Biological Macromolecules Carbohydrates Lipids Proteins Nucleic Acids Carbohydrates

Biological Macromolecules Carbohydrates Lipids Proteins Nucleic Acids


Carbohydrates C: H: O 1: 2: 1

Carbohydrate Functions • • Short-term energy. Cell identification. Cell communication. Structural materials.
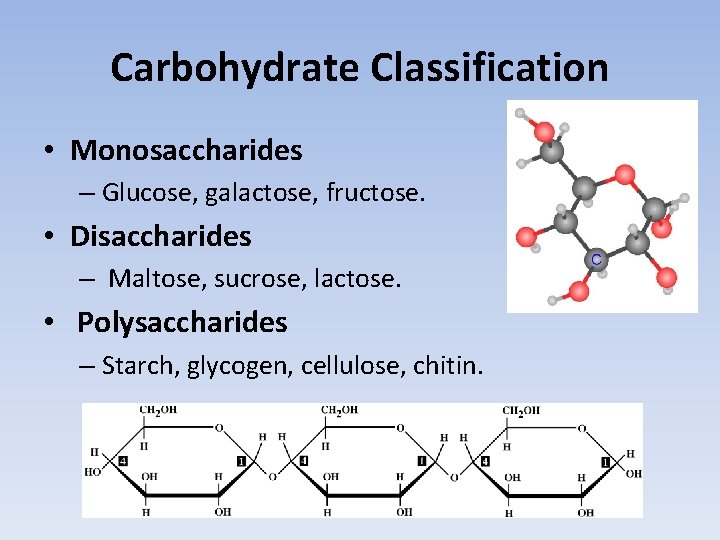
Carbohydrate Classification • Monosaccharides – Glucose, galactose, fructose. • Disaccharides – Maltose, sucrose, lactose. • Polysaccharides – Starch, glycogen, cellulose, chitin.
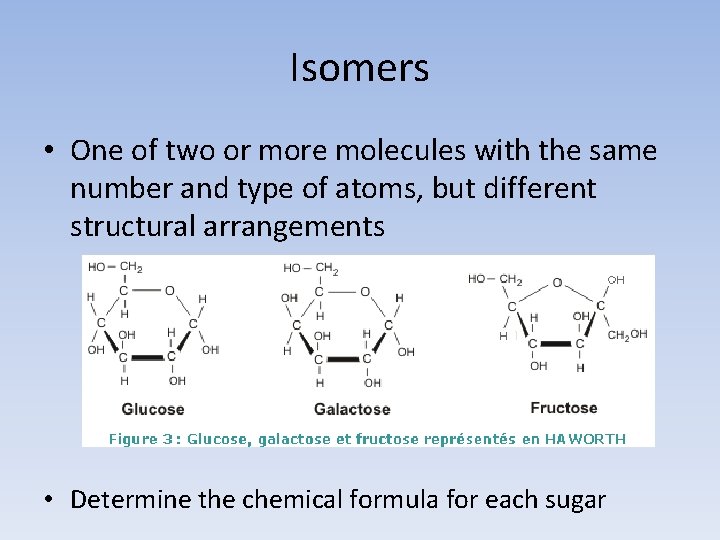
Isomers • One of two or more molecules with the same number and type of atoms, but different structural arrangements • Determine the chemical formula for each sugar
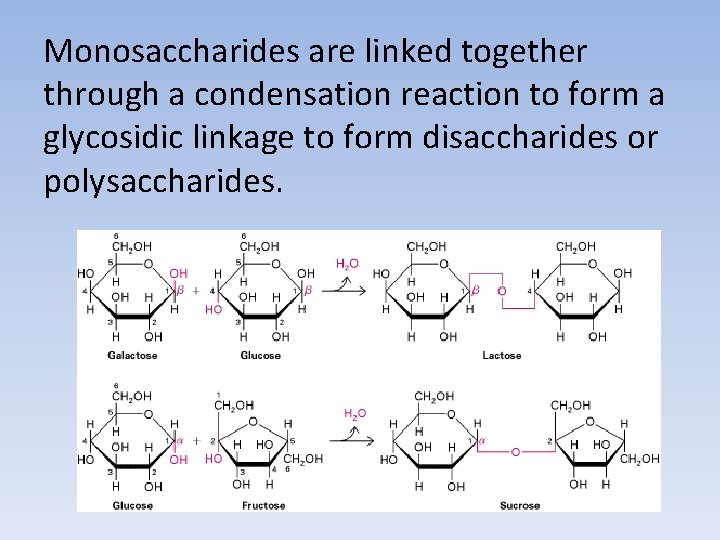
Monosaccharides are linked together through a condensation reaction to form a glycosidic linkage to form disaccharides or polysaccharides.
