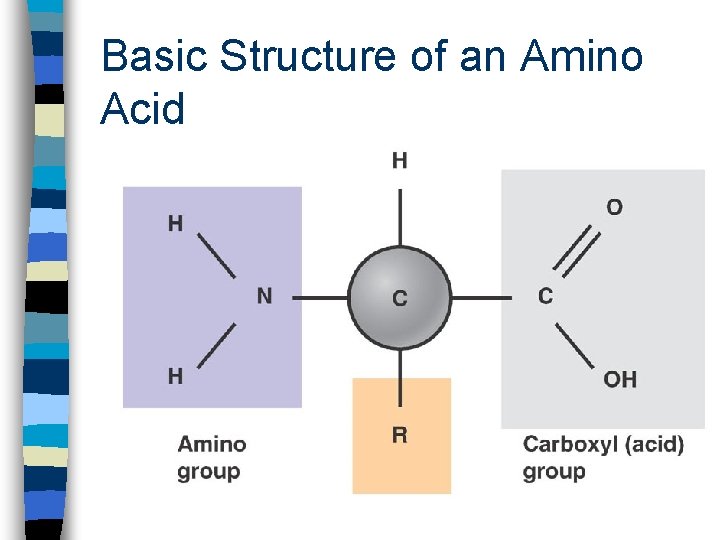
Basic Structure of an Amino Acid
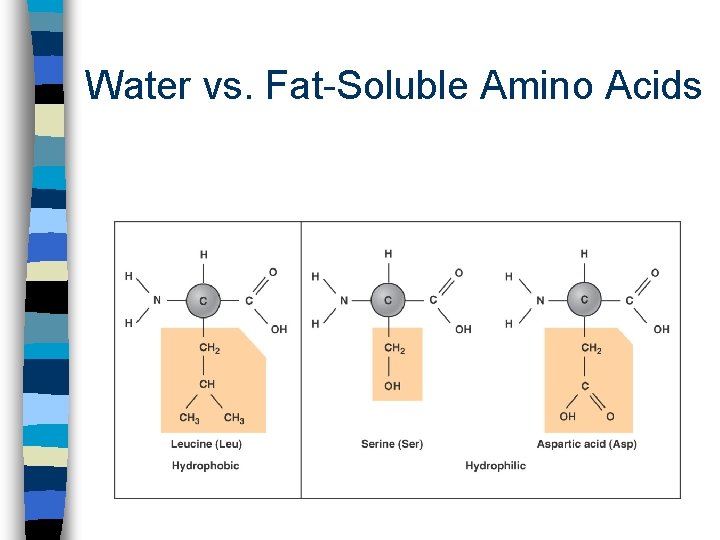
Water vs. Fat-Soluble Amino Acids
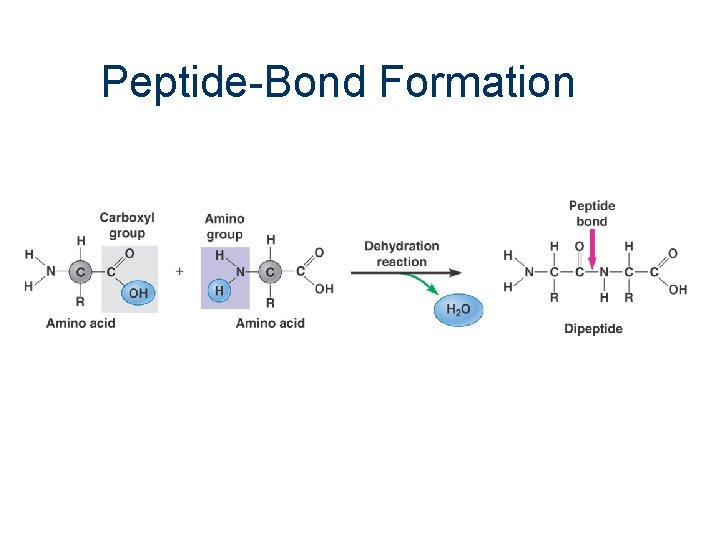
Peptide-Bond Formation

