Biological Exposure Indices BEIs Process and Use Larry

Biological Exposure Indices ® (BEIs ) Process and Use Larry K. Lowry, Ph. D. Chair, ACGIH® BEI® Committee The University of Texas Health Center at Tyler

Where are we going today? • • Current definitions of the BEI®, 2002 The development of BEIs® The key – Documentation Examples Biomonitoring without limits Current and future issues Resources

Biological monitoring. Why? • Assess exposure and uptake by all routes – TLV® not protective – skin – Includes workload – More closely related to systemic effects • Assess effectiveness of PPE • Legal or ethical drivers – Regulations – Control workers’ compensation costs

“Guidelines” for biological monitoring – ® The BEIs

® The BEIs – 2003 BEIs® are intended for use in the practice of industrial hygiene as guidelines or recommendations to assist in the control of potential workplace health hazards and for no other use.

® The BEI – Definition • Biological monitoring … entails measurement of the concentration of a chemical determinant in the biological media of the exposed and is an indicator of the uptake of the substance. • The BEI® determinant can be the chemical itself; one or more metabolites; or a characteristic reversible biochemical change induced by the chemical.

® BEIs • Represent levels of determinants that are most likely to be observed in specimens collected from a healthy worker who has been exposed to chemicals to the same extent as a worker with inhalation exposure to the TLV®-TWA. • Generally indicate a concentration below which nearly all workers should not experience adverse health effects.

® Current basis for BEIs ® • Bio-equivalent to TLV (traditional) – “BEIs® represent levels of determinants that are most likely to be observed in specimens collected from a healthy worker who has been exposed to chemicals to the same extent as a worker with inhalation exposure to the TLV®-TWA. ” • Most of the BEIs® are based on TLVs®

Current basis • Indicators of early, reversible health effect – Approach developed in late 80’s as relationships did not always exist between airborne exposure and biomonitoring determinant. • Examples: – CO, Acetyl cholinesterase inhibiting pesticides, Cd, Pb, Hg, Hexane-Mn. BK

® The BEI Committee Larry Lowry, Ph. D. , U TX Health Center at Tyler – Chair • • • Phil Edelman, MD, CDC – Vice Chair Mike Morgan, Sc. D, CIH, U. of WA – Past Chair Joe Saady, Ph. D. , VA Division of Forensic Science Leena Nylander-French, Ph. D, CIH, UNC, Chapel Hill John Cocker, Ph. D. , HSE, UK K. H. Schaller, Dipl. Ing. , Univ Erlangen, Germany M. Ikeda, Ph. D. , Kyoto Ind Health Assoc, Japan Gary Spies, CIH, Pharmacia Glenn Talaska, Ph. D. , CIH, Univ of Cincinnati Jan Yager, Ph. D. , EPRI

® BEI development • Volunteer assigned document • Prepares draft Documentation • Sources of data – Human laboratory & workplace data • Limited use of animal data – Simulation modeling with verification – Published peer-reviewed data • Draft Documentation discussed in committee meetings, e-mail
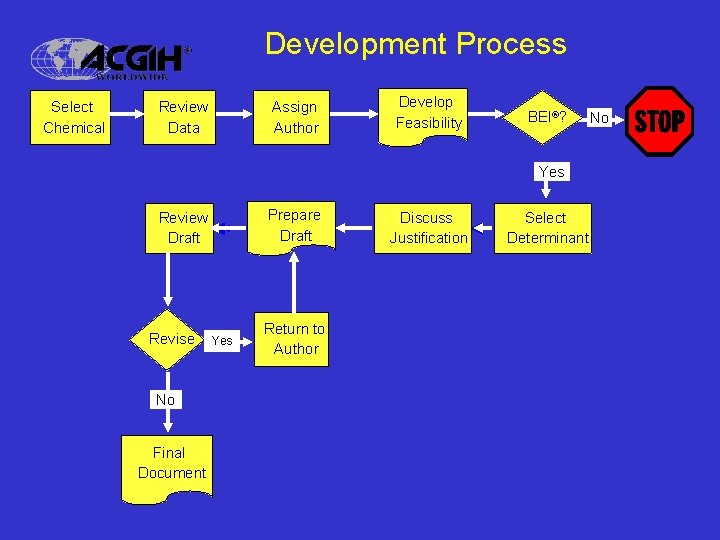
Development Process Select Chemical Review Data Assign Author Develop Feasibility BEI®? Yes Prepare Draft Review Draft Revise No Final Document Yes Return to Author Discuss Justification Select Determinant No

How are chemicals selected? • • • Chemicals with human data Potential for dermal absorption Availability of adequate lab methods Recommendations by others Interest/experience of committee member

The Documentation • Who is the audience? – The practicing occupational hygienist or other practicing occupational health professional • What the Documentation is – Justification supporting the BEI® – Practical information on sampling, background, etc. • What the Documentation is not – An extensive review of toxicological data – A novel research approach to setting guidelines

The Documentation – contents Basis of the BEI® Uses and properties Absorption Elimination Metabolic pathways & biochemical interactions • Possible non-occupational exposure • Summary of toxicology • • •

For each index ® or BEI • • Analytical methods, sampling, and storage Levels without occupational exposure Kinetics Factors affecting interpretation – Analytical procedures and sampling – Exposure – Population • Justification – the key • Current quality of database • Recommendations and references

The notations • B - Background levels expected • Nq- Nonquantitative – Biol. monitoring recommended, no BEI® • Ns- Non-Specific – Needs confirmation • Sq Semiquantitative (but specific) – Screening test – Confirmatory tests