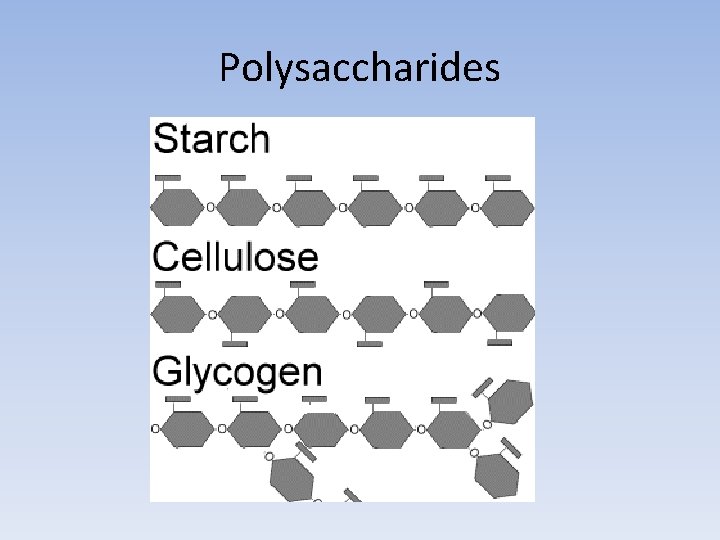
Polysaccharides
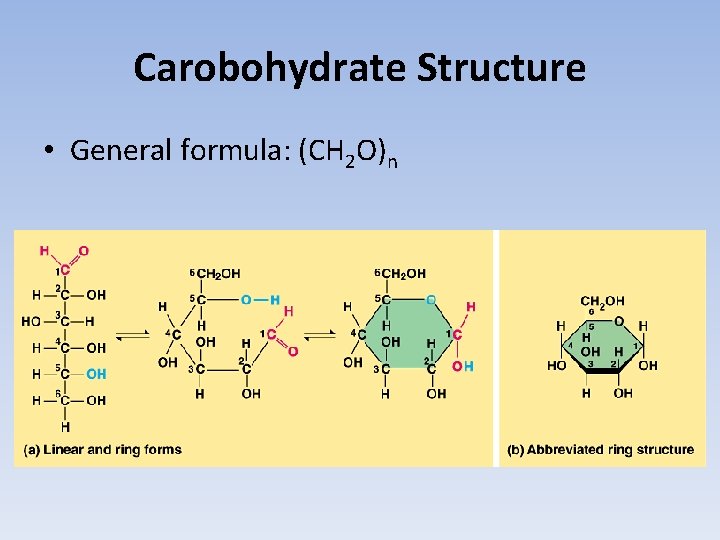
Carobohydrate Structure • General formula: (CH 2 O)n

Lipids Generally nonpolar, hydrophobic molecules. Made up of C, O, H (but fewer O atoms than carbohydrates)

Types of Lipids Type of Lipid Role in Body Triglycerides Energy Storage Phospholipids Steroids Main component of cell membranes Component of cell membranes (cholesterol) and precursors to other steroids and hormones Protection against water loss and insects (plants) Protection against water on surfaces of animals Waxes
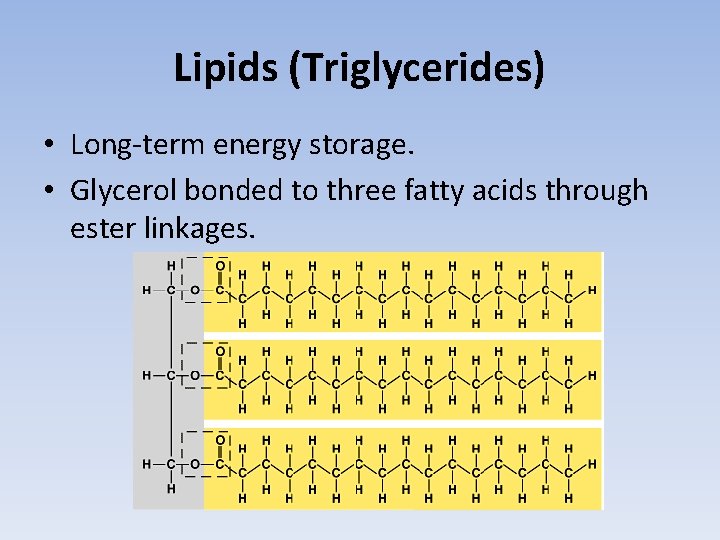
Lipids (Triglycerides) • Long-term energy storage. • Glycerol bonded to three fatty acids through ester linkages.