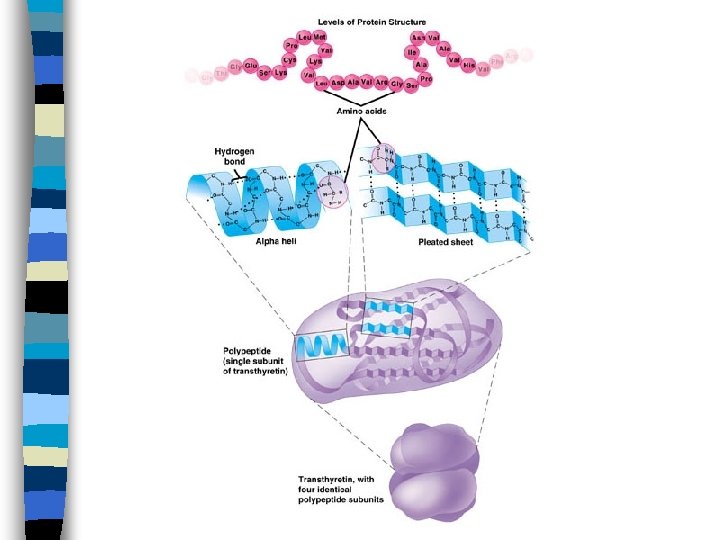
Structural Levels n Primary 1° n Secondary n Tertiary 2° 3° n Quaternary 4°

Primary Structure n Amino acid sequence
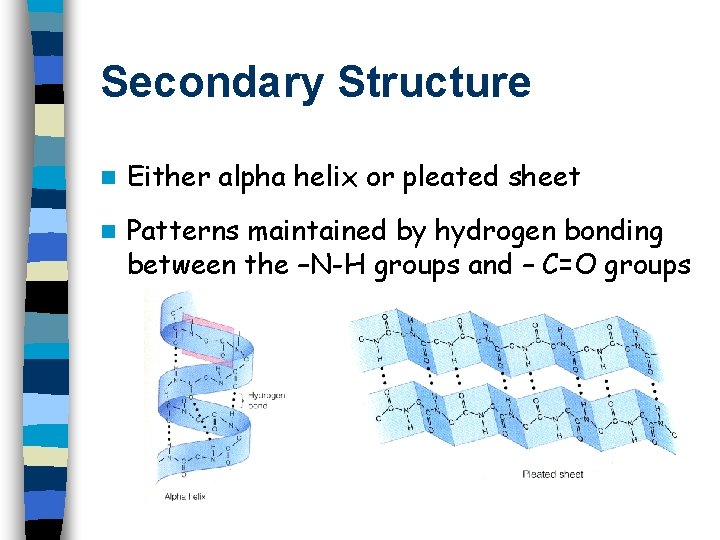
Secondary Structure n Either alpha helix or pleated sheet n Patterns maintained by hydrogen bonding between the –N-H groups and – C=O groups
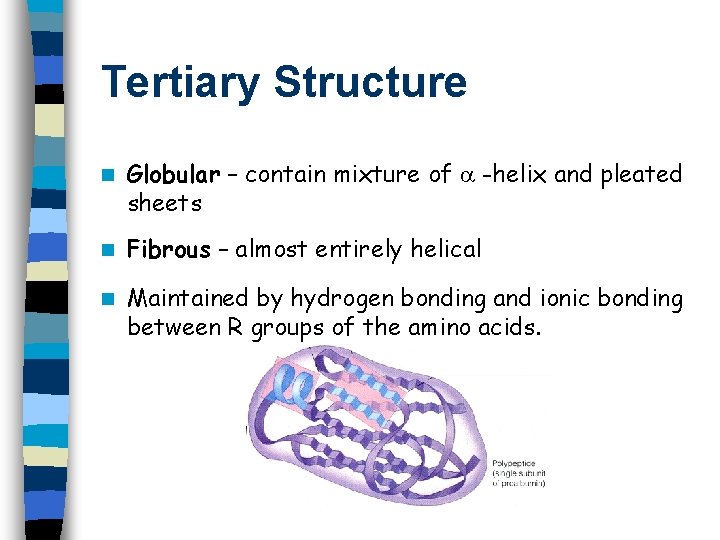
Tertiary Structure n Globular – contain mixture of -helix and pleated sheets n Fibrous – almost entirely helical n Maintained by hydrogen bonding and ionic bonding between R groups of the amino acids.