Practical applications • Bioavailability of metals – Chromium VI (water soluble) fume • Specificity and Sensitivity – Benzene biomonitoring – t, t-Muconic acid in urine (t, t-MA) – S-Phenylmercapturic acid in urine (SPMA)

Bioavailability of metals – Chromium • Physical properties and solubility – Cr (III), very insoluble particulates – Cr (VI) insoluble particulate – the lung carcinogen – Cr (VI) water soluble • Fume as generated in MMA arc welding • Mist as generated in electroplating • Health effects of Cr (VI) water soluble – Fume – lung irritant – Mist – chrome ulcers on skin, mucus membranes

Biological monitoring of Cr exposure • Cr (III) inappropriate – not bioavailable • Cr (VI) insoluble – not bioavailable • Cr (VI) water soluble – If fume, use BEI® based on welding studies – If mist, bioavailability less • See chrome ulcers at “acceptable” BEI® values
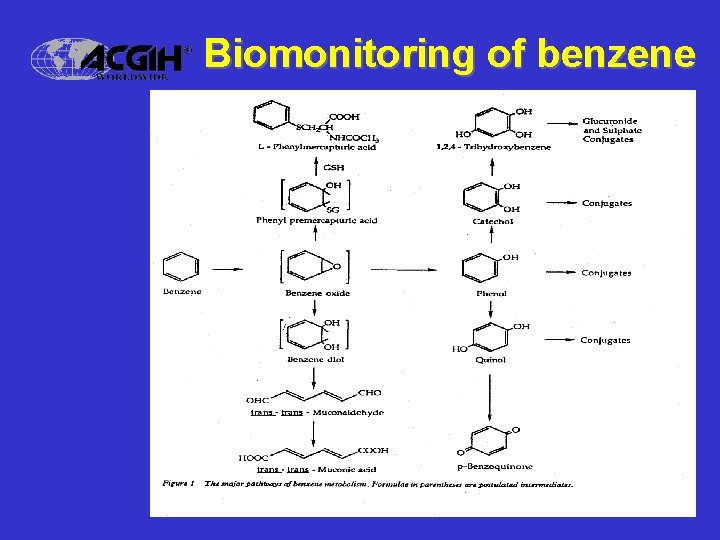
Biomonitoring of benzene

Biomonitoring at ® the current TLV • t, t-Muconic acid in urine (t, t-MA) – Good sensitivity (to 0. 1 ppm benzene) – HPLC methodology – Considerable variability in populations • S-Phenylmercapturic acid in urine (SPMA) – Ultimate sensitivity (to 0. 01 ppm benzene) – GC/MS methodology – Good data base, but expensive

Biological monitoring without limits • What about substances absorbed through the skin and with chronic systemic health effects that occur after a long lag time such as cancer?

The traditional approach • Cannot relate to airborne limits, TLVs® – Irrelevant • Cannot relate to skin absorption – Difficult to quantitate dermal dose • Cannot relate to health effect – Often wrong timeline • What to do?

® The BEI approach • Rationale – Biological monitoring is essential to assess dermal exposure – How do you correlate dermal dose with a biomarker of exposure? • Nq Approach – “Biological monitoring should be considered for this compound based on the review; however, a specific BEI® could not be determined due to insufficient data. ”

Criteria for an Nq • Dermal route of exposure significant • Good measurement methods • Good qualitative data on human exposure and biomarker concentration • Poor quantitative data relating exposure & biomarker • Long lag time, exposure to health outcome • Low or no background in general population

If criteria are met, then • • Develop full Documentation Describe sampling and analysis Define background levels Describe justification for biomonitoring Note the lack of quantitative data Cite guidance values from literature Publish BEI® as Nq (no value)

Examples – MBOCA • Principal route of exposure – dermal • Alleged health effect in humans – cancer • Good methods and human data on exposure-response • Industry practice guidance from the HSE

Health and Safety Executive, UK • Scientific basis to justify guidance values • Use "yardstick or benchmark" approach • Issues – Results – no "safe" or "unsafe" exposure levels – Results – estimates of exposure areas and allow intervention to reduce exposures – No legal status • Examples – MBOCA and MDA

The “yardstick or benchmark” approach • Good analytical methods • All specimens analyzed by one laboratory or with a single method • Establish "best industry practice" using an upper 90% confidence limit of the "best" industries • Benchmarks – guidance value to provide users with assessment of their results

Current issues • Carcinogens? – Is there a safe level of exposure? – The German EKA approach • Mixtures and interactions – Metabolism/toxicokinetics on pure chemical – Workers exposed to mixtures – How does this effect BEI®? • Biomarkers of effect – irreversible effects? • Data gaps – lack of human data • Animal data – should this be used?

Skin absorption Justification for ® BEI – Existing BEIs® for substances with substantial skin absorption • MBOCA – Nq • EGME/EGMEA – Nq • EGEE/EGEEA – 100 mg/g creatinine – (based on TLV® of 5 ppm) – Is this a valid approach? • Are Nq notations appropriate? • Should a chemical without a “skin” notation have a BEI®?

The future • As TLVs® drop, BEIs® based on TLVs® drop – Cannot distinguish exposure at TLV® from background • What do we do for substances that have no human data? • What is the future of modeling techniques? – Can these modeling techniques be validated? • Should animal data be used? • What about mixtures?

Other guidelines

Germany The BATs from the DFG

The HSE – UK Biological monitoring guidelines

Guidance from WHO – How to do biological monitoring

Other Guidelines New edition, 2001

Thank you for your attention Your questions please

Scheduled Break Take a minute to stretch!
- Slides: 40