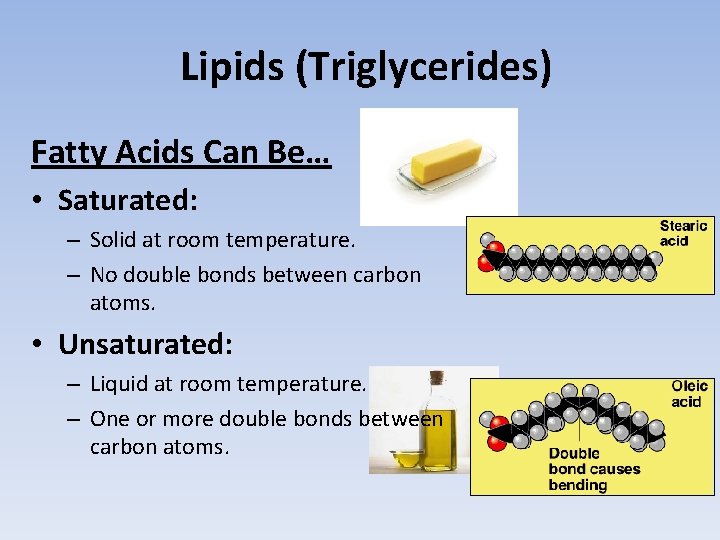
Lipids (Triglycerides) Fatty Acids Can Be… • Saturated: – Solid at room temperature. – No double bonds between carbon atoms. • Unsaturated: – Liquid at room temperature. – One or more double bonds between carbon atoms.
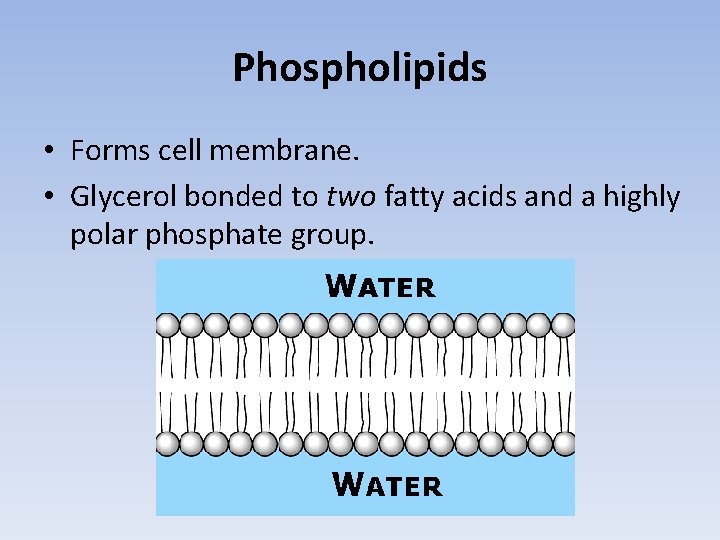
Phospholipids • Forms cell membrane. • Glycerol bonded to two fatty acids and a highly polar phosphate group.