Quaternary Structure n Results from bonding interactions between different polypeptides or subunits
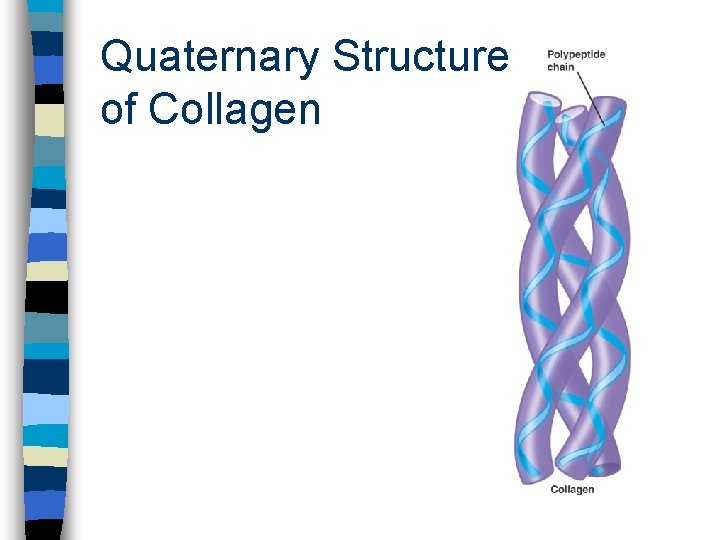
Quaternary Structure of Collagen
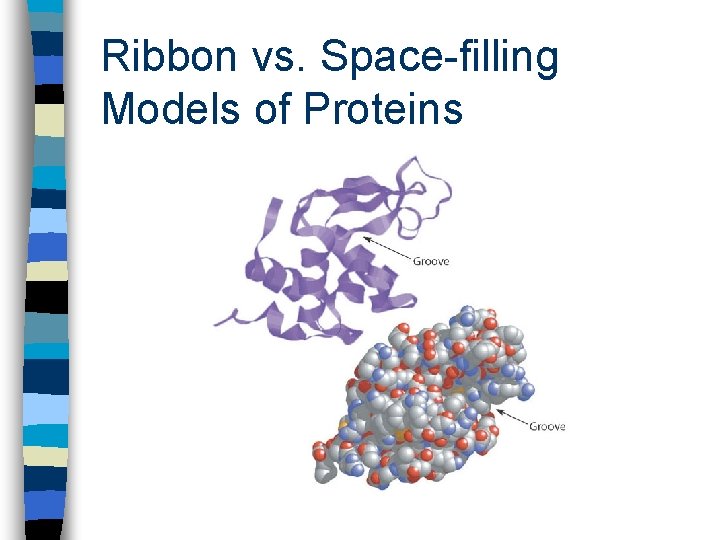
Ribbon vs. Space-filling Models of Proteins
Enzymes

Enzymes in General n speed up metabolic reactions that would normally take years to undergo by lowering the “activation energy” needed for a reaction to occur n are named by adding the suffix “ase” to part of the name of the substrate (ex. sucrase)
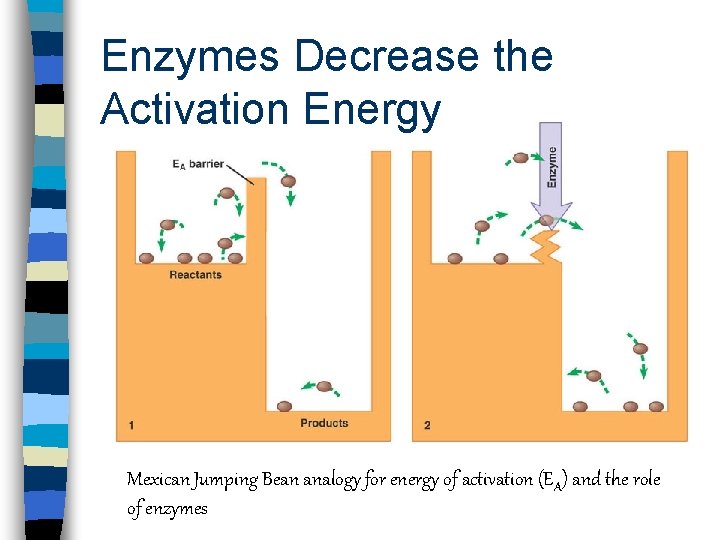
Enzymes Decrease the Activation Energy Mexican Jumping Bean analogy for energy of activation (E A) and the role of enzymes

Characteristics of Enzymes n Do not make any reaction occur that would not normally occur naturally n Do not get used up during the reaction n Can work both in the forward and reverse directions of a reaction n Are highly selective to specific substrates