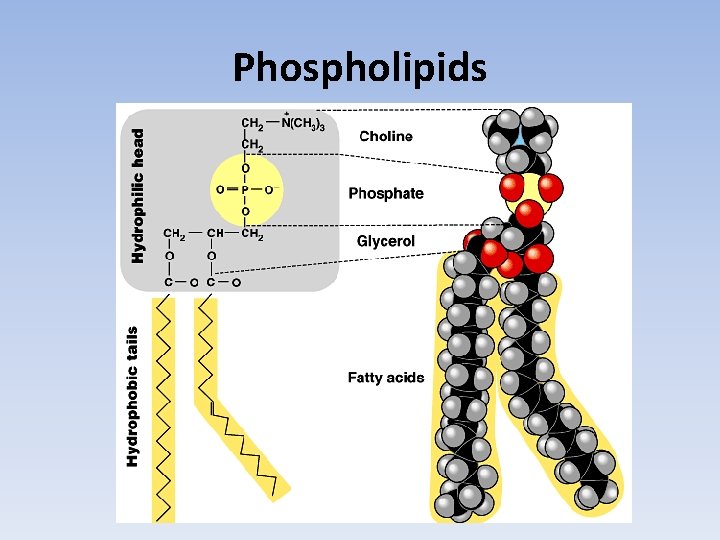
Phospholipids
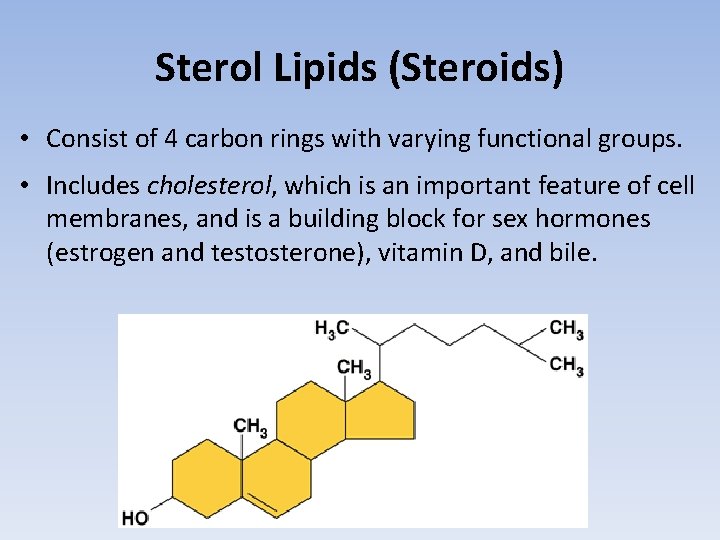
Sterol Lipids (Steroids) • Consist of 4 carbon rings with varying functional groups. • Includes cholesterol, which is an important feature of cell membranes, and is a building block for sex hormones (estrogen and testosterone), vitamin D, and bile.

Waxes • Another type of lipid that is made up of a long chain fatty acid bonded to a long chain alcohol. • Form protective coverings for plants and animals.

Proteins Amino Acid Polymers.

Protein Functions • • • Structural support. Transport of substances. Cell signaling. Movement. Coordination & regulation of activities. Accelerating chemical reactions (as enzymes).
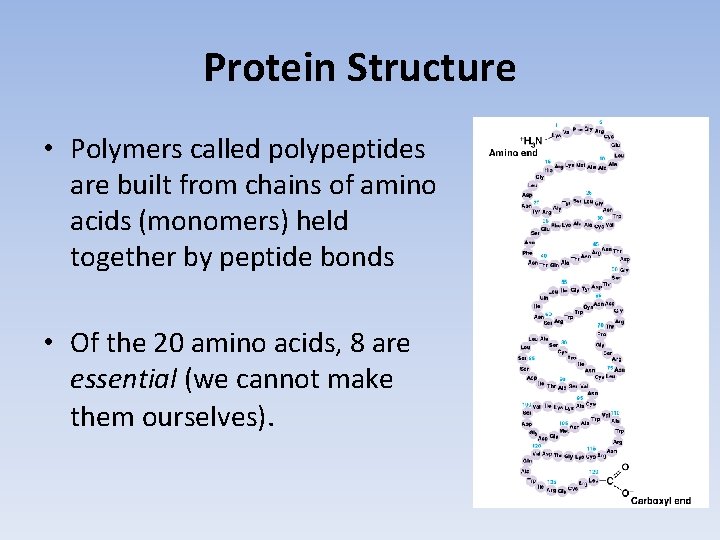
Protein Structure • Polymers called polypeptides are built from chains of amino acids (monomers) held together by peptide bonds • Of the 20 amino acids, 8 are essential (we cannot make them ourselves).
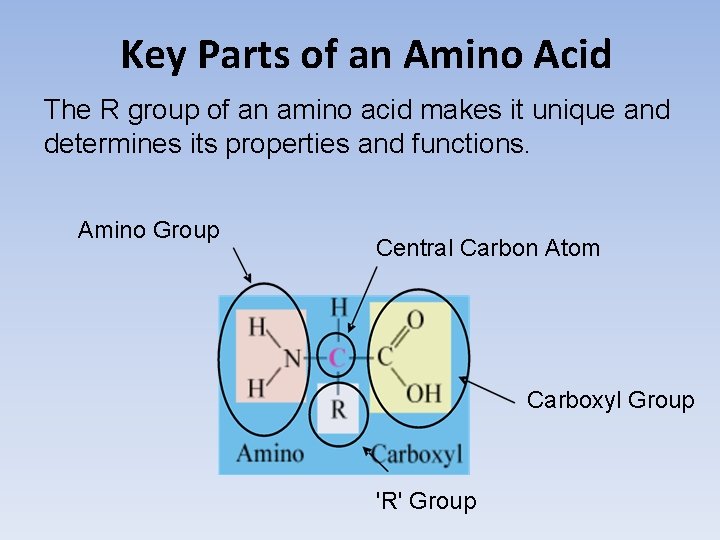
Key Parts of an Amino Acid The R group of an amino acid makes it unique and determines its properties and functions. Amino Group Central Carbon Atom Carboxyl Group 'R' Group
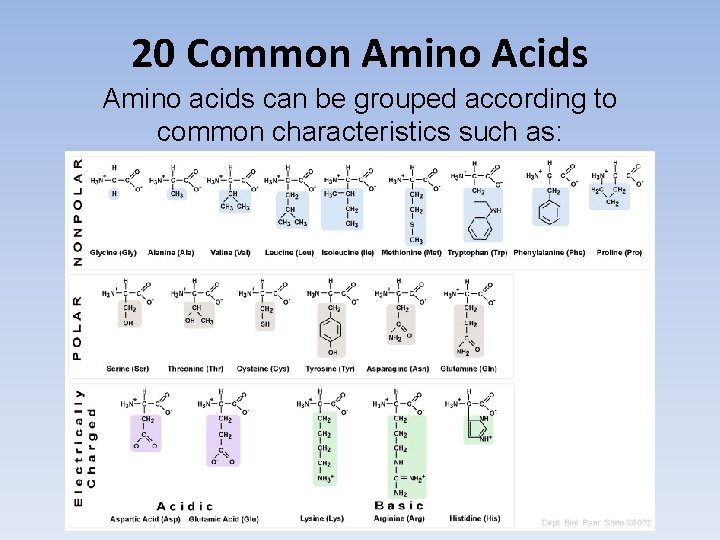
20 Common Amino Acids Amino acids can be grouped according to common characteristics such as:

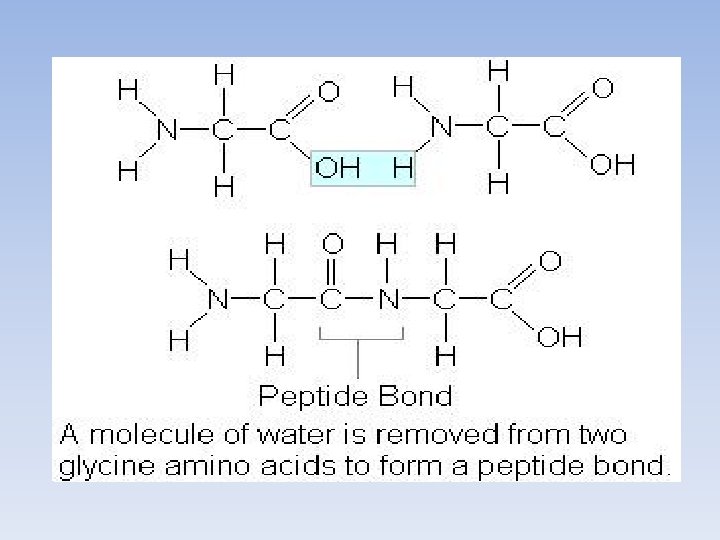
Building Polypeptides • In order to build a protein, amino acids need to be connected together • This is done through dehydration synthesis between the carboxyl and amino group of separate amino acids • The bond that is formed is called a peptide bond.