Induced-Fit Model Enzymes have specifically shaped “active sites” on their surfaces that interact with the substrate(s) n As the substrate enters this active site it induces the enzyme to change shape so that the active site fits even more snugly around the substrate (clasping handshake) n This “induced-fit” strains the pre-existing bonds within the substrate(s) and promotes the formation of new bonds (in products) n
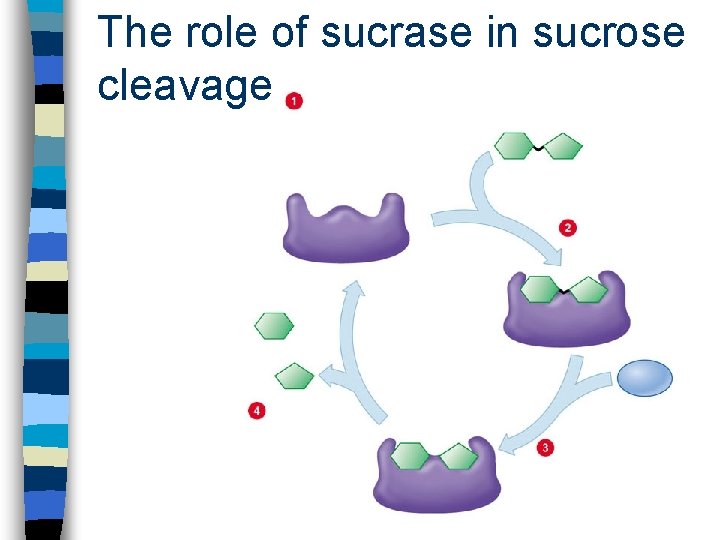
The role of sucrase in sucrose cleavage
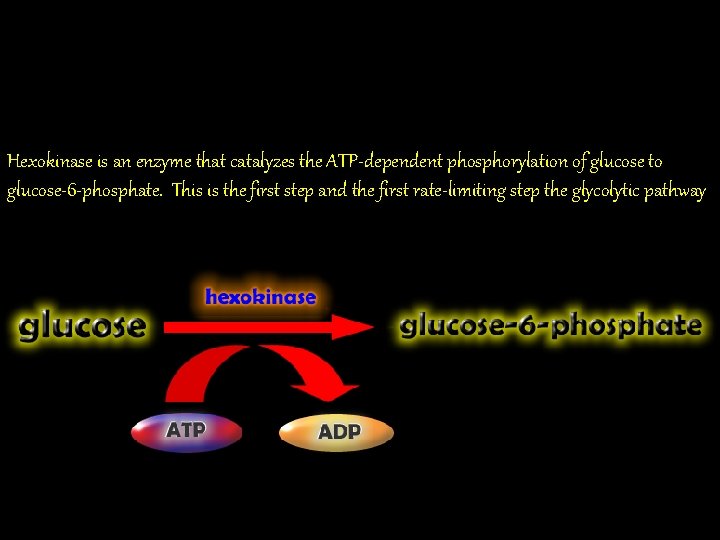
Hexokinase is an enzyme that catalyzes the ATP-dependent phosphorylation of glucose to glucose-6 -phosphate. This is the first step and the first rate-limiting step the glycolytic pathway
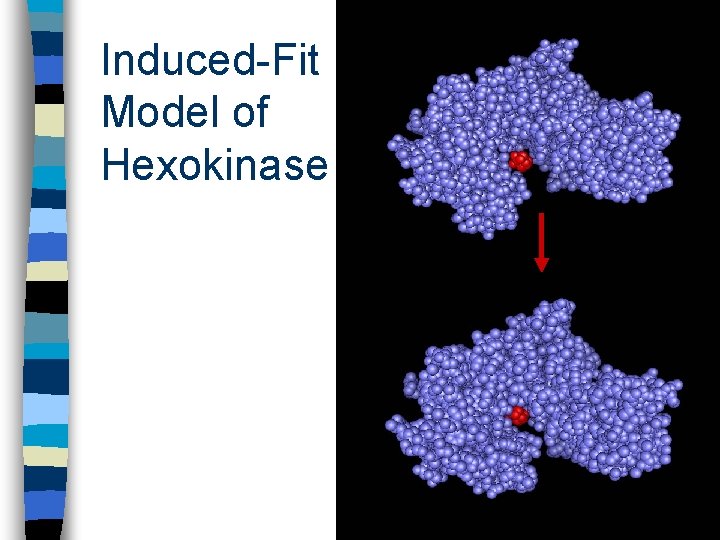
Induced-Fit Model of Hexokinase