Protein Structure The Four Levels of Protein Folding • Primary Structure: The sequence of amino acids in a polypeptide chain, which is determined by the nucleotide sequence of a particular gene. • Secondary Structure: The folding and coiling due to the polar nature of the polypeptide chain as it grows. It can form a coil like α-helix, or a fan shaped β-pleated sheet • Tertiary Structure: The polypeptide chain undergoes additional folding due to side chain interactions, primarily the hydrophobic effect. • Quaternary Structure: Two or more polypeptide chains come together, such as in collagen and hemoglobin.
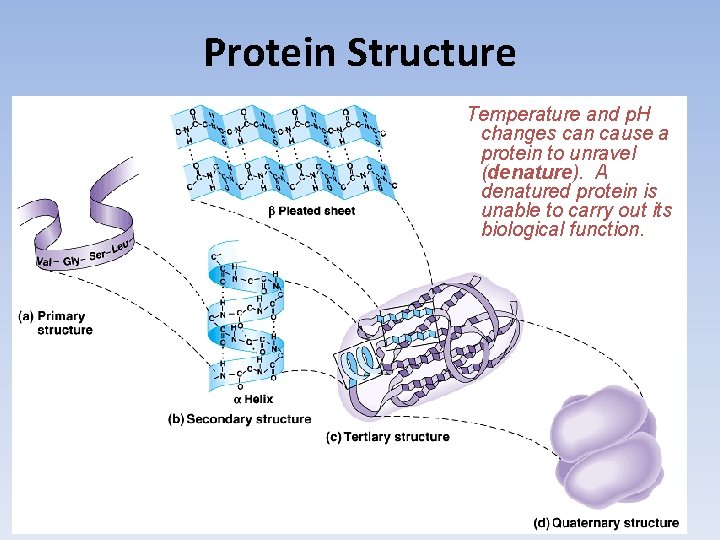
Protein Structure Temperature and p. H changes can cause a protein to unravel (denature). A denatured protein is unable to carry out its biological function.

Nucleic Acids (aka Nucleotides)
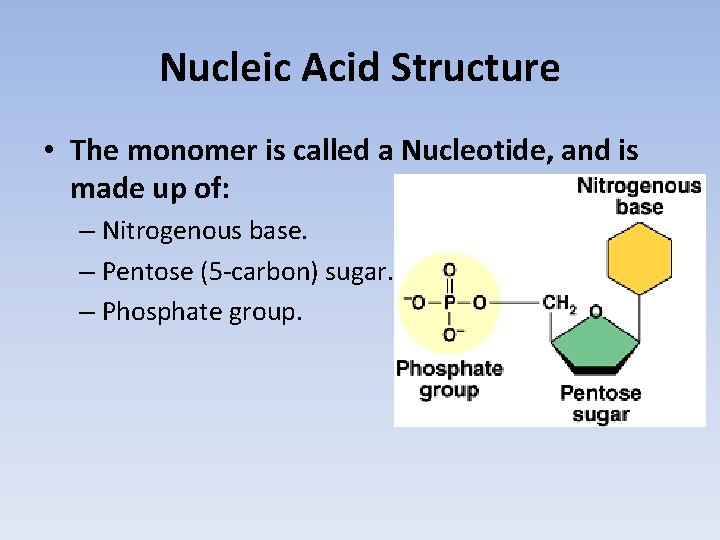
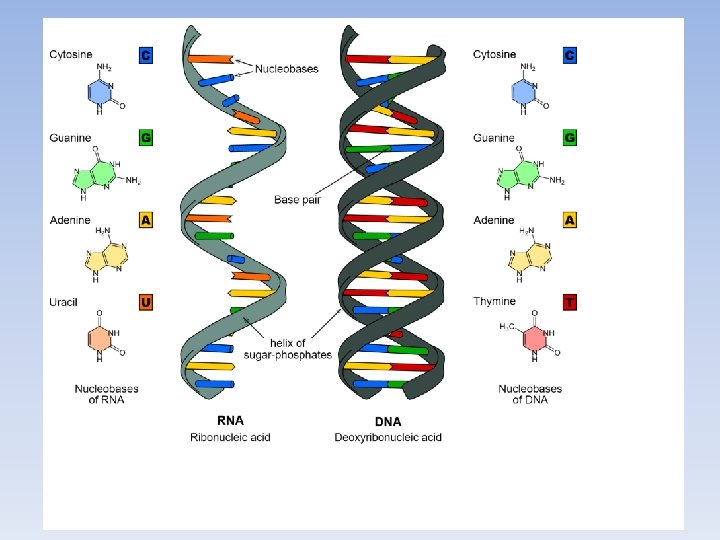
Nucleic Acid Structure • The monomer is called a Nucleotide, and is made up of: – Nitrogenous base. – Pentose (5 -carbon) sugar. – Phosphate group.
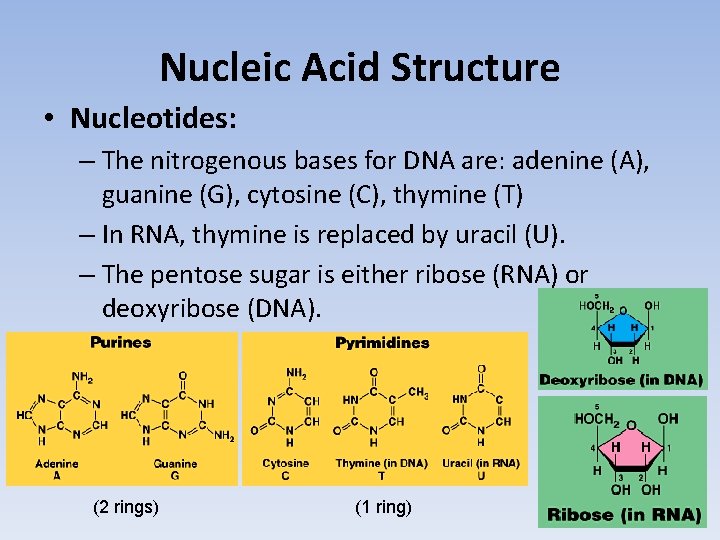
Nucleic Acid Structure • Nucleotides: – The nitrogenous bases for DNA are: adenine (A), guanine (G), cytosine (C), thymine (T) – In RNA, thymine is replaced by uracil (U). – The pentose sugar is either ribose (RNA) or deoxyribose (DNA). (2 rings) (1 ring)

Nucleotide Strands A polymer of nucleotides is often referred to as a strand The phosphate group on one nucleotide often interacts with the hydroxyl group on the sugar of the next nucleotide This forms a phosphodiester bond