Factors Affecting Enzyme Activity n Temperature - causes denaturation of the secondary & tertiary structures of the enzyme, hence changing the shape of the active site, therefore destroying enzymatic action n p. H - same as above n Salinity - same as above
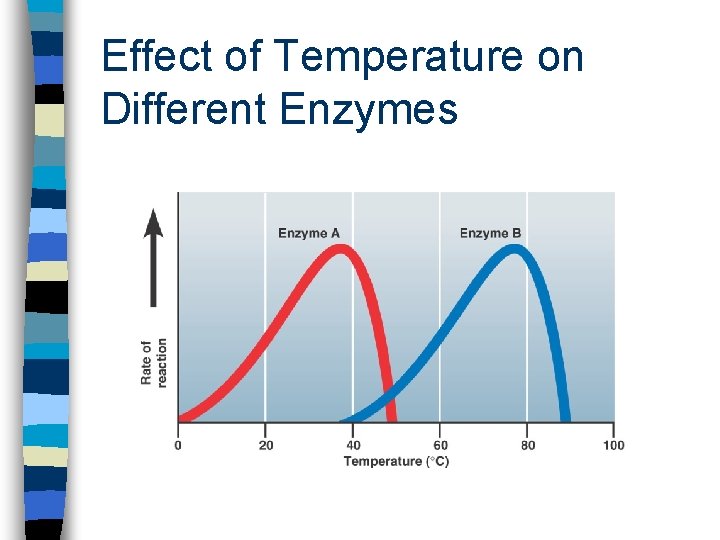
Effect of Temperature on Different Enzymes

Allosteric Control n allo - different n steric - structure n enzymes can be activated or inhibited when a specific substance combines with them at a site other than the active site or within the active site
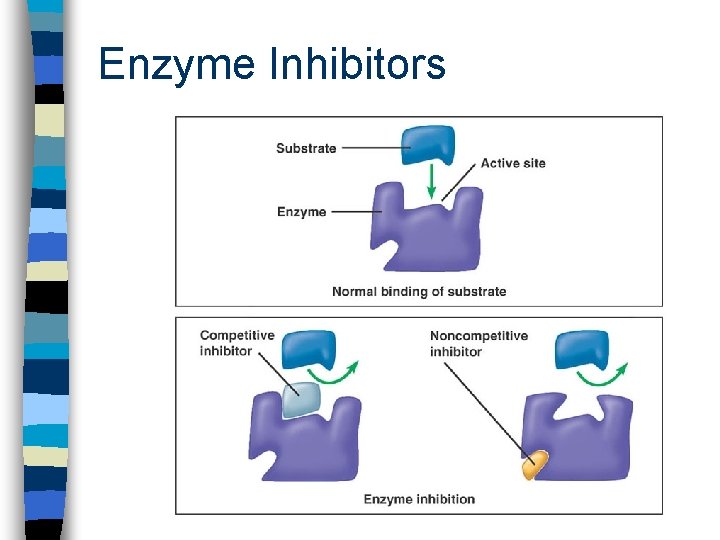
Enzyme Inhibitors

Feedback Inhibition n When an end product accumulates, some of the excess product binds to an enzyme molecule, hence acting as an allosteric inhibitor and therefore blocking the production of more product

Coenzymes n. A coenzyme can alter the shape of the enzyme’s active site allowing a better fit with its substrate n they can also serve as transfer agents of atoms, electrons, H+ ions or functional groups.

Nucleic Acids
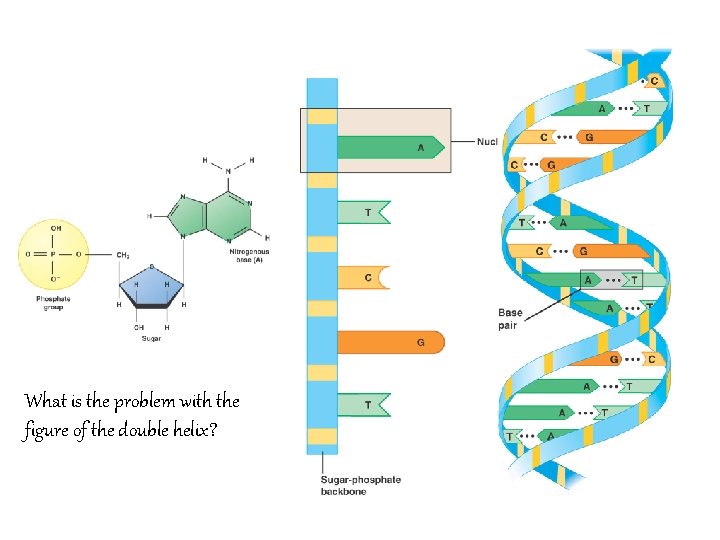
What is the problem with the figure of the double helix?
- Slides: 80