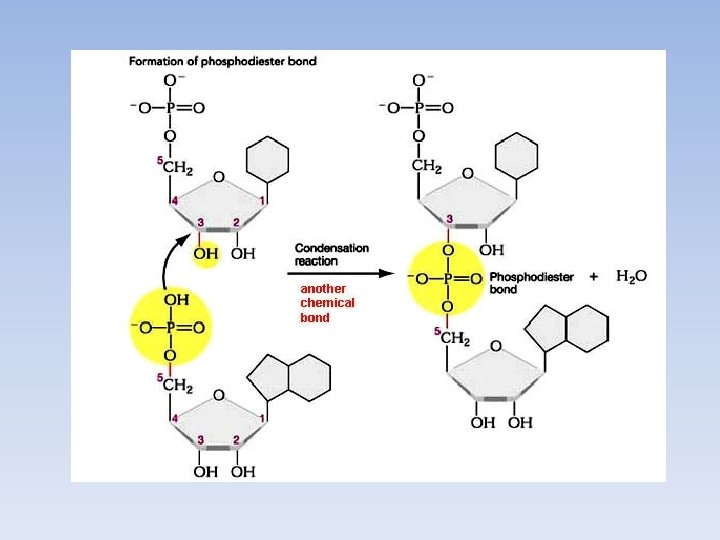
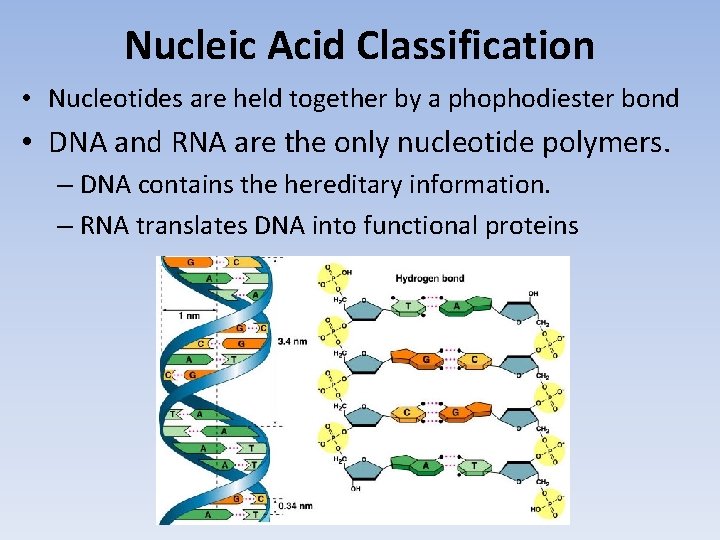
Nucleic Acid Classification • Nucleotides are held together by a phophodiester bond • DNA and RNA are the only nucleotide polymers. – DNA contains the hereditary information. – RNA translates DNA into functional proteins
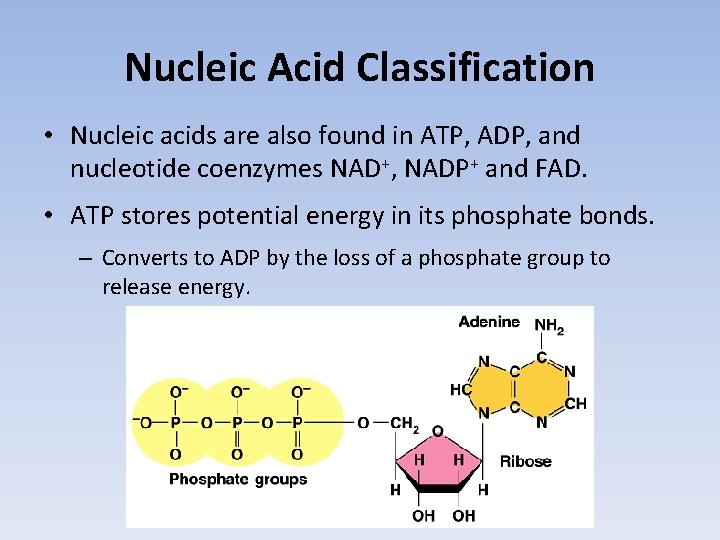
Nucleic Acid Classification • Nucleic acids are also found in ATP, ADP, and nucleotide coenzymes NAD+, NADP+ and FAD. • ATP stores potential energy in its phosphate bonds. – Converts to ADP by the loss of a phosphate group to release energy.
- Slides: 34